Abstract
Objective
The present study aimed to investigate the metabolic activity and adherence capacity of the whole bone marrow cell (wBMC) population and changes in bone marrow (BM) architecture after acute pancreatitis (AP) induction.
Methods
The disease was induced by injecting L-arginine (350 mg/100 g; i.p.). The control animals, animals injected with L-arginine, and animals pre-treated with allopurinol were included in this study. Serum and routine pathohistological analysis were conducted to confirm the induction of AP. The metabolic activity and adherence capacity of wBMC were evaluated in the MTT assay and methylene-blue test, respectively, and the alterations in pancreatic tissue and BM were examined on histological sections. In addition, immunohistochemical expression of β-catenin was assessed in the pancreatic tissue.
Results
The wBMC adherence capacity and their total number significantly increased after AP induction. The adherence capability of wBMC was reduced in rats pre-treated with allopurinol. There were no statistically significant changes in the metabolic activity of wBMCs. In the BM isolated from AP and allopurinol pre-treated animals, a discrete left shift in granulocytopoiesis was found, with a slight increase in the myeloid-to-erythroid ratio.
Conclusions
After AP induction, it was shown that wBMCs expressed increased adherence capacity and unchanged metabolic activity, while the alterations in BM may reflect the general activation of myelopoiesis, which would agree with the increased mobilization of mesenchymal stem cells. Also, a significant decrease in β-catenin expression in the pancreatic tissue was noticed.
Introduction
Acute pancreatitis in experimental animals (AP) could be induced by an injection of a high dose of L-arginine 1 and is followed by elevated serum amylase and pathohistological changes in the pancreatic tissue in the form of edema, acinar cell necrosis, and inflammatory cells infiltration. Also, L-arginine leads to increased nitric oxide (NO) production, resulting in vascular permeability and local inflammatory infiltration. 2 Additionally, reactive oxygen species (ROS) and pro-inflammatory molecules are involved in the development of L-arginine-induced pancreatic injury.3,4 Due to the dosage and time dependency of the effects of L-arginine, this model seems adequate for studying the early phases of AP and mechanisms of extra-pancreatic organ damage. 5
A literature search revealed that there could be some involvement of the cells from distant sites, such as the primary lymphoid organ (bone marrow, BM), during the course of L-arginine-induced AP. 6 Bone marrow tissue consists of hematopoietic stem cells, BM mesenchymal stem cells (MSCs), and the descendent progenitors of adipocytes and osteoblasts. 7 MSCs were previously proven to differentiate and allow cellular regeneration of many tissues, such as gastrointestinal cells, nerve cells, cardiomyocytes, cartilage, and liver tissue. Previous research studies have reported that administration of BM-MSCs alleviated AP in rats, 8 while other authors described that BM mesenchymal stem cells were able to decrease the extent of the tissue injury and excessive inflammation during AP.6,9 Therefore, stem cell biology and BM-MSC transplantation are becoming a field of interest for many therapeutic studies. 10
In the following experimental models, where AP has been induced by the injection of different molecules, such as cerulein, L-arginine, or lipopolysaccharide, MSCs demonstrated to be able to reduce the pancreatitis symptoms, as well as parenchymal damage and necrosis. 11 The previous study described a migration towards the pancreas of MSCs injected intravenously, which is followed by the decrease of IL-1β and TNF-α levels. 12 Despite the role of MSC migration during AP, their role in the treatment is still controversial since this phenomenon is not always present. 13
With these facts in mind, we aimed to study the metabolic activity and adherence capacity of the whole cell population from bone marrow in an infiammatory state caused by L-arginine-AP induction. This was done under in vitro conditions through the evaluation of isolated wBMCs' ability to metabolize MTT and adhere to a plastic surface. Additionally, the changes in BM architecture were evaluated by pathohistological analysis of representative BM sections. Also, apart from the standard AP confirmation, the immunohistochemical staining for β-catenin presence in the pancreatic tissue has been examined.
