Abstract
A 4-y-old cat exhibited neurologic signs such as wobbling, right head tilt, and intention tremor, and MRI revealed a mass in the cerebellum. The cat died 5 mo after initial presentation, and no neoplastic lesions, other than the cerebellar mass, were observed at autopsy. Histologically, large atypical cells resembling Hodgkin cells, with single large inclusion-like nucleoli, and those resembling Reed–Sternberg cells, with symmetrically arranged nuclei, had infiltrated the left side of the cerebellum and were admixed with small lymphocytes. These atypical cells were positive for feline leukemia virus (FeLV), CD20, BLA36, vimentin, p16, p53, and Pax5, and negative for CD3, CD79a, and Iba1 by immunohistochemistry. Multiplex PCR for immunoglobulin heavy-chain gene rearrangement revealed monoclonal proliferation of B-lymphocytes. We describe this feline primary cerebellar B-cell lymphoma that displayed Hodgkin lymphoma–like tumor cells with FeLV protein expression.
Primary central nervous system (CNS) lymphoma is rare, both in humans and in animals. The literature on cytologic classification and immunohistochemistry of primary CNS lymphoma in animals is sparse, although there are a few confirmed cases of T-cell–5,11 or B-cell–origin12,13 lymphomas in cats. Feline intracranial lymphomas most commonly affect the brainstem, cerebrum, or meninges 15 ; however, primary cerebellar lymphoma has not been reported to our knowledge. We describe a feline case of primary cerebellar B-cell lymphoma that displayed Hodgkin lymphoma–like tumor cells with feline leukemia virus (FeLV) protein expression. Extranodal manifestation of Hodgkin lymphoma is rare, although there are a few case reports on primary CNS Hodgkin lymphoma in humans. 6
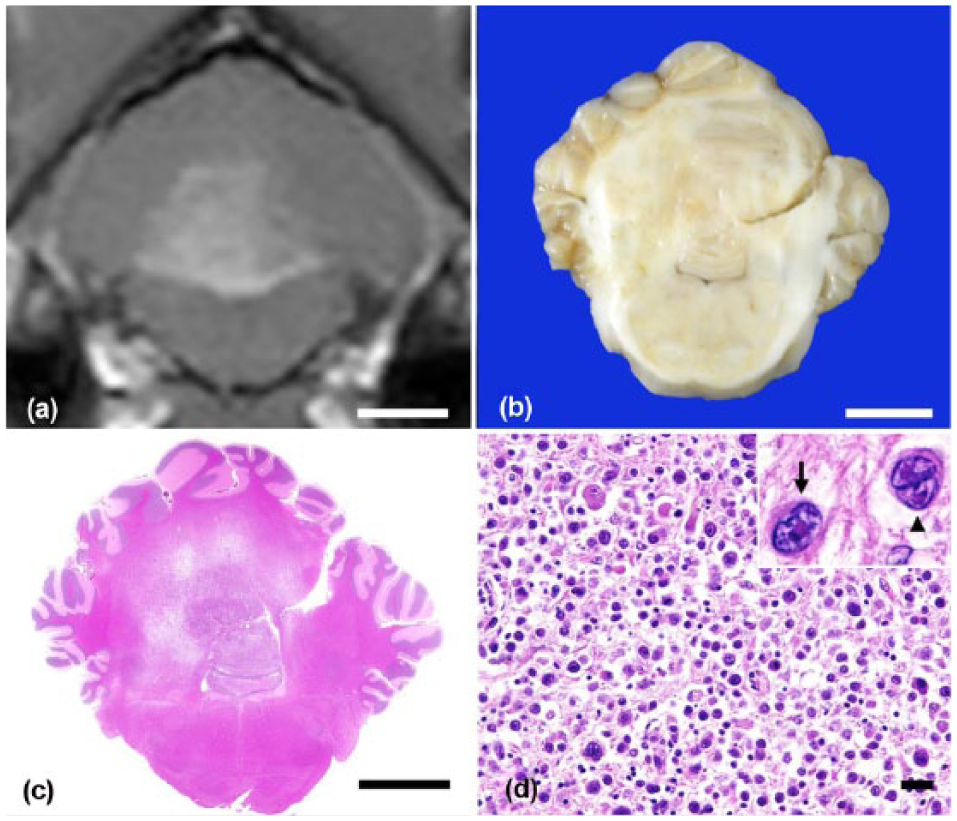
