Abstract
A 61-d-old fennec fox (Vulpes zerda), 11 d after receiving a multivalent, modified-live virus vaccine containing canine distemper virus (CDV), canine adenovirus 2 (CAdV-2), parainfluenza virus, parvovirus, and canine coronavirus, developed oculonasal discharge, and subsequently convulsions, and hemoptysis, and died. Microscopic changes in the cerebrum were evident, including neuronal degeneration and necrosis; intracytoplasmic eosinophilic inclusion bodies were observed in astrocytes. CDV was detected in the brain tissue by immunohistochemistry. Pulmonary lesions of multifocal necrotizing bronchopneumonia had Cowdry type A intranuclear inclusions in the bronchial epithelial cells. Electron microscopy revealed crystalline arrays of adenovirus-like particles within the intranuclear inclusions. Additionally, the hemagglutinin gene of CDV and the CAdV-2 DNA polymerase gene were detected in the fennec fox; sequence analysis showed 100% identity with those of the vaccine strain viruses. To our knowledge, vaccine-induced CDV and CAdV-2 coinfections using molecular analysis have not been reported previously. Therefore, vaccine strains should be considered prior to CDV vaccination in nondomestic carnivores.
Canine distemper virus (CDV; Paramyxoviridae, Canine morbillivirus) is an enveloped, negative-sense, single-stranded RNA virus that infects numerous mammalian carnivores, including wild animals. CDV causes serious clinical signs in dogs, including pyrexia, anorexia, nasal discharge, diarrhea, lymphopenia, encephalitis, and death. 1 Canine adenovirus (CAdV; Adenoviridae, Canine mastadenovirus A) is a non-enveloped, double-stranded DNA virus, which infects canids. There are 2 types of CAdV (CAdV-1 and CAdV-2), which are distinguishable by their genetic, antigenic, and pathogenetic characteristics. 2 CAdV-1 is the etiologic agent of infectious canine hepatitis in the fox.4,13 CAdV-2 has been implicated in the etiopathogenesis of infectious tracheobronchitis in dogs, also known as kennel cough. 9 However, there is no current evidence to suggest that CAdV-2 is pathogenic in foxes. 2 CDV and CAdV-2 coinfections have been described in dogs 12 ; there have been no reports of CDV and CAdV-2 coinfections in the fox species, to our knowledge.
Fennec foxes (Vulpes zerda) are the smallest species in family Canidae. They are native to the northernmost areas of African countries but are often kept in captivity in zoos or as pets. A natural outbreak of CDV infection in fennec foxes has been reported. 23 No specific treatment for canine distemper has been proven effective; current treatment consists of supportive care to prevent secondary infections. Therefore, effective vaccination should be considered for CDV-susceptible animals.
We describe pathologic and molecular features of a fatal case of vaccine-derived CDV and CAdV-2 coinfection in a fennec fox that exhibited respiratory and central nervous system (CNS) signs during the post-vaccination period.
A female fennec fox, born in a private home in Japan, remained indoors and had no contact with any other carnivores. At 61 d old, the fox was vaccinated with a multivalent, modified-live virus (MLV) vaccine containing CDV, CAdV-2, parainfluenza virus, parvovirus, and canine coronavirus (Vanguard Plus5/CV; Zoetis).
At 11 d post-vaccination (dpv), the fox exhibited conjunctivitis and nasal discharge (Fig. 1A). At 15 dpv, the fox displayed anorexia and dyspnea. Subsequently, the fox was taken to a veterinary clinic and treated with an antibiotic, 8 mg/kg of cefovecin sodium (Convenia; Zoetis) subcutaneously, and 5 mL/kg/h of lactated Ringer solution intravenously. At 17 dpv, the fox had episodes of hemoptysis (Fig. 1B) and convulsions; an antiepileptic drug (1 mg/kg of diazepam; Cercine; Teva Takeda Pharma) was administered intravenously. The fox died on 18 dpv. The fox was submitted for a postmortem examination.
