Abstract
An outbreak of inclusion body hepatitis caused by fowl adenovirus serotype 8 (FAdV-8) has caused significant economic losses in the poultry industry worldwide. However, a rapid serology test kit specific to FAdV-8 is not available to date. We developed a fiber-based ELISA using the purified GST-fiber of FAdV-8 as coating antigen to measure antibodies against FAdV-8. Specificity analysis showed that our ELISA could react with sera against FAdV-7, -8a, and -8b, but not with sera against the other pathogens tested. Moreover, detection of positive sera with our ELISA had 83% and 94% agreement with an indirect immunofluorescence assay (IFA) and a commercial ELISA from BioChek, respectively. Our ELISA was also effective in the detection of antibodies against FAdV-8 in sera from both experimentally infected and clinically vaccinated chickens. Our FAdV-8 fiber-based ELISA can be a valuable tool to specifically and sensitively detect antibodies against FAdV-7 and/or -8 in infected or vaccinated chickens.
Based on its genomic sequence and antigenic characteristics, fowl adenovirus (FAdV; Fowl aviadenovirus) has been clustered into 5 species (FAdV-A to -E) with 12 serotypes (1–7, 8a, 8b, 9–11).7,12,13 FAdV infections are usually subclinical in chickens; chickens with acute infection may develop inclusion body hepatitis (IBH), hepatitis hydropericardium syndrome, or gizzard erosion and ulceration.12–14 Of the 12 FAdV serotypes, FAdV-6, -7, -8a, and -8b, which belong to FAdV-E, are thought to be the main pathogens for IBH.8,10,12,14 Outbreaks of IBH caused by FAdV-8 have resulted in huge economic losses to the poultry industry worldwide.1,5,8,9,11–13,16,18–20 However, commercial tests specific for FAdV-8 and efficient vaccines against FAdV-8 are not available to date.
Fiber proteins located on the surface of virions not only play a vital role in mediating infection, but also induce virus-specific antibodies against FAdV.4,6,15 Therefore, fiber proteins could be used for developing serologic test reagents or for producing subunit vaccines against FAdV. Fiber-based ELISAs for the detection of antibodies against FAdV-1 or -4 have been reported. 2 However, an ELISA specific for the serologic detection of antibodies to FAdV-8 is not available.
To generate a rapid and specific ELISA for detection of antibodies against FAdV-8, we cloned the fiber gene of FAdV-8 into a pGEX-6P-1 vector, fused the gene with glutathione S-transferase (GST; ClonExpress II one step cloning kit; Vazyme Biotech, Nanjing, China), and developed primers specific for the FAdV-8 fiber gene (Table 1). PCR was performed (Phanta super-fidelity DNA polymerase; Vazyme Biotech). The PCR amplification mixture contained 38 μL of water, 5 μL of 10× buffer, 1 μL of 10 mM dNTP, 2 μL (10 μmol) of each primer, 1 μL of template (viral DNA or pGEX-6P-1 plasmid), and 1 μL of Phanta super-fidelity DNA polymerase. PCR conditions were: 1 cycle of 95°C for 5 min, 30 cycles of 95°C for 30 s, 55°C for 30 s, and 72°C for 2 min, and finally 1 cycle of 72°C for 10 min. The purified PCR product for the fiber gene was then ligated with the purified linear pGEX-6P-1 (Exnase II; ClonExpress II kit; Vazyme Biotech) and transformed into regular chemically competent cells as described previously. 17 Positive recombinants were confirmed by sequencing and designated as GST-fiber.
Primer sequences used to construct the recombinant GST-fiber.

To test whether the recombinant GST-fiber could efficiently express the fiber protein of FAdV-8, GST-fiber was transformed into competent BL21 (Escherichia coli strain BL21) cells, and the transformed BL21 cells were induced by 1 mM IPTG overnight at 16°C. The expression of GST-fiber fusion protein was analyzed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). GST-fiber fusion proteins were efficiently expressed as soluble fusion protein. The expressed GST-fiber proteins were then purified (Glutathione sepharose 4B; GE Healthcare Life Sciences, Uppsala, Sweden). The molecular weight of the purified GST-fiber fusion protein was ~ 84 kDa (Fig. 1A). To further confirm the expression and antigenicity of the GST-fiber fusion protein, anti–GST-tagged monoclonal antibody (ABclonal, Wuhan, China) and chicken sera seropositive (by IFA and western blot) to FAdV-8 were used as primary antibody for western blot analysis; GST-fiber fusion protein could be recognized efficiently (Fig. 1B, 1C). Hence, the GST-fiber fusion protein expressed here can be used as an antigen to develop a rapid serologic test for antibodies to FAdV-8.
