Abstract
Polycystic kidney disease (PKD) is one of the most common hereditary diseases in cats, with high prevalence in Persian and Persian-related cats. PKD is caused mainly by an inherited autosomal dominant (AD) mutation, and animals may be asymptomatic for years. We screened 16 cats from various breeds exhibiting a renal abnormality by ultrasound examination and genotyped them for the c.10063C>A transversion on exon 29 of the polycystin-1 (
Introduction
Domestic cats are potential models for 218 genetic diseases and phenotypes, of which 72 are known to have causative mutations shared by cats and humans, including polycystic kidney disease (PKD; www.omia.org). PKD is characterized by uni- or bilateral renal cyst formation, and is a systemic, progressive hereditary disease with clinical signs that may develop at any age; cysts can also form in ductal organs, such as liver, seminal vesicle, pancreas, and arachnoid membrane.19,27 In humans, PKD has genetic heterogeneity, with autosomal dominant (AD) and autosomal recessive (AR) inheritance patterns in various genes. Polycystin-1 (
In cats, PKD was first described in a Persian cat family with renal cysts
5
and an AD inheritance pattern6,14 mediated by a point mutation of C → A at sequence position c.10063 in exon 29 of the
The aims of our study were to determine whether the
Materials and methods
Sample collection
We studied 310 domestic cats aged 2 mo to 6 y old, from Turkey. Buccal swab samples were collected for genetic analysis from various cats of various breeds: 35 Angora, 56 Van, 26 Siamese, 22 Scottish Fold, 40 Persian, 9 Exotic Shorthair, 5 British Shorthair, and 117 mixed-breed cats (Ankara University Local Ethical Board of Animal Experiments, Approvals 2017-18-149 and 2018-4-33). Ultrasonography was performed (linear probe 7.5 MHz, convex probe 3.5 MHz, AU5 Harmonic; Esaote Biomedica).
Pathology examinations
A postmortem examination was performed on a Persian cat that died as a result of renal failure. Kidney and liver sections were fixed in 4% paraformaldehyde solution for 48 h and processed routinely for histologic (hematoxylin and eosin, and Masson trichrome stains) and immunohistochemical examinations. 21 Cysts were photographed, and their dimensions were measured by light microscopy (DFC450C digital color microscope camera; Leica).
Immunohistochemistry
Immunohistochemical assays of kidney and liver tissues were performed (UltraVision detection system; Thermo Fisher Scientific), according to the manufacturer’s directions, to demonstrate expression of caspase-3, B-cell lymphoma 2 (Bcl-2), transforming growth factor–beta (TGF-β), and tumor necrosis factor–alpha (TNF-α). The primary antibodies used were: anti–caspase-3 (Ready to use; NeoMarkers), to show the activated apoptotic pathway; anti–Bcl-2 (sc-7382, dilution 1:50; Santa Cruz Biotechnology) for the evaluation of apoptosis inhibition; anti–TGF-β (sc-130348, dilution 1:100; Santa Cruz Biotechnology) to evaluate the initiation of fibrosis; and anti–TNF-α (sc-52746, dilution 1:100; Santa Cruz Biotechnology) to show the degree of inflammation.
In brief, tissue sections were deparaffinized in xylene and hydrated through graded alcohols. To unmask antigens, tissue sections were placed in a microwave oven in citrate buffer, for 20 min at the high setting. Endogenous peroxidase activity was inhibited by 0.1% H2O2 in methanol for 10 min, and nonspecific labeling was blocked by pre-incubation with normal goat serum for 10 min. Thereafter, the sections were incubated with anti–caspase-3, anti–Bcl-2, anti–TGF-β, and anti–TNF-α for 60 min in a humidity chamber at room temperature. Sections were then incubated with biotinylated secondary antibody for 15 min, labeled with horseradish peroxidase for 15 min, and 3-amino-9-ethylcarbazole (AEC) chromogen substrate solution (Thermo Fisher Scientific) for ~ 10 min. Sections were counterstained with Mayer hematoxylin for 1 min and coverslipped with a water-based mounting medium (Thermo Fisher Scientific). The density of immunopositive staining was photographed by light microscopy (BX51 microscope with DP25 digital camera; Olympus).