A 4-y-old neutered male, mixed-breed cat that had shown neurologic signs, including wobbling and right head tilt, was referred to the Veterinary Medical Center at the University of Tokyo. Magnetic resonance imaging (MRI) showed a high signal area in the left cerebellum. The cat was seropositive for FeLV antigen and feline immunodeficiency virus (FIV) antibody. Treatment with prednisolone led to transient improvement, but the cat started to exhibit dysstasia with intention tremor. Four months after the initial examination, MRI revealed enlargement of the lesion (Fig. 1a). The cat died 1 mo later, and an autopsy was performed. At the time of autopsy, no significant findings were noted except for pulmonary congestion. Cytologic examination of the bone marrow revealed no significant finding. Samples of most visceral organs and the brain were collected and fixed in 10% neutral-buffered formalin. On cut surface, a gray nodular lesion (1.0 × 1.0 cm) occupied the left side of the cerebellum, and the border of the lesion was indistinct (Fig. 1b). Tissues were embedded in paraffin, sectioned at 4 μm thickness, and stained with hematoxylin and eosin (H&E) and Congo red. Immunohistochemistry was performed (Table 1). Horseradish peroxidase–labeled polymer (EnVision+ system, Dako, Tokyo, Japan) was used as a secondary antibody. Antigen–antibody complexes were visualized with the 3,3’-diaminobenzidine (DAB) chromogen, and the sections were counterstained with hematoxylin. For clonality analysis, DNA was extracted from the paraffin sections (QIAamp DNA FFPE tissue kit, Qiagen, Valencia, CA). Multiplex polymerase chain reaction (PCR) was performed to detect immunoglobulin heavy-chain gene or T-cell receptor gene rearrangement as described previously.9,10 PCR was performed in duplicate.
Primary antibodies and the results of immunohistochemistry.