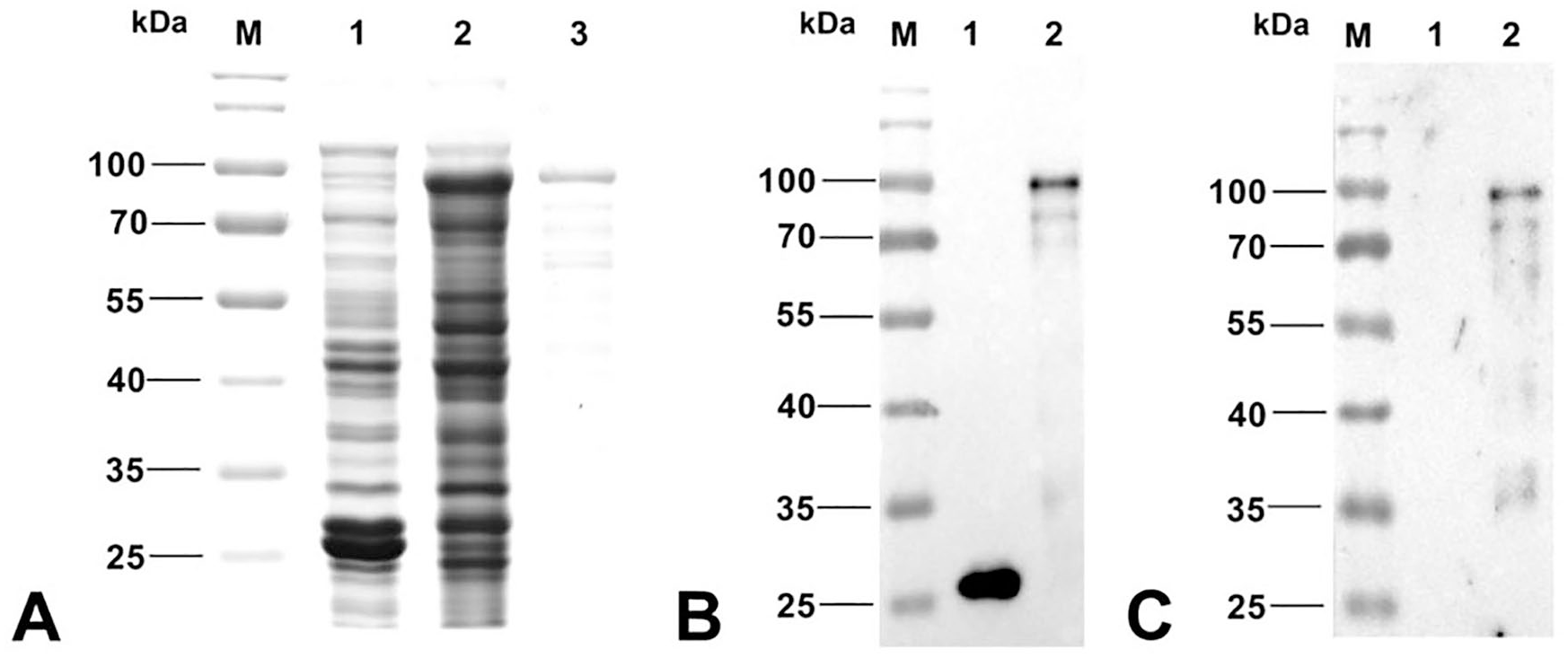
The expression and purification of GST-fiber fusion protein.
To develop a fiber-based ELISA for FAdV-8, 1 μg of the purified GST-fiber fusion protein/well (diluted in 0.1 M carbonate–bicarbonate buffer, pH 9.6) was used to coat ELISA plates overnight at 4°C; the plates were then blocked with 5% skimmed milk in phosphate-buffered saline with Tween 20 (PBST; pH 7.4, containing 0.05% Tween 20) for 1 h at 37°C. After the plates were washed once with PBST, 100 μL of sera diluted 1:400 in dilution buffer (PBST with 1/100 volume of 3 mg/mL of BL21 total lysate) were added to the plates. Dilution buffer, negative sera, and positive sera were set as blank, negative, and positive controls, respectively. Plates were incubated for 1 h at 37°C, followed by 3 washes with PBST. Then, 100 μL of rabbit anti-chicken antibody labeled with horseradish–peroxidase (diluted 1:50,000 in the dilution buffer) were added to the plates and incubated at 37°C for 1 h. After 3 more washes with PBST, 100 μL of freshly prepared substrate solution 3,3’,5,5’-tetramethylbenzidine was added to each well. Color development was performed in the dark for 10 min, and the reaction was stopped by addition of 50 μL of 2 M sulfuric acid. The absorbance values at 450 nm (OD450) were measured with an ELISA reader. When the OD450 value of the sample was > 0.184, the sample was regarded as positive. This cutoff was determined based on the negative control sera by calculating the arithmetic mean plus 3 × the standard deviation.
