Abstract
In the present study, a chromogenic in situ hybridization for the identification of Giardia duodenalis in paraffin-embedded tissue samples was developed. The sensitivity and specificity of the probe was validated by testing it on cultured reference samples of different assemblages of G. duodenalis as well as culture and tissue samples containing other protozoa and infectious agents. The probe gave a positive reaction with the Giardia samples and a negative reaction with all other samples. Further, the probe was used for screening of histological slides of intestine from different animal species (99 canine samples, 85 feline samples, and 202 porcine samples) for the presence of G. duodenalis trophozoites. With this assay, the parasites were detected in samples from 8 dogs (8.08%), 6 cats (7.06%), and zero pigs. The results clearly indicate that the described method is useful for detection of Giardia trophozoites in routinely processed intestinal tissue of different animal species.
Introduction
Giardia is the most common enteric protozoan pathogen of human beings, domestic animals, and wildlife. 19 Taxonomy of this genus is still a matter of uncertainty. Currently, there are 6 recognized species (G. duodenalis [syn. G. intestinals, G. lamblia], G. agilis, G. muris, G. ardeae, G. psittaci, and G. microti). Giardia duodenalis, which is known to infect a broad variety of species, has been further divided into 7 genetic assemblages (A–G). A recent study 18 suggested recognizing the different assemblages as separate species. As this proposition has not yet been unanimously accepted, the traditional, yet unsatisfactory, assemblage concept will be used in the present paper. Although such an important pathogen, both in human beings and animals, the recognition of the trophozoites in histological tissue sections is challenging, because the parasites do not clearly stand out from sloughed enterocytes, cell debris, or ingesta. Specific labeling techniques, such as immunohistochemistry (IHC), 2,23 fluorescent antibody test, 10 or fluorescent in situ hybridization (FISH) 5,7,9,16 have been described but rarely used, due to disadvantages inherent in each of the applied techniques. Immunohistochemistry is dependent on the availability of good quality and specific primary antibodies, and the fluorescent-based methods are hampered by the fact that they need specific microscopic equipment and that the morphology of the tissue cannot be adequately evaluated simultaneously.
The present study proposes to apply chromogenic in situ hybridization (CISH), which has already been successfully established for other protozoa, for the specific identification of G. duodenalis in intestinal tissue and biopsy samples. Based on positive experience with other protozoa, 6,15,20,24 a part of the 18S (small subunit) ribosomal (r)RNA gene was chosen as target for the probe design. The assay was validated and optimized on paraffin-embedded samples of cultured G. duodenalis. A number of canine, feline, and porcine intestinal samples were screened with the probe in order to evaluate the usefulness of the technique in routinely embedded tissue samples and to show the prevalence of Giardia infections in different animal species based on trophozoite detection in tissue samples.
Materials and methods
Probe design
An ISH probe for the detection of all genetic assemblages of G. duodenalis was designed after extensive homology studies using the Sci Ed Central software package a on all available GenBank sequences of the 18S rRNA gene of G. duodenalis. A region of 37 bp with complete identity among all 7 genetic assemblages was chosen as probe sequence. The selected probe sequence was 5’-CCTCGGCTCCGCGGCGGGGGCTGTGGGCCCGCCTCGC-3’. Subsequently, this sequence was submitted to Basic Local Alignment Search Tool (BLAST; http://blast.ncbi.nlm.nih.gov/Blast.cgi) to search against GenBank sequences and to exclude unintentional cross-reactivity with other organisms. The probe sequence was sent to Eurofins MWG Operon b for probe synthesis and labeling of the 3’-end with digoxigenin. Subsequently, ISH was carried out using the newly designed probe on different protozoal cultures and tissue samples.