Materials and methods
Animals and housing
Healthy male Wistar rats (200–250 g) were housed at the Vivarium of the Institute of Biomedical Research, Medical Faculty, Niš, Serbia. All animals were maintained under standard husbandry conditions with a temperature of 23 ± 2°C, relative humidity of 55 ± 10%, and a 12/12 h light/dark cycle. Animals were fed with standard, commercial laboratory food pellets and had water available ad libitum. The experiments were performed in accordance with the declaration of Helsinki and the European Community guidelines for the ethical handling of laboratory animals (EU Directive of 2010; 2010/63/EU), and the experimental protocols were commenced after being approved by the animal ethics committee (decision number: 323-07-00,278/2017-05/2).
Experimental procedure: Induction of pancreatitis
Acute pancreatitis (AP) was induced by injecting L-arginine in a single dose of 350 mg/100 g of body weight (i.p.), chosen according to a review 1 and our recent studies.14,15 Three groups of Wistar rats (six animals per group) were included in our experiment, which was the number of animals suggested for this kind of research. 1 Animals from the first group (control/vehicle-treated) received the vehicle in a dose of 0.4 mL/kg, while the ones from the second group were treated with allopurinol, a specific xanthine oxidase inhibitor which was previously suggested to be an adequate positive control drug in same the experimental setting,1,14,15 at a dose of 100 mg/kg (0.1 mL per animal) 1 h prior to L-arginine. Group 3 was injected only with L-arginine. All animals were sacrificed after 24 h by an overdose of ketamine (Ketamidor 10%, Richter Pharma AG, Wels, Austria).
The blood sample was collected by a cardiac puncture, and serum levels of amylase, lipase, and lactate dehydrogenase (LDH) were determined in animals belonging to all three groups using an automated biochemical analyzer (Beckman Coulter AU680 (Olympus America Inc)). Pancreatic tissue and bone marrow samples collected at necropsy were immediately submerged in 10% neutral buffered formalin.
Isolation of whole bone marrow cell (wBMC) population
The wBMC population was isolated using the method previously described. 16 Briefly, rat femurs were dissected, and the bone marrow extraction was performed with a cannula using an RPMI medium. After centrifugation, the supernatants were discarded, and the precipitates were resuspended in the RPMI medium. The cell suspensions were set to 2.5 × 106 cells per mL. Cells were transferred to 96-well microtiter plates and allowed to adhere to the surface for 1.5 h at 37°C under a 5% (v/v) CO2 atmosphere. After that, the metabolic activity and adherence capacity of wBMCs were determined by the MTT or methylene-blue assays, respectively.
MTT assay (Determination of metabolic activity)
The metabolic activity of the wBMC population was determined after the incubation period using a standard MTT (5 mgmL−1) assay. 17 The formation of formazan crystals was evaluated 4 h later, and each well’s absorbance was recorded at 540 nm (Multiscan Ascent, Labsystems, Finland). The activity of the cells was expressed as % of activity based on the formed formazan relative to the one produced by cells isolated from the control (vehicle-treated) group. All experiments were done in triplicate and repeated three times.
Methylene-blue assay (Adherence ability evaluation)
The adherence ability of isolated wBMC was evaluated using a methylene blue-staining protocol. 17 Briefly, after 1.5 h of adherence, the cells were fixed in methanol and stained for 30 min with 1% (w/v) methylene blue solution. Excess dye was removed by a subsequent wash with distilled water, and the remaining cell-retained dye was extracted using a mixture of ethanol and 0.1 moldm−3 aqueous hydrochloric acid (1:1, v/v). The absorbance of the extracts from each well was recorded at 620 nm, and the results are presented as % of adhered cells relative to the absorbance measured for the cultured wBMCs isolated from the control (vehicle-treated) group. All experiments were done in triplicate and repeated three times.
Analysis of pancreatic tissue sections
Following the fixation period, pancreatic tissue samples were dehydrated in ascending grades of alcohol, cleared in xylene, and then embedded into paraffin wax. Histologic 4-μm thick sections were stained with H&E and analyzed following the previously described standard method. 15 For the detection of β-catenin in the pancreatic tissue, primary antibody (DAKO, USA) diluted up to 2.5–5 μg/ml in PBS was applied to hydrogen peroxide-blocked tissue and left to incubate overnight in a moist chamber at 4°C. The sections were counterstained with appropriate secondary antibody, and finally, the visualization was effectuated using diaminobenzidine and counterstained with Mayer’s hematoxylin. The distribution of the stain and its intensity were examined under the light microscope (Olympus BX43, Olympus Corporation, Tokyo, Japan).