To evaluate the specificity of the ELISA for FAdV-8, we tested the ELISA with sera against different serotypes of FAdVs, including FAdV-1, -4, -5, -6, -7, -8a and b, -9, and -10, and sera against other avian pathogens including influenza A(H9N2) virus, chicken anemia virus, Marek disease virus (Mardivirus), Newcastle disease virus (Avian orthoavulavirus 1), avian reticuloendotheliosis virus (Reticuloendotheliosis virus), infectious bronchitis virus (Avian coronavirus), and egg-drop syndrome virus (Duck atadenovirus A). The above sera were generated either in our laboratory or commercially (Sinopharm Yangzhou VAC Biological Engineering, Yangzhou, China) by using the corresponding pathogens. The OD450 values of the chicken sera against FAdV-7, -8a, -8b were > 0.8, whereas those from other sera tested were all < 0.184 (Fig. 2A). Thus, the sera against FAdV-7, -8a, -8b were positive in our ELISA, and the sera against other viruses tested were all negative, demonstrating the specificity of our ELISA for FAdV-7, -8a, and -8b.
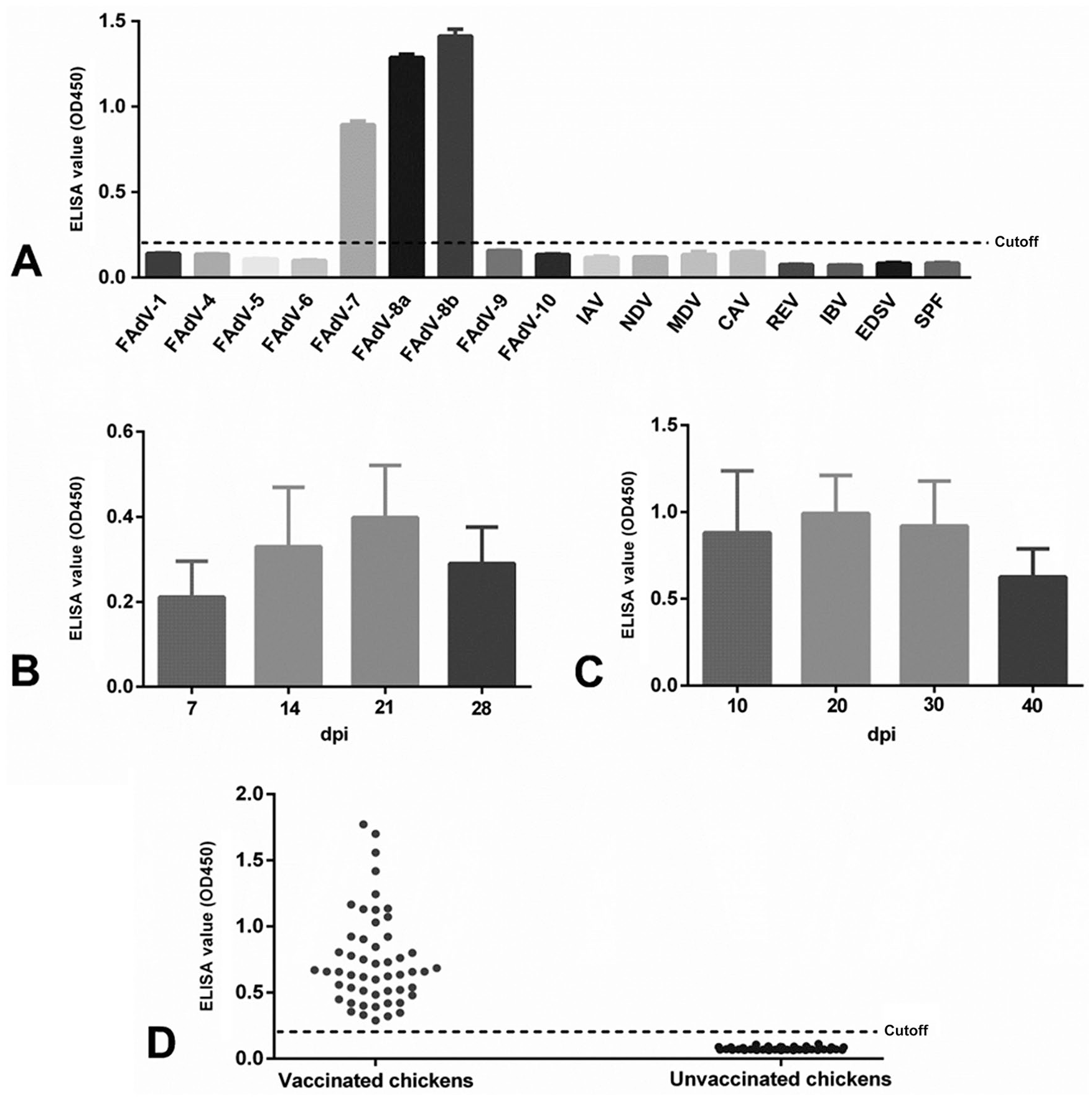
ELISA for detection of antibodies against fowl adenovirus serotypes 7 and 8 (FAdV-7, -8).