Molecular genetic analysis
DNA was extracted from the buccal swabs (Quick-DNA universal kit; Zymo), following the manufacturer’s instruction. Briefly, swabs were shredded and placed into centrifuge tubes; 200 µL of genomic lysis buffer was added to the samples and let stand at room temperature for 10 min. The mixture was then transferred to a spin column; DNA was eluted with 50 µL of elution buffer. Obtained DNA was quantified spectrophotometrically (NanoDrop; Thermo Fisher Scientific) and visualized for integrity on dyed (SafeView Classic; ABM) 1% agarose gel; electrophoresis under 100 V. Quantified DNA was stored at –20°C until further analyzed.
The 559-bp region harboring the causative mutation c.10063C>A was amplified by using specific oligonucleotides forward (5′-CAGGTAGACGGGATAGACGA-3′) and reverse (5′-TTCTTCCTGGTCAACGACTG-3′).
23
PCR was set up with 1 pmol of each oligonucleotide, 200 µM dNTP, 1× buffer, 1.5 µM MgCl2, 2 IU of
WGS analyses of the Siamese cat family were performed (HiSeq 2500; Illumina) by a private service provider (BGI). Briefly, gDNA of the 4 individuals was fragmented randomly in separate sample tubes. Sequencing libraries were constructed by adapter ligation (Nextera DNA flex library prep; Illumina), with amplification by PCR and gel purification of the expected bands. DNA cluster preparation was performed and sequenced by paired-end sequencing. Whole genomes were resequenced with an average of 20 coverage. Obtained fastq files were aligned to the latest
Results
Clinical examinations and ultrasonography
Among 310 cats, 16 had various signs including fatigue, anorexia, and vomiting, and were referred to veterinary clinics. Clinical examination by palpation revealed an increase in total kidney volume; ultrasonography was performed. Anechoic and/or cystic lesions were seen in the cortex of both kidneys in 12 cats; bilateral renomegaly was seen in 4 cats. Cystic lesions were not evident in any of the other parenchymal organs.
PCR-RFLP and WGS analysis
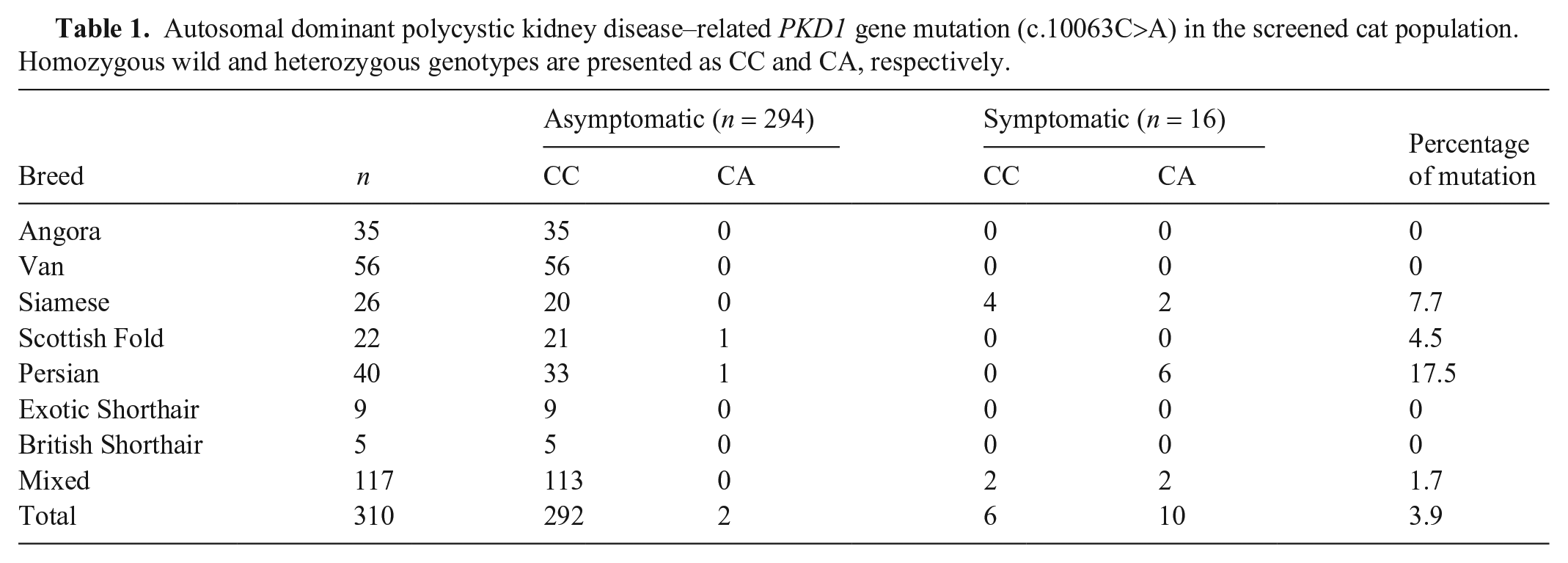
Extracted DNA amounts varied 20–150 ng, and PCR was used to amplify regions of interest from all of the samples. Ten of the 12 cats with cystic kidneys were found heterozygous for the
Autosomal dominant polycystic kidney disease–related
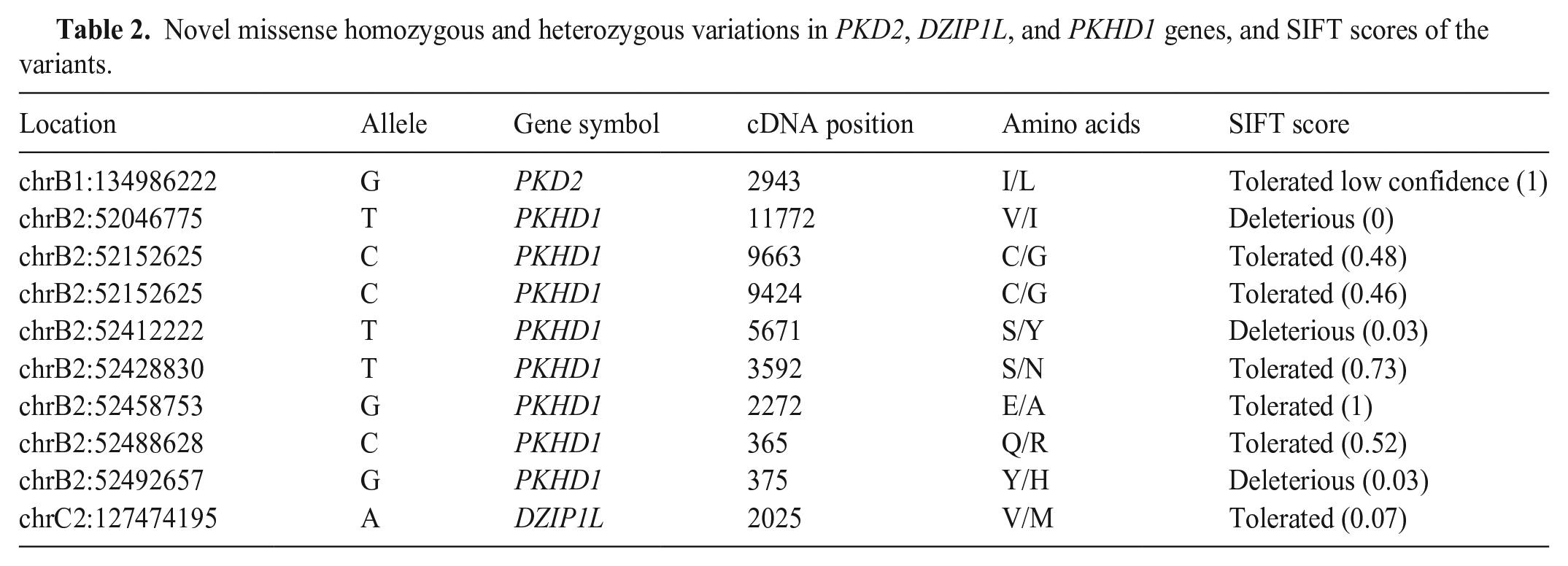