Analysis of bone marrow sections
Following the 12-h fixation, samples were decalcified with EDTA, dehydrated in ascending grades of alcohol, cleared in xylene, and then embedded into paraffin wax. Histologic 4-μm thick sections were stained with H&E and Giemsa. The evaluation of BM structure (cellularity, composition, and maturation of cell lineages, possible presence of inflammation, necrosis, and stromal alterations) was performed on the light microscope (Olympus BX43, Olympus Corporation, Tokyo, Japan), while the blood cell count was determined using an automated hematological analyzer.
Statistical analysis
The results of the experiments were expressed as the mean ± SD. Statistically significant differences were determined by a one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test for multiple comparisons (GraphPad Prism version 5.03, San Diego, CA, USA). Probability values (p) less than or equal to 0.05 were considered to be statistically significant.
Results
Levels of serum α-amylase, lipase, and LDH in different experimental groups
In order to confirm the occurrence of AP serum levels of α-amylase, lipase, and LDH were determined in all three groups. Levels of all three studied enzymes were found to be statistically significantly higher in the AP group (Figures 1(A) to (C)), while only levels of α-amylase and LDH were significantly increased in the group of rats treated with allopurinol prior to AP (Figures 1(A) and (C)). Serum α-amylase (A), lipase (B), and LDH (C) activity in rats from different experimental groups. Data are presented as mean ± SD (n = 6). One-way ANOVA, Tukey’s post hoc test: *p < 0.001, **p < 0.01 vs. control group.
Number of wBMCs obtained from different experimental groups after AP induction
Number of the whole bone marrow cells (wBMC) obtained for different experimental groups and counted in Trypan blue (expressed in 106/ml).

Data are presented as mean values ±SD (n = 6); *p < 0.05 vs. Control.
Metabolic activity of wBMCs after AP induction