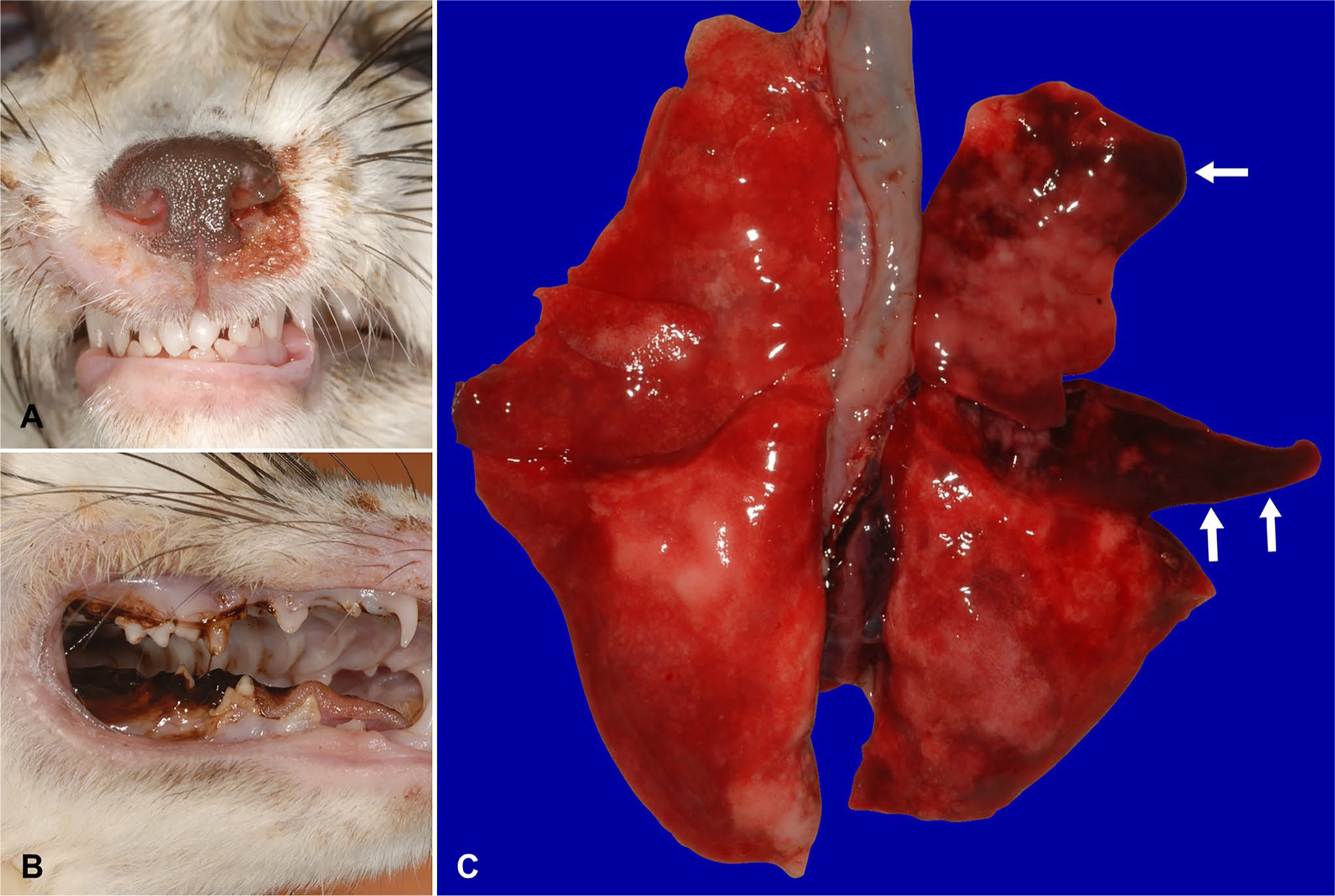
Gross anatomic lesions of vaccine-induced canine distemper virus and canine adenovirus 2 coinfection in a fennec fox.
Tissue samples (brain, lung, trachea, bronchi, heart, esophagus, liver, stomach, intestine, spleen, and kidneys) were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin for histologic examination. Sections of the brain, intestines, and lungs were examined by immunohistochemistry (IHC) using a polyclonal antibody against CDV (Ikeda strain), as described previously. 15 Secondary reactions were performed (peroxidase-conjugated Histofine simple stain MAX PO; Nichirei) and visualized using 3,3’-diaminobenzidine (DAB) and H2O2. IHC slides were counterstained with Mayer hematoxylin. For negative controls, the primary antibody was omitted. For electron microscopic examination, the formalin-fixed lung was washed in 0.1 M phosphate buffer solution and post-fixed in osmium tetroxide. The fixed specimens were dehydrated in alcohol and embedded in epoxy resin according to standard techniques. Ultrathin sections were stained with uranyl acetate and lead citrate and then examined with a transmission electron microscope (JEM-1400Flash; JEOL).