WGS analysis of the causative dominant and recessive genes in the Siamese cat family revealed several novel variations in all genes as well as missense homozygous and heterozygous mutations in
Novel missense homozygous and heterozygous variations in
Histopathology and immunohistochemical analyses
Autopsy of the Persian cat that died in renal failure revealed multiple macroscopic cysts in the cortex of both kidneys (Fig. 1A) and liver (Fig. 1B). Histologic examination of the kidney revealed multiple cystic structures containing homogeneous pink material. Adjacent to cysts, normal, degenerate, cystic, and atrophic tubules and glomeruli were observed. In some tubules, severe epithelial degeneration and necrosis were observed, as well as debris and residues of 2 or 3 tubules, along with microcystic areas (Fig. 1C). In some of the proximal tubules, thyroidization and epithelial hyperplasia were observed, indicating regeneration. Interstitial fibrosis was common in the cystic-degenerate areas. The areas of fibrosis were confirmed with Masson trichrome staining (Fig. 1D).
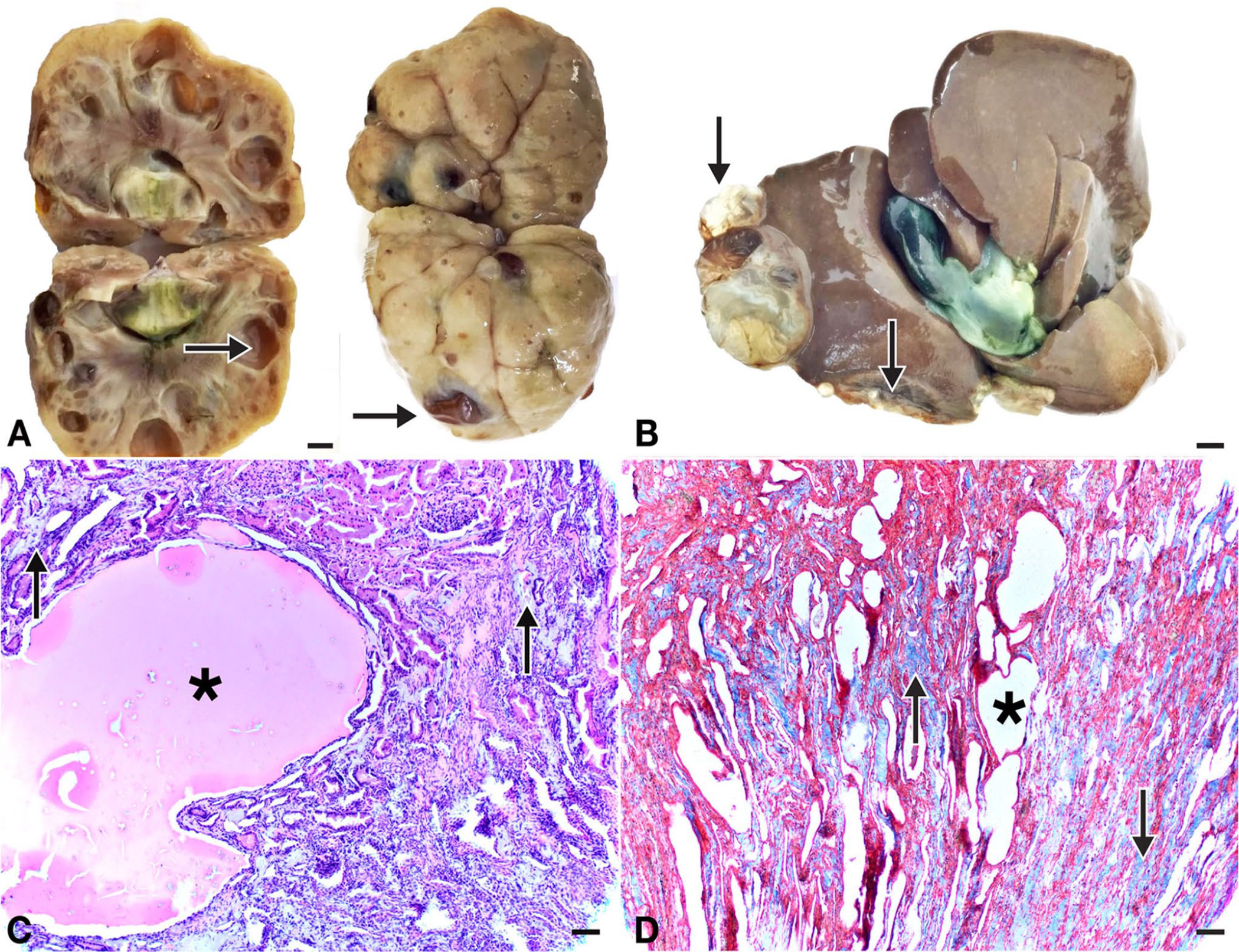
Polycystic kidney and liver in a Persian cat.