To assess the metabolic activity in wBMCs we used the MTT assay. Cells isolated from animals with AP did not have statistically significant changes in mitochondrial enzyme activities when compared with the mitochondrial functioning of cells obtained from control animals (Figure 2). Additionally, the treatment with allopurinol showed an approximately equal level of wBMC metabolic activity as the wBMCs isolated from the control (vehicle-treated) rats (Figure 2). Metabolic activity of wBMCs isolated from rats belonging to different experimental groups estimated by the MTT assay. Data are presented as mean ± SD; the statistical significance was calculated by one-way ANOVA.
Adherence capacity of wBMC after AP induction
In order to investigate the adherence capacity of wBMCs after the disease development, a methylene-blue assay was conducted. Cell adherence was statistically significantly increased after pancreatitis induction (Figure 3). Allopurinol significantly decreased cell adherence compared to the wBMCs in the AP group; however, the data were not significantly different from the ones in the control group (Figure 3). Adherence capacity of wBMCs isolated from rats belonging to different experimental groups estimated by the methylene-blue assay. Data are presented as mean ± SD; the statistical signifiance was calculated by one-way ANOVA followed by Tukey’s post hoc test. *p < 0.0001 vs. control group; #p < 0.001 vs. AP.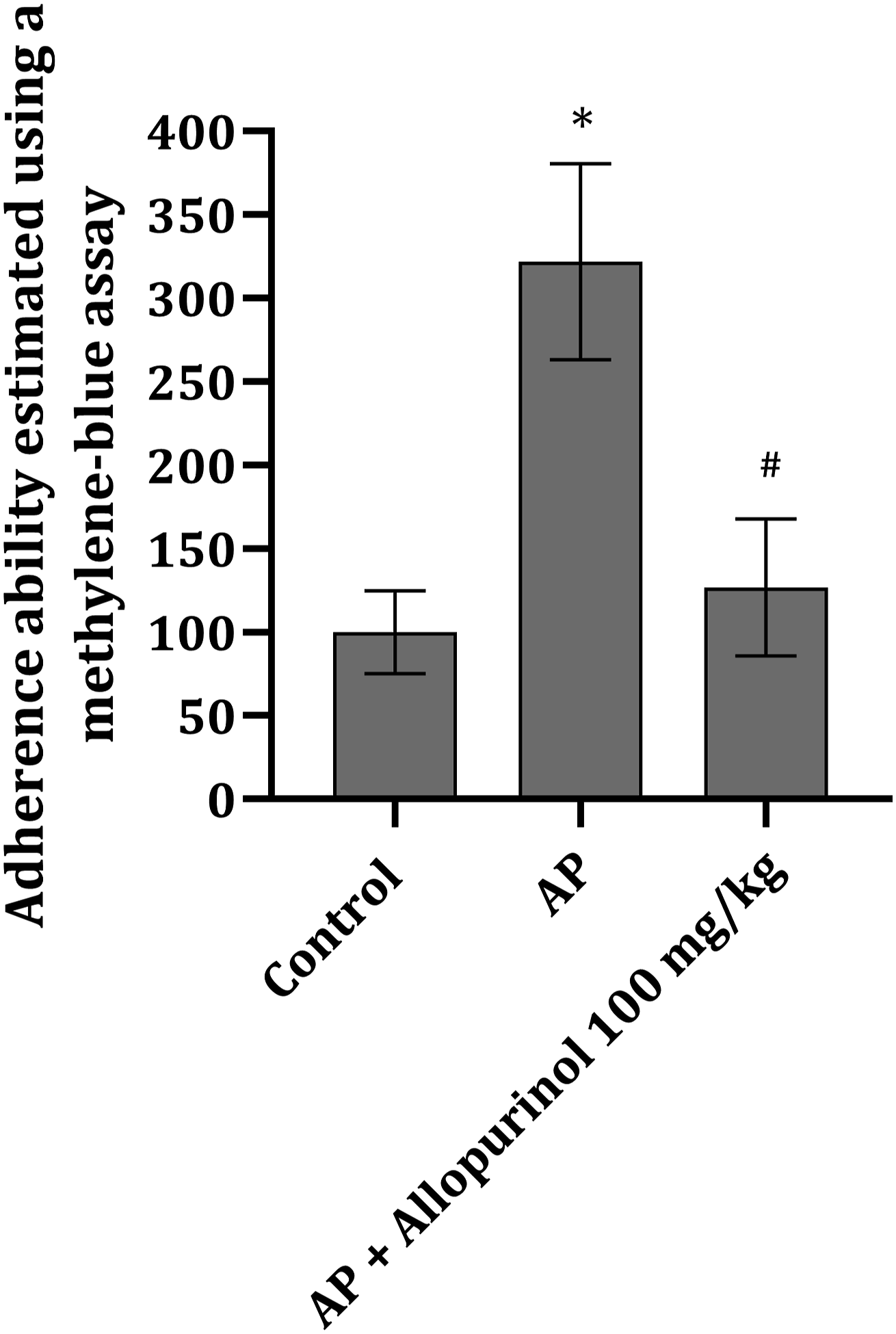
Pancreatic tissue alterations in animals with L-arginine-induced pancreatitis and β-catenin distribution
In the pancreas tissue of healthy rats (control) regular tissue morphology was observed, with no edema between the lobules and with uniform nuclei and granules' distribution within the acinar cells (Figure 4(A)). In the group of rats exposed to a high dose of L-arginine a significant disturbance in the lobule arrangement (tissue edema), inflammatory cell infiltrate, blood vessel hyperemia, and acinar cell degeneration was found (Figure 4(B)). In animals treated with allopurinol prior to L-arginine, similar changes as in the group of AP were noted. However, the intensity of changes was less pronounced and occasionally barely noticeable (Figure 4(C)). Micromorphological appearance (magnification ×100) of pancreatic tissue stained with H&E obtained from healthy rats (A), those with AP induced by L-arginine (B), and those treated with allopurinol (100 mg/kg) prior to L-arginine (C); circled-inflammatory cell infiltration, asterisk-blood stasis.