Brain and lung tissues, and oral, rectal, and conjunctival swabs, were collected from the fennec fox. Each sample was mixed with phosphate-buffered saline (PBS). The mixture was used for virus isolation and viral gene detection.
A72/cSLAM cells, expressing canine signaling lymphocyte activation molecule (SLAM), 19 were grown in Dulbecco modified eagle medium (DMEM; Thermo Fisher Scientific) containing 10% fetal calf serum (FCS), 100 U/mL of penicillin, and 100 μg/mL of streptomycin (Thermo Fisher Scientific). Vero cells (Vero 9013, JCRB9013) purchased from the Health Science Research Resource Bank (HSRRB, Japan) were cultured in eagle minimum essential medium (EMEM; Thermo Fisher Scientific) containing 5% FCS, 100 U/mL of penicillin, and 100 μg/mL of streptomycin. The cells were maintained in a humidified 5% CO2 atmosphere at 37°C.
The sample mixture was centrifuged at 2,000 × g for 15 min at 4°C. The supernatants were filtered through 0.45-μm centrifuge tube filters (Costar Spin-X; Corning). Filtrates were inoculated on A72/cSLAM and Vero cells. Cells were incubated with DMEM or EMEM containing 2% FCS and 1% antibiotic–antimycotic (Thermo Fisher Scientific) at 37°C. The cells were maintained and passaged until a cytopathic effect (CPE) was observed. After 5 blind passages, samples without CPE were judged as negative for virus isolation.
To determine the nucleotide sequence of the complete hemagglutinin (H) gene of CDV in the brain and of the vaccine, the H gene was amplified by reverse-transcription PCR (RT-PCR; RNA LA PCRTM kit (AMV) v.1.1; Takara Bio). Reverse transcription was performed using random 9-mer oligonucleotide primers, and PCR was performed using primer pairs HF (5′-AACTTAGGGCTCAGGTAGTC-3′) and HR (5′-AGATGGACCTCAGGGTATAG-3′). 6 PCR products were electrophoresed on 0.8% agarose gel and extracted (MinElute gel extraction kit; Qiagen).
To determine the nucleotide sequence of the DNA polymerase gene of CAdV, DNA was extracted from the isolated virus and the vaccine (DNeasy blood & tissue kit; Qiagen) according to the manufacturer’s instructions. The DNA polymerase gene was amplified by nested PCR (Ex Taq; Takara Bio). The first PCR was performed using primer pairs polFouter (5′-TNMGNGGNGGNMGNTGYTAYCC-3′) and polRouter (5′-GTDGCRAANSHNCCRTABARNGMRTT-3′). The second PCR was performed using primer pairs polFinner (5′-GTNTWYGAYATHTGYGGHATGTAYGC-3′) and polRinner (5′-CCANCCBCDRTTRTGNARNGTRA-3′). 21 PCR products were electrophoresed on 2.0% agarose gel and extracted (MinElute gel extraction kit).
Nucleotide sequences of both the H gene of CDV and the DNA polymerase gene of CAdV were determined (BigDye terminator v.3.1 cycle sequencing kit; Thermo Fisher Scientific) according to the manufacturer’s instructions.
Phylogenetic trees based on the amino acid sequences of the CDV H gene were constructed using a distance-based (neighbor-joining) method with MEGA 7.0 software. 16 Bootstrap values were calculated based on 1,000 replicates.