In situ hybridization
Chromogenic ISH was performed according to a previously described protocol. 6 Briefly, 3-µm paraffin-embedded sections were deparaffinized then rehydrated. For proteolysis, the slides were treated with proteinase K c (2.5 µg/ml) in Tris-buffered saline for 30 min at 37°C. Afterward, the slides were rinsed in distilled water, dehydrated in alcohol (95% and 100%), and air dried. The slides were incubated overnight at 40°C with a hybridization mixture, 100 µl of which were composed of 50 µl of formamide, d 20 µl of 20× standard sodium citrate (SSC), 10 µl of dextran sulfate e (50% w/v), 12 µl of distilled water, 5 µl of boiled herring sperm DNA f (50 mg/ml), 2 µl of 50× Denhardt solution, e and 1 µl of Giardia probe at a concentration of 10 ng/ml. On the second day, the slides were washed with decreasing concentrations of SSC (2×, 1×, 0.1×; 10 min each) to remove non-hybridized probe. Afterward, the slides were incubated with anti-digoxigenin–alkaline phosphatase Fab fragments c (1:200) for 1 hr at room temperature. Visualization of the hybridized probe was carried out after an additional washing step using the color substrates 5-bromo-4-chloro-3-inodyl phosphate c and 4-nitro blue tetrazolium chloride. c Color development was stopped with Tris–ethylenediamine tetra-acetic acid buffer (pH 8.0) after 1-hr incubation. The slides were counterstained with hematoxylin and mounted under coverslips using synthetic aqueous mounting medium. g
Positive and negative control samples
Five protozoal cultures embedded in paraffin wax, representing 3 genetic lineages of G. duodenalis (assemblage A: source human; assemblage B: sources dog and chinchilla; assemblage E: sources sheep and pig), h were used as positive controls for the designed ISH probe. Cultures containing Monocercomonas colubrorum, 24 Pentatrichomonas hominis (GenBank accession no. AY349187), 12 Trichomonas gallinae, Tetratrichomonas gallinarum, 15 Tritrichomonas foetus, 25 and Tritrichomonas augusta (GenBank accession no. AY055802), 12,25,h,i and formalin-fixed and paraffin-embedded tissue samples containing Histomonas meleagridis, Trichomonas gallinae, Entamoeba invadens, and Cryptosporidium baileyi served as negative controls. All protozoal cultures containing an undefined number of parasites were fixed in 7% buffered formalin and embedded in paraffin wax. Prior to embedding, all cultures were soaked with rice starch (3.3 mg/ml) for 5 hr and centrifuged at 6,000 × g for 10 min to produce a pellet. The pellet was overlaid with agar, hardened at 4°C, carefully removed from the tube, and embedded in paraffin wax.
In addition, formalin-fixed and paraffin-embedded intestinal samples from 3 different animal species (rabbit [Oryctolagus cuniculus], chinchilla [Chinchilla lanigera], degu [Octodon degus]) with coproscopically diagnosed Giardia infections were used to analyze the functionality of the designed Giardia probe directly in tissue. The samples were obtained from the archive of the Institute of Pathology and Forensic Veterinary Medicine (Vienna, Austria) and were chosen after examination of hematoxylin and eosin–stained histological sections had revealed protozoa morphologically consistent with Giardia. Additionally, to rule out nonspecific hybridization, all protozoal culture and tissue samples containing Giardia were tested with an irrelevant probe specific for Cryptosporidium spp. 6
Test samples
The probe was used as a screening tool for the detection of Giardia trophozoites in a retrospective study of paraffin-embedded archived intestinal tissues from dogs, cats, and pigs.
Canine and feline samples
Selection criteria were between 8 weeks and 12 months of age with an anamnesis of diarrhea. Both necropsy samples and duodenal biopsy samples were included. A total of 99 canine tissue samples from the small intestine (mostly jejunum, in some cases also duodenum or ileum; 91 necropsy cases, 8 biopsies) and 85 feline samples from the small intestine (82 necropsy cases, 3 biopsies) were included. In 58 dogs (58.6%) and 40 cats (47%), typical macroscopic and histological lesions of parvovirus enteritis or panleukopenia were present.
Porcine samples
Paraffin-embedded samples from jejunum of 202 pigs (weight range: 4–113 kg; majority between 10 and 30 kg), which had been investigated for different Brachyspira species in the course of another project, were used. 13 The pigs had been affected with miscellaneous chronic diseases, among which diarrhea, wasting, and respiratory problems were the most common.
All samples were tested with the CISH procedure described above. In each run, a positive control (paraffin-embedded Chinchilla sample) was included. The evaluation of the slides was done by light microscopy and the quantity of the parasites was assessed using the following score system: 0: no parasites present in the entire section; 1: an average of 1–5 parasites per 10× field, assessed in 10 randomly chosen fields; 2: an average of 6–30 parasites per 10× field, assessed in 10 randomly chosen fields; 3: more than 30 parasites per 10× field, assessed in 10 randomly chosen fields.
Results
In silico analysis of the probe
The probe was checked for homologies with other Giardia species using the Align plus tool from the Sci Ed software package. a There was 1 nucleotide (nt) mismatch with G. psittaci (AF473853), 2 mismatches with G. microti (AF006676), and more than 10 mismatches including a 7 nt insertion with G. ardeae (Z17210). Sequences containing the region of interest were not available from G. muris and G. agilis. The BLAST search revealed no close similarities with other microbial sequences.
Evaluation of the ISH assay
In the paraffin-embedded Giardia culture samples, the parasites were clearly identified by their dark purple coloration (Fig. 1A). The specificity of the staining was proven by the morphological characteristics of the labeled Giardia trophozoites. Paraffin-embedded culture samples or tissue samples containing other protozoa showed no signals indicating that the probe had no cross-reactivity with other protozoa with potential morphological similarity (Fig. 1B–C). Routinely paraffin-embedded intestinal samples of animals with prediagnosed Giardia infection showed clear labeling of Giardia trophozoites attached to the intestinal villi or within the intestinal contents displaying the characteristic morphological features of this parasite. Hybridization with the Cryptosporidium probe produced no signal (Fig. 1D). These investigations proved that the Giardia probe was well suited for identification of Giardia trophozoites in routinely fixed and embedded intestinal samples.