The distribution of β-catenin in the pancreatic tissue of animals belonging to the control and allopurinol pretreated group was found to be almost identical, i.e. diffuse expression mainly localized in the membrane of the acinar cells of the exocrine pancreas (Figures 5(A) and (C)). Animals that have been treated only with L-arginine demonstrated a diffuse decrease of β-catenin, with the preservation of protein expression only around blood vessels and excretory ducts. Distribution of β-catenin in pancreatic tissue (magnification ×100) obtained from healthy rats (A), those with AP induced by L-arginine (B), and those that have been treated with allopurinol (100 mg/kg) prior to L-arginine (C).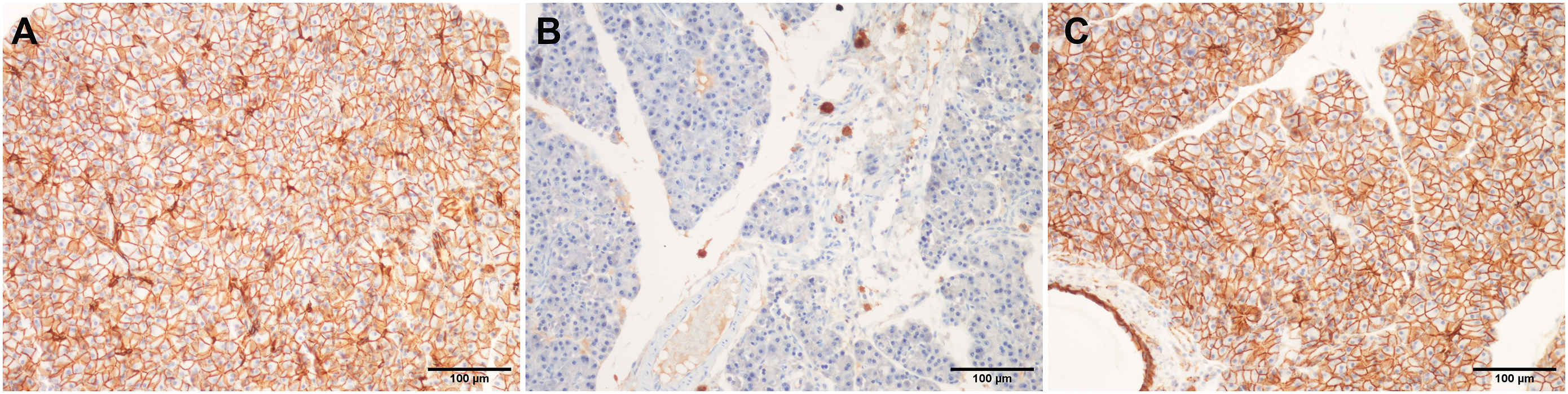
Bone marrow alterations in animals with L-arginine-induced pancreatitis
Pathohistological assessment of BM samples in animals from all three groups revealed normal cellularity of the hematopoietic tissue, with a preserved progressive maturation of granulocytic and erythroid cells (Figure 6). Cellularity was similar in all analyzed samples, but certain subtle differences were observed between the groups. Normal myeloid-to-erythroid (M:E) ratio in rat BM was preserved in all specimens (normal range 1.07–1.93).18,19 However, the cellularity was on the higher end in the AP group. In the allopurinol pre-treated group, BM contained fewer cellular areas, while dilatation of sinusoids and central veins were marked. Erythroid islands showed compact, regular structure and distribution. Granulopoiesis was notably enhanced in the AP and allopurinol pre-treated animals, with a discrete left shift and an increased number of maturing, hyposegmented larger elements. Megakaryocytes, the largest hematopoietic cells with abundant eosinophilic cytoplasm and lobular, segmented large nuclei, were slightly increased in number in the AP group. No significant lymphoid or plasma cell infiltration was found (Figure 6). The micromorphological appearance of BM samples obtained from rats from the control group (
Discussion
Biochemical analysis of the serum enzymes reflecting pancreatic tissue function revealed that injection of L-arginine induces AP, and these results are in line with the ones previously published.14,15 Animals from the positive control group received allopurinol, a well-known and clinically utilized xanthine oxidase inhibitor, which has been suggested as an adequate drug for this kind of study since by inhibition of O2 radical generation, the severity of the disease can be decreased. 1 Microscopic examination of the pancreatic tissue revealed that L-arginine application induces significant cellular disturbance, both degeneration and necrosis, with extensive edema and inflammatory cell infiltration followed by blood stasis (hyperemia). Rats treated with allopurinol were found to have the same changes to a notably lesser extent (Figure 4(C)).