The lung was grossly and incompletely deflated, diffusely and mildly edematous, and the right cranial and middle lobes had multifocal-to-coalescing, dark red, consolidated areas (Fig. 1C). Microscopically, pulmonary lesions of multifocal necrotizing bronchopneumonia had Cowdry type A intranuclear inclusions in bronchial epithelial cells (Fig. 2A). Electron microscopy revealed crystalline arrays of viral particles (72 nm diameter) in the intranuclear inclusions in the bronchial epithelium. The virus particles were nonenveloped and hexagonal with electron-lucent or electron-dense cores (Fig. 2B), suggestive of CAdV infection. Grossly, meningeal vessels were congested. Microscopic changes in the cerebrum included neuronal degeneration, necrosis, and rarefaction of the neuropil. There was no evidence of perivascular lymphocyte infiltration or demyelination in the brain, which is commonly seen with CDV infections. Intracytoplasmic eosinophilic inclusions were observed in astrocytes (Fig. 2C). Further, CDV antigen was detected in nerve cells of the cerebrum and cerebellum by IHC (Fig. 2D). In other regions, CDV antigen was only detected in a few mesenchymal cells of the small intestinal lamina propria. The thymus was involuted, the mesenteric lymph node was atrophied, and microscopic changes in the spleen were confirmed as marked lymphoid depletion (Fig. 2E). Respiratory signs were considered to be associated with bronchopneumonia and neurologic signs with neurodegeneration and necrosis in the CNS.
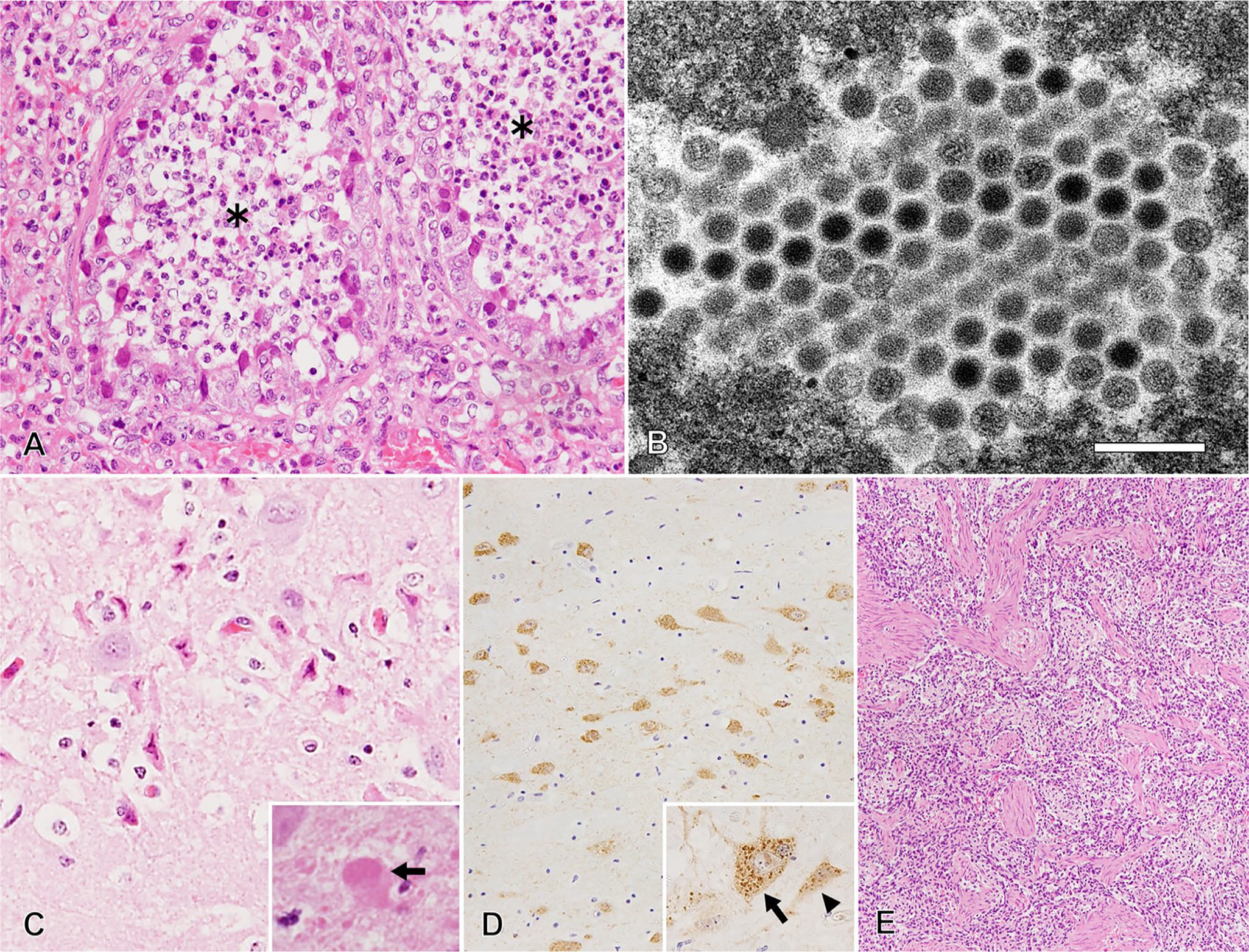
Histologic lesions of vaccine-induced canine distemper virus (CDV) and canine adenovirus 2 coinfection in a fennec fox.