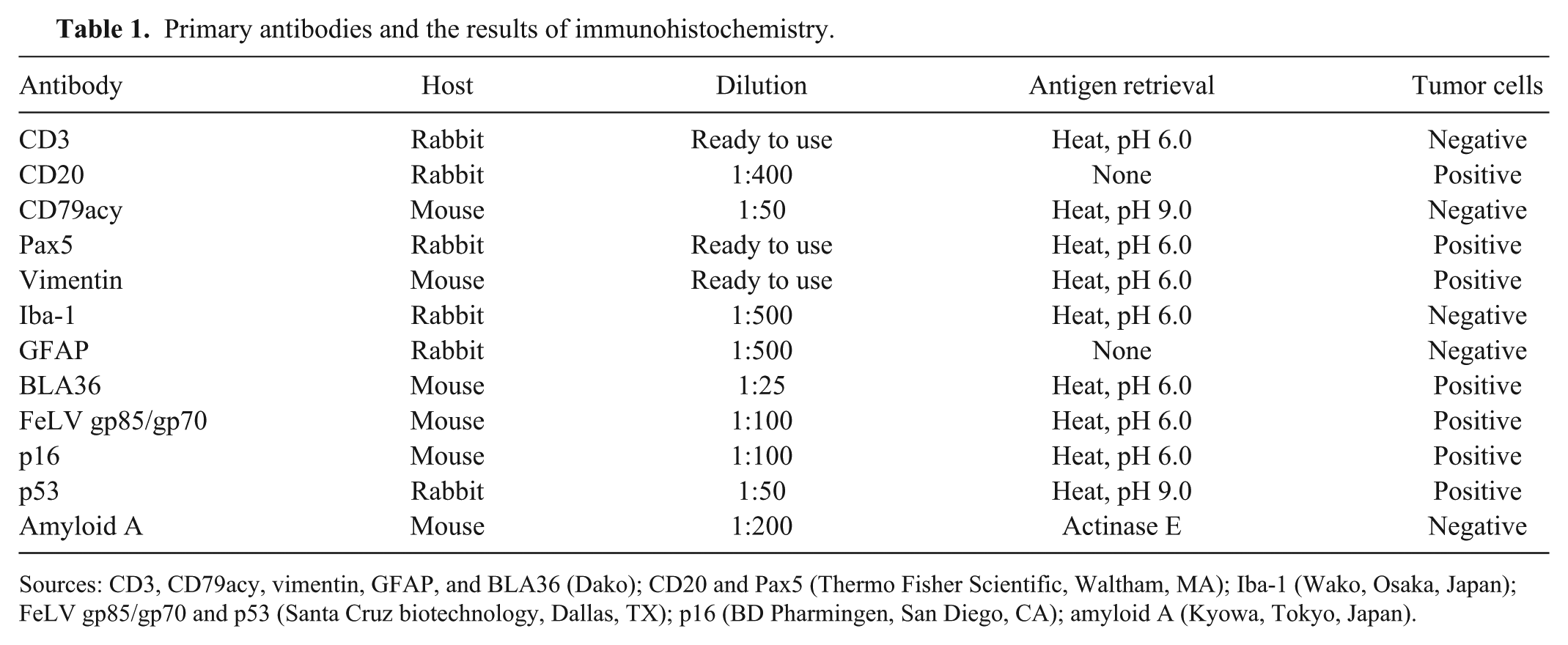
Sources: CD3, CD79acy, vimentin, GFAP, and BLA36 (Dako); CD20 and Pax5 (Thermo Fisher Scientific, Waltham, MA); Iba-1 (Wako, Osaka, Japan); FeLV gp85/gp70 and p53 (Santa Cruz biotechnology, Dallas, TX); p16 (BD Pharmingen, San Diego, CA); amyloid A (Kyowa, Tokyo, Japan).
Histologically, the structure of the cerebellar medulla was obscured (Fig. 1c). In this area, large atypical round cells with abundant amphophilic cytoplasm were observed, and they infiltrated the cortex, meninges, and choroid plexus. These tumor cells showed marked anisokaryosis and anisocytosis. The nuclei of the tumor cells were oval, with thick, irregular, or indented nuclear membranes (Fig. 1d). Both tumor cells with single large inclusion-like nucleoli resembling Hodgkin cells, and those with symmetrically arranged nuclei resembling Reed–Sternberg cells, were common (Fig. 1d). These cells were ~40 μm in diameter. Mitotic figures were frequent (1 per high power field). A number of lymphocytes, macrophages, and reactive astrocytes were admixed with the tumor cells. Proliferation of tumor cells was not found in organs outside the cerebellum.
The tumor cells were strongly positive for FeLV gp85/gp70, cluster of differentiation (CD)20, B-lymphocyte antigen (BLA)36, vimentin, p16, and p53; weakly positive for Pax5 (Fig. 2a–g); and negative for CD3, CD79a, and Iba1. Most of the lymphocytes were CD3-positive T-cells (Fig. 2h). Multiplex PCR analysis revealed a single clear band of amplification product of the immunoglobulin heavy-chain gene but not of the T-cell receptor gene (Supplemental Table 1; Supplemental Fig. 1). A moderate amount of Congo red–positive amyloid was observed in the liver, kidney, and left thyroid. The amyloid was positive for amyloid A by immunohistochemistry.