In the present study, we detected a significant increase in rat wBMC adherence capacity after AP induction (Figure 3). In the literature, it was described that BM mesenchymal stem cells have a significant potential to adhere to tissue culture surfaces and also could be multipotent for differentiation into other cell types, under in vitro or after implantation in vivo 20 In addition, increased adherence of BM cells can be considered a beneficial property associated with the possible migration of these cells in vivo that follow AP induction. One potential outcome of wBMC migration is the final amelioration of the inflammatory process in the pancreas, which is in correlation with the results of other investigators.6, 9 Namely, the literature data showed that an injection of BM mesenchymal stem cells partly protected the pancreatic tissue against AP-induced damage through the regulation of inflammatory cytokine release by an exocrine pancreas secretion. 6 Another study confirmed that transplanted BM mesenchymal stem cells were able to migrate and prevent further pancreatic tissue injury as well as systemic inflammatory response occurring during AP. 9
Role of β-catenin in the physiological function of pancreatic tissue is relatively well studied (in cell-type-dependent role in proliferation). 21 However, its role in disease pathogenesis has not been completely elucidated so far. It was found that both during acute and chronic pancreatitis acinar cell proliferation is increased. 22 Thus, the role of β-catenin could not be overlooked. 21 During cell damage, β-catenin detaches from the inner surface of the cell membrane and relocates into the nucleus (or Golgi complex), where it exerts many stress/protective roles, including transcriptional activation of numerous genes. 21 Interestingly enough, we found a significant reduction of β-catenin expression in the pancreatic tissue of rats exposed only to L-arginine, while the treatment of allopurinol either prevented a decline in its expression or increased the expression during the AP. Allopurinol may influence the retention of β-catenin membrane immunoreactivity by contributing to the preservation of cell membrane in L-arginine-induced AP; however, the inhibitory activity of allopurinol to enzymes involved in post-translational modifications of β-catenin cannot be excluded. It is suggested that by enhancing proliferation, pharmacological β-catenin agonists might enhance regeneration and improve the outcome of AP. This is clearly visible in the group treated with allopurinol, where a significant diminution in tissue damage was detected, which could be associated with β-catenin expression that is known to prevent apoptosis. 23
Additionally, in light of their excessive adherence and further migration route, our results confirmed a statistically significant increase in the total cell number in a pool of isolated wBMCs (Table 1). BM hematopoietic tissue is very dynamic and reacts promptly to injury and inflammation occurring in various distant locations. One of the earliest responses during inflammation includes the accelerated release of neutrophils from the BM reserve pools. 24 This reflects in peripheral blood leukocytosis. On the other hand, the leukocyte cell number in peripheral blood remained the same in the present study. Namely, the results from leukocyte blood analysis revealed the silencing of white blood cell (WBC) number (4.9 × 103/μl) during the course of the experiment (i.e. after 24 h post-AP induction), which is in line with the control WBC value (5.4 × 103/μl) (the data not shown). This finding is in correlation with the conclusion that 24 h after AP induction, a total recovery of rat WBCs is present, although the number of these cells was elevated 6 h after AP induction. 25 This elevation during the first 6 h might be associated with a release of MSCs and their migration to the pancreatic tissue to aid the recovery; however, this seems to be possible only in the group of animals treated with allopurinol and L-arginine (Figure 4(C)), but not in the group treated with L-arginine only (Figure 4(B)). The regeneration of the exocrine pancreas is plausible by MSCs from bone marrow, 26 but also from the stem cells within the pancreas. This way of reparation and therapy is much more abundant in the case of diabetes but less in exocrine pancreatic dysfunction.
On the other hand, our further results have shown an unchanged level of wBMC metabolic functioning, i.e. mitochondrial enzymes activities after AP induction (Figure 2). These results might be explained by the fact that cells from BM are exposed to very different and complex microenvironmental influences in in vivo conditions (such as an inflammatory state during AP development), which lead to a specific shaping of these cells’ activity and their unaltered functioning under in vitro conditions. In that sense, our present analysis of BM sections confirmed the absence of any cell infiltrations or alterations in BM structure, which consequently caused unchanged viability of wBMC and their normal metabolic functioning. Another explanation might be that the cells which might be more metabolically active (MSCs) 27 exited the bone marrow at the beginning of the experiment leaving only the cells whose activity is increased, thus matching the one in control. Also, a plausible explanation is that the MSCs have increased their metabolic activity during AP while the other cells decreased, and now the ability of wBMC population to metabolize is almost identical in these two groups.