The CDV H gene was detected in the brain, and the sequence was determined and deposited into the DNA Data Bank of Japan (DDBJ; accession LC498611). This detected CDV was named Pr17131O-CDV, although CDV was not isolated. The CAdV DNA polymerase gene was detected in the lung, and CAdV was isolated from the oral swab using both A72/cSLAM and Vero cells. The isolated virus was named Pr17131O-CAdV, and the nucleotide sequence of the DNA polymerase gene was determined and deposited into DDBJ (accession LC498612), indicating that the isolated virus was CAdV-2. Based on these findings, a clinical diagnosis of CDV and CAdV-2 coinfection was made in this fennec fox.
Vaccine-induced CDV infection has been documented in various canids, including domestic dogs.7,11,17,18 If pups become ill within 2–3 wk after CDV vaccine administration, the development of vaccine-induced disease is always suspected. However, it has been difficult to determine whether the disease was induced by the vaccine virus or by field viruses infecting pups shortly before or after vaccine administration. 17 Most previous studies regarding vaccine-induced CDV-associated diseases have been based on indirect evidence, such as the circumstances of the vaccination, histopathologic findings, and serology.
The development of vaccine-induced disease is caused by various factors, including host factors, such as age, genetic constitution, and immune status, as well as by the degree of virus attenuation. There are 2 major categories of commercial CDV vaccines: MLV vaccines, and canarypox-vectored recombinant vaccines. Recombinant CDV (rCDV) vaccines have proven to be generally safe and efficacious for nondomestic carnivores.8,14 Unfortunately, the alternative rCDV vaccines have not been available in Japan.