Immunohistochemical analyses revealed Bcl-2 (Fig. 2A); degenerate tubule epithelium showed strong caspase-3 immunoreactivity (Fig. 2B), and TNF-α (Fig. 2C) immunoreactivity in degenerate tubule epithelium. TNF-α and caspase-3 immunopositive reactions had similar localization. TGF-β activity was detected in fibrotic areas and tubule epithelium (Fig. 2D).
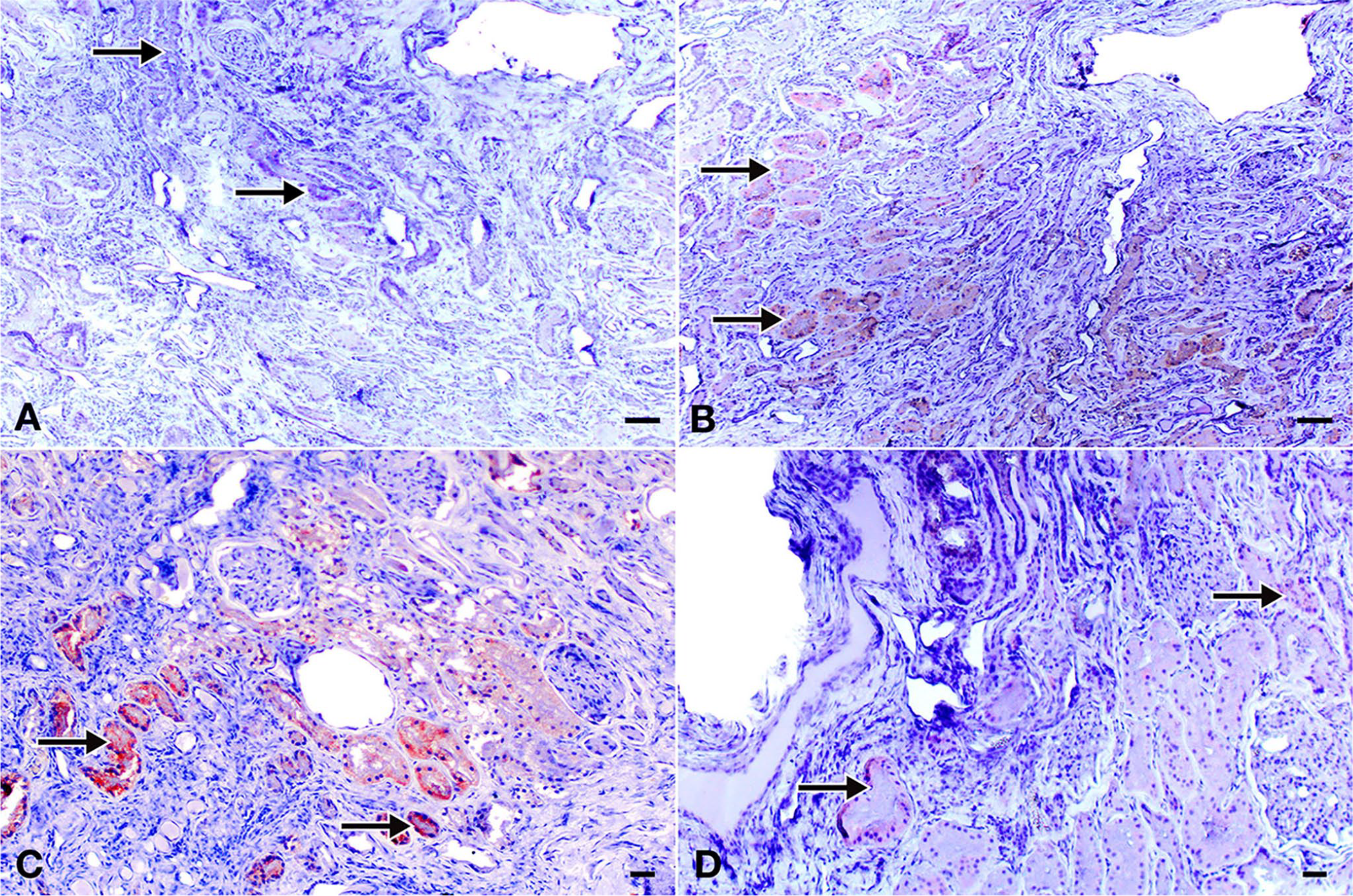
Immunoperoxidase staining of the polycystic kidney in a Persian cat. Indirect avidin–biotin complex immunoperoxidase test, AEC chromogen with Mayer hematoxylin counterstaining. Bars = 100 µm.
Discussion
We found PKD prevalence of 17.5%, 2.7%, and 3.9% in Persian, Persian-related cats (Scottish Fold, Exotic Shorthair, and British Shorthair), and the total cat sample, respectively. In previous studies, which were mainly of Persian and Persian-related cat breeds, the prevalence of PKD was 26% (lowest) to 49% (highest).1,2,8,19 In our study, the mutation was not detected in Exotic and British Shorthair breeds, which might be the result of the small number of subjects. Moreover, molecular examination showed that none of the cats from Angora and Van breeds had the
Previous studies also reported that several cats had renal cysts visible on ultrasonographic examination, but that their 29th exon of
Uncontrolled mating of cats harboring the mutation caused the spread of the mutant allele in the cat population.22,23 Persian cat ancestry is a known major factor in the spread of the PKD mutation,2,8,19 but other breeds, such as Siamese, should also be considered at high risk. These individuals should be screened before accepting them into the breeding programs. According to our WGS analysis, one of the Siamese cats had homozygous non-synonymous mutations in the