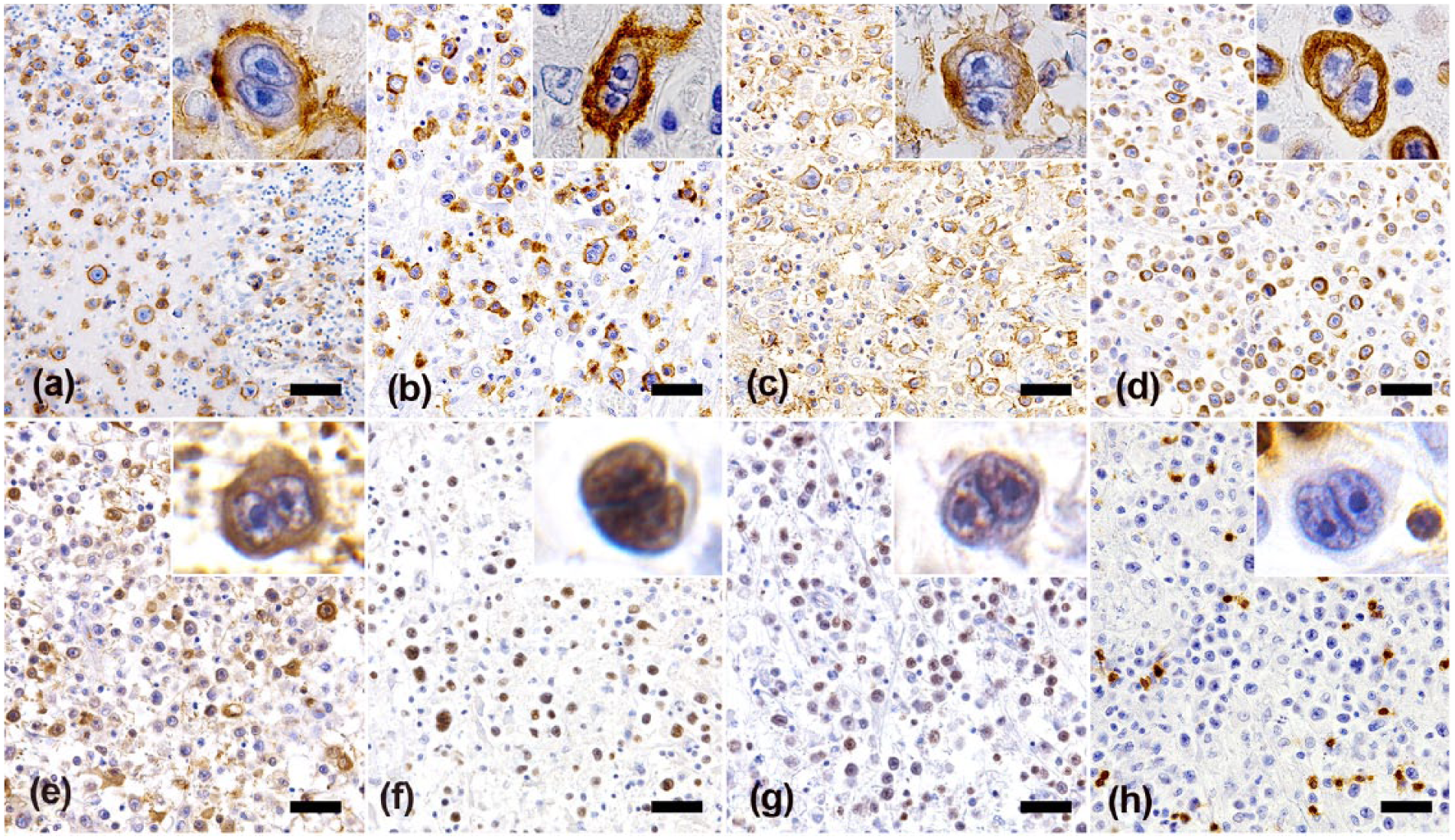
Tumor cells are positive for FeLV gp85/gp70 (a), CD20 (b), BLA36 (c), vimentin (d), and p16 (e). The nuclei of the tumor cells are positive for p53 (f), and weakly positive for Pax5 (g). Small lymphocytes admixed with the tumor cells are positive for CD3, but the large atypical cells are negative (h). Insets: higher magnifications of tumor cell. Bars = 100 μm.