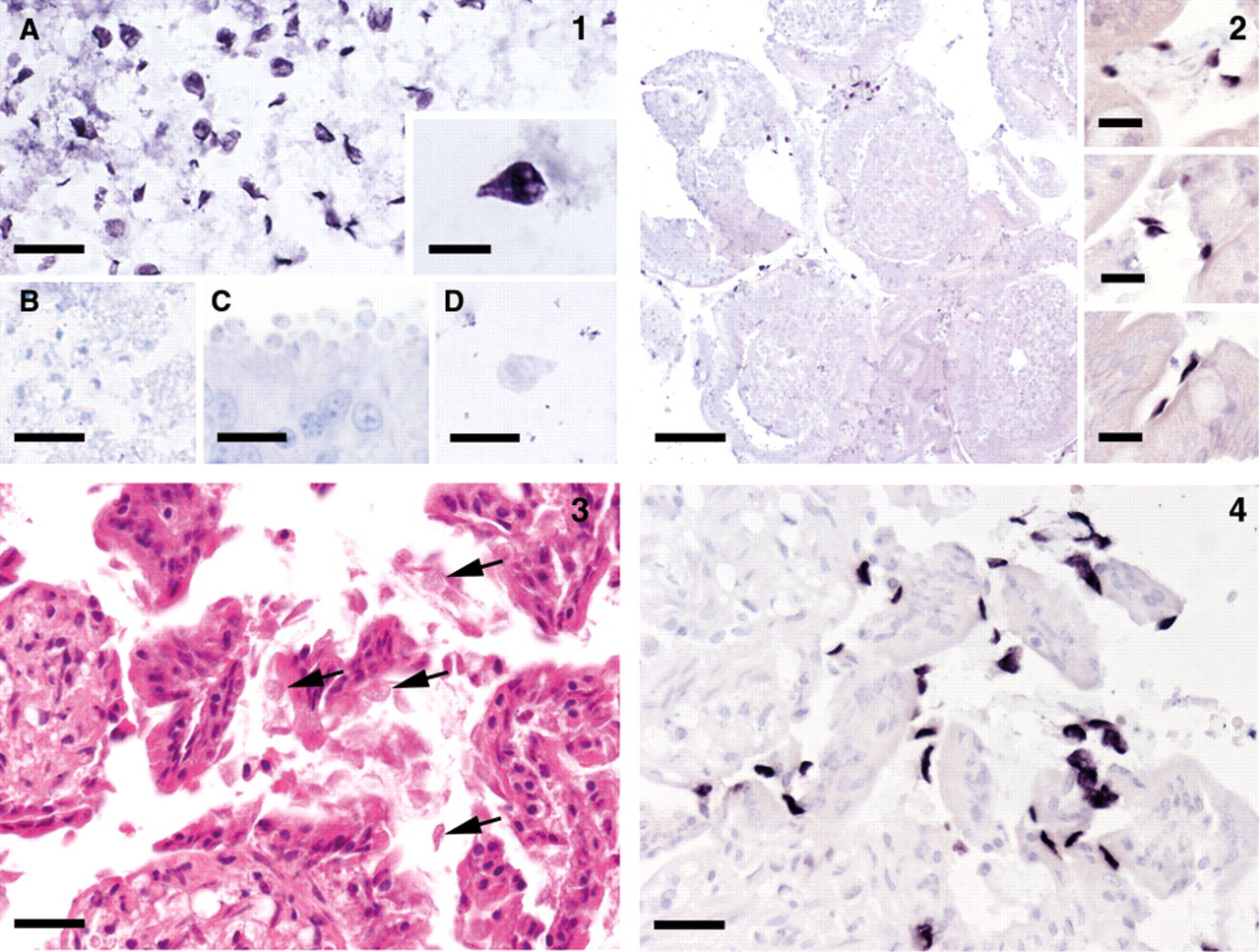
Specificity tests of the Giardia in situ hybridization on paraffin-embedded parasite cultures and tissue sections.
Canine tissue samples
In 8 samples (7 necropsy cases, 1 biopsy; 8.08%), Giardia parasites could be found, which were mostly attached to the mucosal villi, but frequently also present within the intestinal lumen (Fig. 2). The quantities were scored as 3 (severe) in 3 cases and 1 (slight) in 5 cases. One of the dogs was affected with canine parvovirus enteritis. In some cases, there was moderate autolysis of the intestinal mucosa; the identification of Giardia, however, was not impaired.