Our recent publication described that AP induced by a high dose of L-arginine (i.e. severe AP) is characterized by a significant elevation of serum amylase level and also with pathohistological changes in the rat pancreatic tissue. 14 Allopurinol, as a specific xanthine-oxidase inhibitor (100 mg/kg) was chosen based on previously published data that used this drug as a positive control in the AP model induced with L-arginine.1,3,4 Xanthine oxidase, a major source of ROS, is expressed only in inflammatory states (xanthine dehydrogenase is present in normal tissue), which was recently published. 28 It was previously described that treatment with allopurinol effectively ameliorated cerulein-induced pancreatitis. 29
In our present study, 24 h after the damaging insult, i.e. the administration of L-arginine, the assessment of BM sections indicated a stimulation of granulocytopoiesis, mirrored through a white lineage reactive left shift and an increase of the M:E ratio (Figure 6). Cellularity was higher in AP animals than in those pre-treated with allopurinol. Moreover, allopurinol pre-treated animals showed slightly lower cellularity than healthy controls and vascular changes in BM as well. The effects of allopurinol on rat BM have not been well characterized. In the human population, allopurinol is known to cause adverse hematological side effects, which are associated with BM suppression, and lead to anemia, leukopenia, and thrombocytopenia.30,31 However, our results revealed that the discrete changes we found in allopurinol pre-treated rats are insufficient to reflect any damaging effects of this drug on hematopoiesis after AP induction.
Study limitations
One of the study limitation involves the uncalculated sample size, however, this was not performed due to the fact that in previous studies the same number of animals used per group gave satisfactory statistically significant differences between the control and experimental group. Some of the other limitations of the study might include insufficient immunological profile of the examined WBM cell population and some other interactions between WBM cells and pancreatic tissue, or with some other tissue affected by L-arginine application.
Recommendations and future prospective
The results of the present study only provide a small piece of information regarding the role of WBM cell population and its role in AP. Also, data provided by our study reveal the association between adhesion molecules and inflammation during the specific AP induced by L-arginine. Future studies would be focused on the modulation of the immune response in AP induced by L-arginine and on the specific determination of WBM cell immunological profile. Using knockout models and/or inhibitor of β-catenin will be more conclusively determine the role of this crucial pathway in the model of L-arginine-induced pancreatitis and provide a more efficient link about the direct involvement of allopurinol-mediated β-catenin effects.
Conclusions
It can be concluded that there is a statistically significant increase in adherence capacity and the total number of wBMCs in a fulminant infiammatory condition such as AP induced by L-arginine. Additionally, the unchanged metabolic activity of these cells was detected. After the administration of L-arginine and disease induction, the evaluation of BM sections indicated a stimulation of granulocytopoiesis, mirrored through a white lineage reactive left shift and an increase of the M:E ratio. However, no significant lymphoid or plasma cell infiltration was found. Although subtle, the changes in BM from AP animals may reflect the general activation of myelopoiesis, which would be in accordance with the increased mobilization and activation of mesenchymal stem cells. Also, a decrease in β-catenin immunoreactivity in the pancreatic tissue of rats treated only with L-arginine but not in the group with allopurinol and L-arginine further reveals the changes occurring during the course of pancreatitis.
Footnotes
Acknowledgements
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Grant No: 451-03-47/2023-01/200113 and 451-03-68/2022–14/200124).
Authors’ contributions
NMS, KVM, PR, DS and NR conceived and designed the experiment; all authors collected the data and contributed to data analysis and interpretation; NMS, KVM, SS and NSR wrote the manuscript draft. All authors critically reviewed, revised and approved the present manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Grant No: 451-03-47/2023-01/200113 and 451-03-68/2022–14/200124).
Ethics approval
Ethical approval for this study was obtained from the Animal Ethical Committee of Faculty of Medicine, University of Niš, Serbia (decision number: 323-07-00,278/2017-05/2).
All experiments were performed in compliance with the legislation covering the use of animals for scientific purposes, the declaration of Helsinki and EU Directive 2010/63/EU for animal experiments (EU Directive 2010/63/EU For Animal Experiments. Legislation for the Protection of Animals Used for Scientific Purposes. http://ec.europa.eu/environment/chemicals/lab_animals/legislation_en.htm; Accessed August 20, 2021.).
Data availability
The authors confirm that the data supporting the findings of this study are available upon request from corresponding author.