Our case received the multivalent MLV vaccine when ~ 2 mo old, and there was no outbreak of CDV at this point in the breeder facility. Further, the first clinical signs of conjunctivitis appeared 11 dpv, which is consistent with the incubation period of CDV. 1 In other fox species, chicken embryo or cell culture strain MLV CDV vaccines have been tested, and they were proven to be immunogenic and safe.10,20 In contrast, canine kidney cell culture strain MLV vaccines are typically considered to have more adverse effects on wildlife species.8,10 The Rockborn strain, which is grown in primary dog kidney cells, has been suspected to retain residual virulence after several reports of vaccine-related disease.5,7,17 In addition, increased virulence of the dog-kidney-cell attenuated CDV has been suspected of having been triggered by its contemporaneous presence in vaccine formulations (i.e., the live modified CAdV-1 vaccine). 17
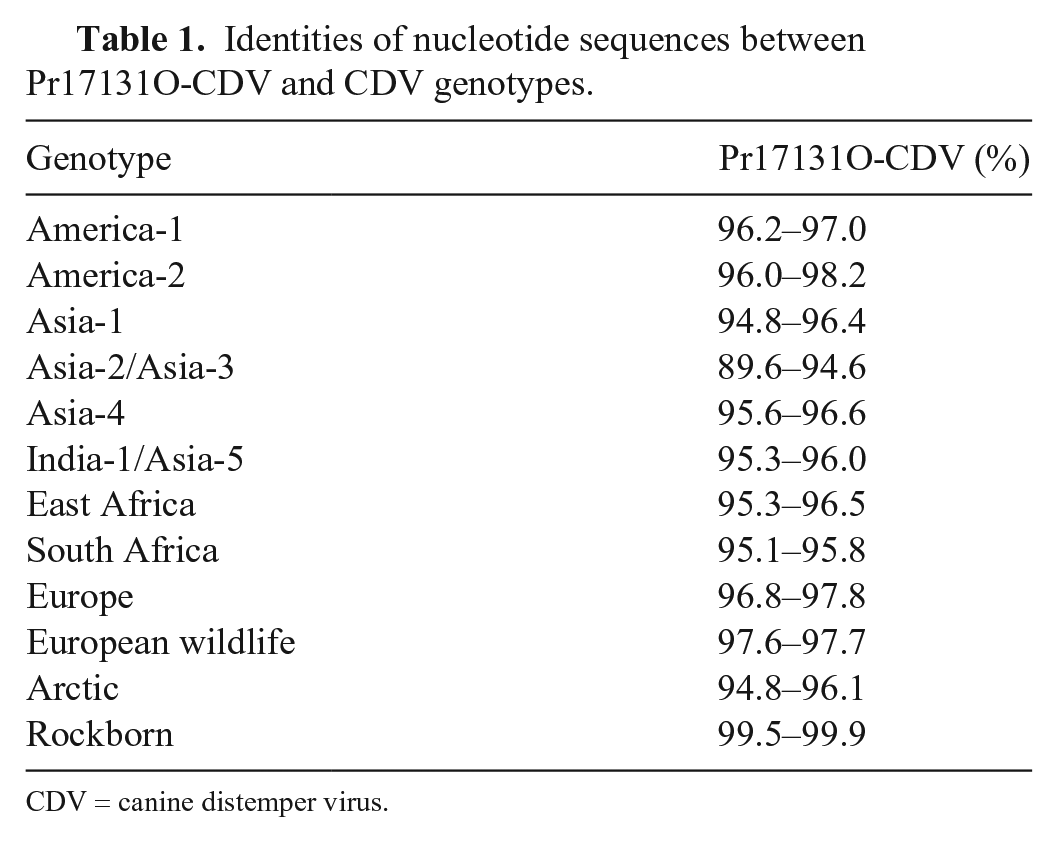
Sequence analysis showed 100% CDV identity of the H gene between the vaccine-amplified strain and Pr17131O-CDV. In addition, the Rockborn strain (GU810819), matching Pr17131O-CDV, was identified in the sequence databases. The H gene, which plays vital roles in the infectious process, is profoundly suitable for phylogenetic analysis because it is considered one of the most variable genes in the CDV genome.6,22 In our study, phylogenetic analysis showed that Pr17131O-CDV was different from the Asian wild-type genotypes spreading in Japan (Fig. 3, Table 1, Suppl. Table 1). Our findings support vaccine-induced CDV infection in the fennec fox.
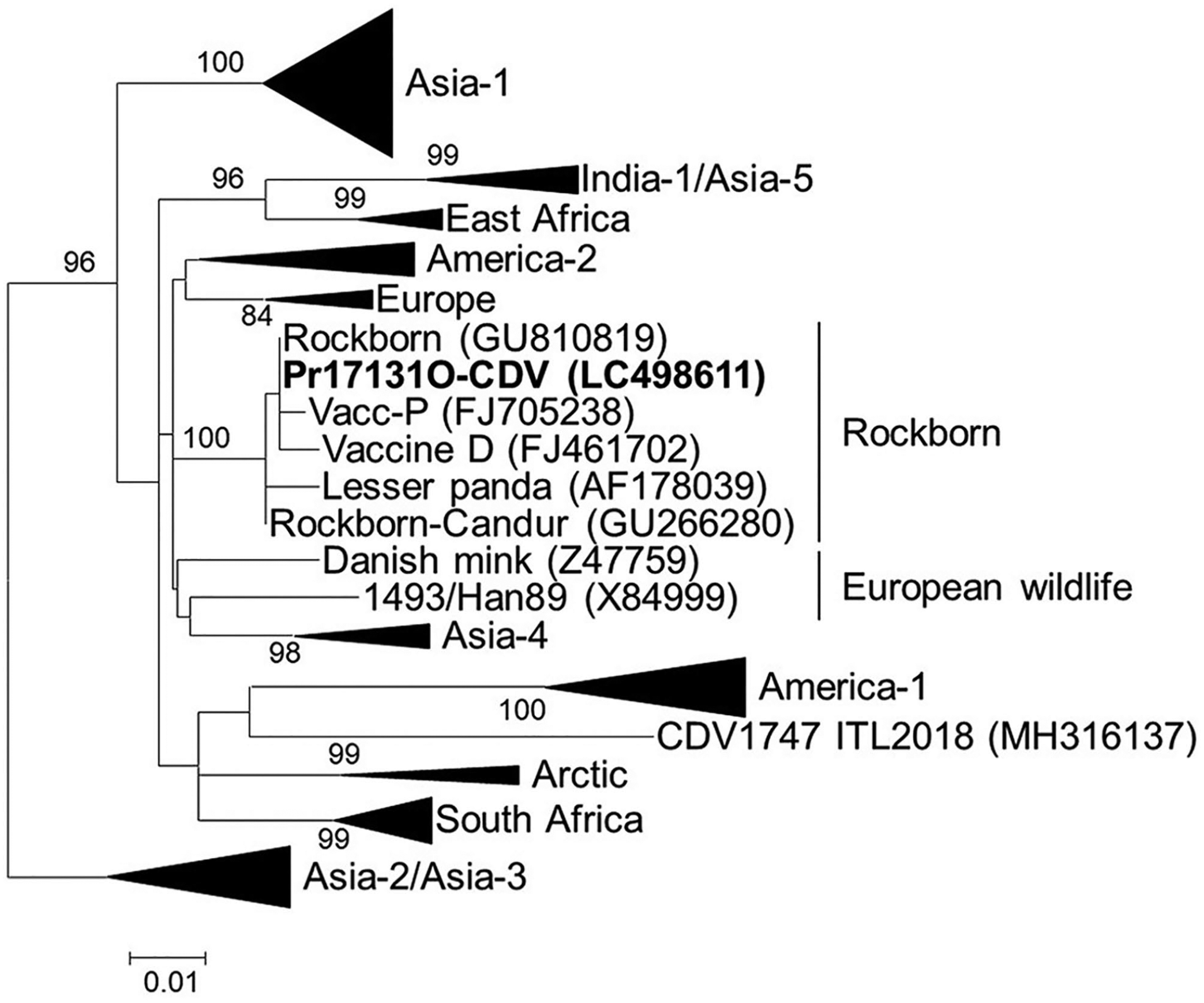
Phylogenetic tree showing genetic relationships of the Pr17131O-CDV/Rockborn strain to other canine distemper virus (CDV) genotypes. Phylogenetic trees based on the amino acid sequences of the H gene (607 amino acids) were constructed using a distance-based (neighbor-joining) method with MEGA 7.0 software. GenBank accessions are shown in parentheses. The accessions of the reference strains in collapsed branches (triangles) are in Suppl. Table 1. Scale bar indicates the number of nucleotide substitutions per site.
Identities of nucleotide sequences between Pr17131O-CDV and CDV genotypes.