In humans with PKD, ~ 300 mutations have been identified in the
In PKD, the balance between tubular degeneration and activation of necrosis and apoptosis is an important factor in the occurrence of cysts. The affected polycystin-1 protein in
Progressive tubular degeneration and microscopic cysts seen in the immediate vicinity of the macroscopic cysts suggests that the pathogenesis of this genetic disease might be related to induction of programmed cell death in affected cells. In addition to cystic structures in the kidney tissue, fibrosis and interstitial tissue enlargement have been noticed, and increased TGF-β expression around these fibrotic areas is noteworthy. This suggests that, in animals with PKD, renal failure may not be only caused by cyst formation, but renal fibrosis might also be an important factor. There are numerous studies on the role of TGF-β in renal fibrosis and progressive renal failure. 28 Fibrosis might be noted as an important factor in renal failure in PKD, and apoptotic markers might be particularly useful in illumination of etiopathogenesis.
Genes associated with cilia formation and those causing ADPKD and ARPKD should be screened for mutations in cat populations to explore differential cyst formation and disease severity in PKD. Furthermore, WGS, clinical analysis, and histopathology should be considered in tandem to discover the candidate genes in cats.
Footnotes
Acknowledgements
We thank the veterinary practitioners who provided materials for our study. We also thank M. Krzywinski for editing the figures.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