Feline tissue samples
In 6 samples (all necropsy cases; 7.06 %), Giardia parasites were present, which were mostly attached to the villi, but frequently also present within the intestinal lumen (Figs. 3–4). The quantities were scored as 3 (severe) in 4 cases, 2 (moderate) in 1 case, and 1 (slight) in 1 case. Two of the cats were affected with feline panleukopenia. Moderate autolysis of the intestinal mucosa was present in several cases which, however, did not impair the identification of Giardia.
Porcine tissue samples
In none of the porcine tissue samples could Giardia parasites be identified. The results of the ISH in samples of the different animal species investigated are summarized in Table 1.
Results of in situ hybridization for presence of Giardia sp. in the small intestinal samples of dogs, cats, and pigs.

Discussion
In the present investigation, a novel CISH procedure for the detection and identification of G. duodenalis trophozoites in paraffin-embedded tissue sections was developed, validated, and applied. In situ hybridization has been used for the identification of Giardia previously, but mainly in context with identification of cysts in environmental samples and with fluorescently labeled probes. 5,7,10,14 Only infrequently has ISH been applied on pathology specimens. 16 Also the authors of this study worked with FISH technique with the requirement for dark field microscopy and the disadvantage of less morphological information on surrounding tissue alterations compared to bright field microscopy. Chromogenic ISH, on the other hand, combines specific identification of the infectious agent with the possibility for simultaneous evaluation of associated tissue lesions. The technique has been successfully used for the demonstration of various protozoal parasites in pathology specimens, such as Histomonas meleagridis, Entamoeba sp., Cryptosporidium sp., members of the order Trichomonadida, and others. 6,15,20,24 In particular, Giardia trophozoites are difficult to identify in conventionally stained histological slides, as they may easily be confounded with cell debris, secretion products, or ingesta. Also, with Giardia as the target organism, CISH with the developed probe gave clear signals and proved its excellent suitability for specific detection of protozoa in tissue sections. Partial autolysis of the intestinal samples, which is a frequent feature of routine diagnostic submissions, had no negative effect on the outcome of ISH. Also, concurrent necrotizing intestinal infections such as parvovirus enteritis and panleukopenia did not inhibit the detection of Giardia trophozoites.
The prevalence of Giardia infections in animals of the susceptible age groups affected with diarrhea was at the lower end of comparable studies based on detection of Giardia in fecal samples. 3,4,22 It has to be taken into account that, with the exception of biopsies, the samples included in the current study were from lethal cases in which enteritis could be both, main, or additional diagnosis. A considerable number of dogs and cats were affected with parvovirus infection. However, there was no increased frequency of Giardia infections in this group.
A possible problem when using the newly developed technique as a tool for estimating the prevalence of Giardia is the limited examination of only 1 intestinal section. A possible discontinuous distribution of Giardia trophozoites in the intestinal canal could result in a missed infection by using this approach. This disadvantage could be counterbalanced by investigation of several intestinal samples representing various locations of the small intestine. In addition, this method can be recommended as an excellent tool for confirming or excluding a potential Giardia infection in biopsy samples of young animals with intestinal problems, in which this disease represents an important differential diagnosis.
None of the investigated porcine small intestinal samples showed a positive result, which is in line with the very low prevalence demonstrated in a German study, 8 but was in contrast to other investigations that demonstrated Giardia cysts in fecal samples in varying prevalences. 1,11,17,21,26 Taken together, CISH has been proven to be a robust and reliable method for identification and localization of Giardia trophozoites in formalin-fixed, paraffin-embedded tissue samples of different animal species.
Footnotes
Acknowledgements
The authors thank Prof. Jaroslav Kulda, Charles University Prague, and Prof. Michael Hess, University of Veterinary Medicine Vienna, for their generous gifts of cultured samples of Giardia and other protozoa. The invaluable technical support of Karin Fragner and Klaus Bittermann is gratefully acknowledged.
a.
Scientific & Educational Software, Cary, NC.
b.
Eurofins MWG Operon, Ebersberg, Germany.
c.
Roche, Basel, Switzerland.
d.
Fluka, Sigma-Aldrich Chemie GmbH, Buchs, Switzerland.
e.
Sigma-Aldrich, St. Louis, MO.
f.
Invitrogen Corp., Carlsbad, CA.
g.
Aquatex, VWR International, Vienna, Austria.
h.
Culture collection of the Department of Parasitology, Faculty of Science, Charles University, Prague, Czech Republic.
i.
Clinic for Avian, Reptile and Fish Medicine, Department for Farm Animals and Veterinary Public Health, University of Veterinary Medicine, Vienna, Austria.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work received financial support from the Austrian Science Fund (FWF) grant P20926.