To further confirm the FAdV-7 and -8 specificity of our ELISA, the sera collected at different times from chickens experimentally infected with FAdV-7 and -8 were tested individually. All animal experiments were performed in accordance with institutional animal care guidelines and protocol 06R015, which was approved by the Animal Care Committee, Yangzhou University, China. The antibody against FAdV-7 could be detected efficiently as early as 7 d post-infection (dpi); the antibody level peaked at 21 dpi and then decreased gradually (Fig. 2B). Moreover, the antibody against FAdV-8 could be efficiently detected as early as 10 dpi; the antibody level peaked at 20 dpi and then decreased gradually (Fig. 2C). The antibody against the fiber protein of FAdV-7 or FAdV-8 was detected efficiently early after infection, and the effectiveness of detection lasted for ≥28 dpi for FAdV-7 and ≥40 dpi for FAdV-8, indicating the feasibility of using our ELISA for the detection of FAdV-7 and -8 infection.
To further test the sensitivity of our ELISA, antibody in 3 chicken sera positive against FAdV-8 (sera A–C) with serial dilutions (1:400; 1:800; 1:1,600; 1:3,200; 1:6,400; 1:12,800; 1:25,600; and 1:51,200) were detected successfully by both our ELISA and IFA (LMH cells [leghorn male hepatoma cell line] infected with FAdV-8 were used as antigen). Our ELISA was more sensitive than the IFA. The minimum titers of sera A–C for detection by our ELISA were 1:6,400, 1:25,600, and 1:3,200, respectively, whereas the titers of the sera for detection by IFA were 1:800, 1:1,600, and 1:800, respectively (Table 2).
Comparison of our ELISA and IFA results for detection of antibody against fowl adenovirus serotype 8 (FAdV-8) in 3 chickens (A–C).
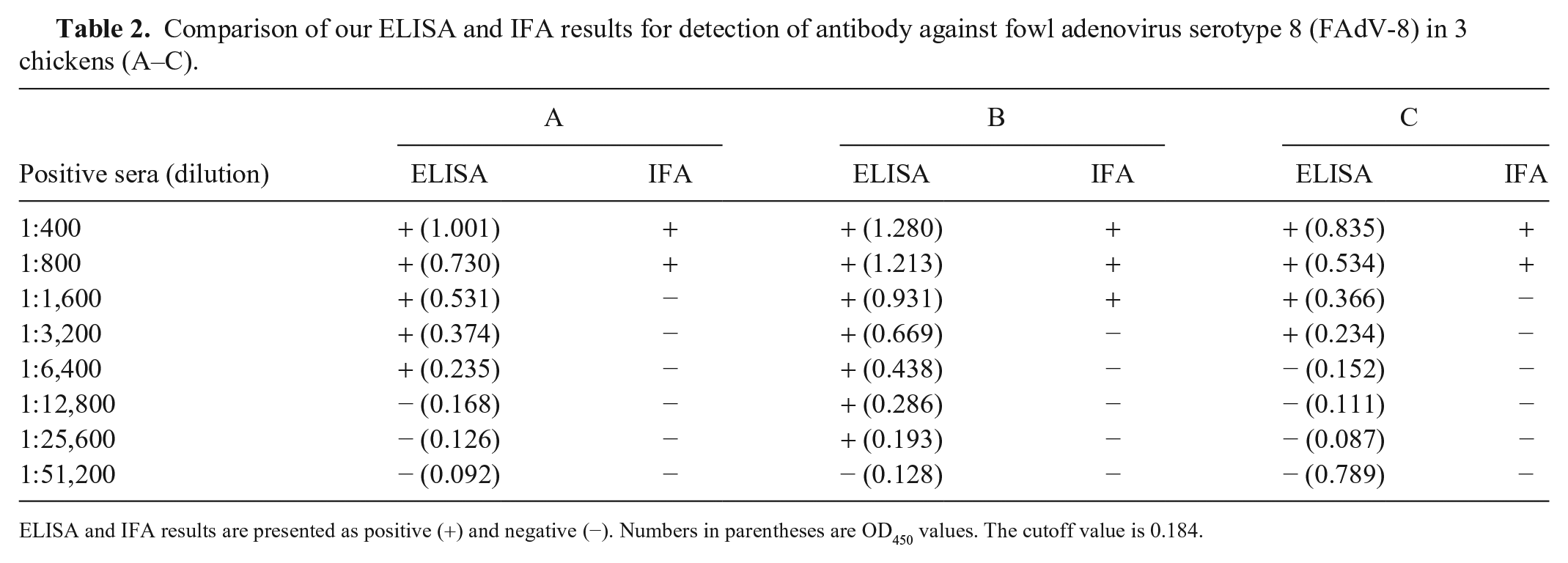
ELISA and IFA results are presented as positive (+) and negative (−). Numbers in parentheses are OD450 values. The cutoff value is 0.184.
To test whether our ELISA could be applied to monitor the level of FAdV-8 antibody in chickens vaccinated with inactivated FAdV-8 vaccine, 60 sera from a vaccinated chicken flock and 50 sera from an unvaccinated chicken flock were collected and tested. All 60 sera from the vaccinated chickens were positive in our ELISA, and all 50 sera from the unvaccinated chickens were negative. The mean OD450 from positive sera was 0.731, whereas that from negative sera was 0.077 (Fig. 2D). When checked by IFA, 10 of the 60 sera positive in the ELISA were negative. To further evaluate the 10 sera that were negative in the IFA, we performed a western blot assay. In the western blot, we used as antigen the lysates of the DF-1 