After 7 mo of clinical disease, no evidence of metastasis was found at autopsy and histologic examinations. Based on gross, microscopic, and immunohistochemical results, primary cerebellar lymphoma was diagnosed. The morphology of the tumor cells was quite characteristic. Hodgkin cells and Reed–Sternberg cells (H/RS cells) are diagnostic hallmarks of Hodgkin lymphoma in humans. H/RS cells have also been described in lymphoma cases of dogs and cats.8,17 Human Hodgkin lymphoma has distinctive features such as a high prevalence of Epstein-Barr virus infection, high occurrence in young age, spread by local extension, and favorable clinical outcome with treatment. The most recognized counterpart of Hodgkin lymphoma in animals is the T-cell–rich, large B-cell lymphoma, although there is an ongoing debate whether this entity is a true counterpart or a different entity. The tumor often occurs in the cervical area of cats and shows similar morphologic and clinical features to those of Hodgkin lymphoma in humans. Veterinary pathologists have described this type of lymphoma as either “Hodgkin-like lymphoma” 17 or “T-cell–rich B-cell lymphoma” 3 ; however, in each study, the authors suggested that the feline disease is a counterpart of Hodgkin lymphoma, particularly of lymphocytic and histiocytic subtypes. Unfortunately, gene rearrangement analysis was not performed in these previous studies.
In general, Hodgkin lymphoma is considered to be derived from B-cells in the germinal center or post-germinal center of the lymph node, although tumor cells are negative for a common B-cell marker, CD79a. Anti-BLA36 antibody was raised against a human Hodgkin cell line; this antibody reacts with Reed–Sternberg cells as well as with activated B-lymphocytes. 7 A nuclear transcription factor related to B-cell differentiation, Pax5, is expressed in Hodgkin lymphoma as well as in diffuse large B-cell lymphomas (DLBCL) but not in other lymphomas. Hodgkin lymphoma tumor cells are faintly positive for Pax5, but those of DLBCL are strongly positive. 4 The expression of another B-cell marker, CD20, has been thought to indicate a better prognosis for Hodgkin lymphoma. 16 Vimentin, CD30, and CD15 are markers used to differentiate T-cell–rich B-cell lymphoma from Hodgkin lymphoma in humans (i.e., these markers are negative in T-cell–rich B-cell lymphomas but positive in Hodgkin lymphomas). 14 In our feline case, the results of immunohistochemistry and clonality analysis indicated that the tumor cells were of B-cell origin. Moreover, the immunohistochemical results are comparable to those of Hodgkin lymphoma in humans. In humans, the incidence of primary CNS lymphoma has increased in association with the AIDS epidemic. It has been reported that 17.6% of feline intracranial lymphoma cases, including both primary and secondary cases, were seropositive for FeLV; all the cases were negative for FIV. 15 In studies on feline Hodgkin-like lymphoma, 1 of 4 cats was seropositive for FeLV antigen by ELISA 17 ; 8 of 8 cats were negative for FeLV by PCR analysis using DNA extracted from paraffin-embedded tissues. 3 However, immunohistochemistry for virus antigen was not performed.3, 17 In our case, the cat was seropositive for both FeLV and FIV. Also, the envelope proteins of FeLV (gp85 and gp70) were expressed specifically in the atypical tumor cells, indicating viral propagation in the tumor cells. The expression of p16 and p53 in H/RS cells is reported to be associated with Epstein-Barr virus infection in classic human Hodgkin lymphoma. 1 Moreover, p16 and p53 are involved in the pathogenesis of various virus-associated neoplasms. Although the relation between the FIV and FeLV infection and morphologic variation of B-cell lymphoma cells has been suggested, 2 further study is needed to elucidate the implication of viral infection for the atypical morphology in feline lymphoma with a Hodgkin lymphoma–like appearance.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported in part by Japan Society for the Promotion of Science KAKENHI grants 26660235 and 26660236.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