CDV = canine distemper virus.
In our case, hemoptysis occurred secondary to the first clinical signs of conjunctivitis after 4 d. Immunohistochemical findings revealed no CDV antigen in the lungs, but intranuclear bodies associated with CAdV infections were observed in the lesions. Furthermore, sequence analysis showed 100% CAdV-2 DNA polymerase identity between the strain amplified from the vaccine and Pr17131O-CAdV. Additionally, the Manhattan strain (S38212), matching Pr17131O-CAdV, was identified in the sequence databases. Although CAdV-2 infection alone is considered to commonly have low pathogenicity, the severe form results in general poor health and/or immunosuppression in dogs. Secondary bronchopneumonia can occur. 9 The immunosuppressive effects associated with CDV-induced infections have also been shown to be associated with selective destruction or impairment of SLAM-expressing cells because of viral tropism in lymphoid tissues. 3 Based on the histopathologic and electron microscopy findings, CAdV-2 is most likely the etiologic agent that caused bronchopneumonia in this fox.
Supplemental Material
Supplemental_material – Supplemental material for Molecular evidence for vaccine-induced canine distemper virus and canine adenovirus 2 coinfection in a fennec fox
Supplemental material, Supplemental_material for Molecular evidence for vaccine-induced canine distemper virus and canine adenovirus 2 coinfection in a fennec fox by Kenichi Tamukai, Shohei Minami, Rio Kurihara, Hiroshi Shimoda, Ikki Mitsui, Ken Maeda and Yumi Une in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Mr. T. Saito and Ms. R. Suzuki for submitting the fennec fox for autopsy, and Mr. K. Watanabe for electron microscopic examination. We also thank Drs. Y. Watanabe and H. Nanba for providing expert advice.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by a grant from the Japan Agency for Medical Research and Development (19fk0108097).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