cells (chicken fibroblast cell line) transfected with plasmid pcDNA3.1-fiber expressing the fiber protein of FAdV-8; 5 of the 10 sera were positive for the correct blot (58 kDa) with the lysates of the DF-1 cells transfected with pcDNA3.1-fiber, but not with the lysates of the control DF-1 cells. We also compared our ELISA with a commercial ELISA (BioChek, Reeuwijk, Netherlands) to test 18 positive and 18 negative clinical chicken sera and found that the correlation for positive sera and negative sera between the 2 methods was 94% (17 of 18) and 67% (12 of 18), respectively (Table 3). Given that the BioChek ELISA can detect antibodies against all 12 serotypes of FAdV, including FAdV-7 and -8, the low correlation for the negative sera indicates that the clinical chicken sera that were negative in our ELISA for detection of antibody against FAdV-7 and -8 may have contained antibodies against other serotypes of FAdV. The high correlation for the positive sera highlights the utility of our ELISA for detection of antibody against FAdV-7 and -8.
Comparison of results for our ELISA for fowl adenovirus serotype 8 (FAdV-8) and the BioChek ELISA.

Our ELISA results are presented as positive (+) and negative (−). Numbers in parentheses are OD450 values. The cutoff value is 0.184.
BioChek ELISA results are presented as positive (+) and negative (−). Numbers in parentheses are OD405 values. The cutoff value is 0.668.
Although a fiber-based fluorescent microsphere immunoassay (FMIA) has been reported as a novel multiplex tool for simultaneous detection of antibodies against FAdV, 3 the FMIA is expensive, and cannot differentiate the antibody to FAdV-8 from antibodies to other serotypes of FAdV. Similarly, the BioChek ELISA could not differentiate antibody to FAdV-8 from antibodies to other serotypes of FAdV. Notably, given that previous studies demonstrated that antibodies against the fiber proteins are closely related to the neutralizing antibody for FAdV, investigation is still needed into whether detection of antibody against the fiber protein with our ELISA can be an efficient indicator of the effectiveness of protection provided by FAdV-7 and -8 vaccination.
Footnotes
Acknowledgements
We thank Prof. Tuoyu Geng (Yangzhou University) for kindly providing the proofreading for our manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Jiangsu Agricultural Science and Technology Innovation Fund (CX(19)3026), Key Laboratory of Prevention and Control of Biological Hazard Factors (Animal Origin) for Agrifood Safety and Quality (26116120), and the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions.
