Abstract
An accurate and simple-to-perform new version of a competitive ELISA (cELISA) kit that became commercially available in 2015 for testing of cattle for antibody to Anaplasma marginale was validated for detection of Anaplasma ovis antibody in domestic sheep. True positives and negatives were identified using nested PCR (nPCR) as the gold standard. Negative bovine control sera supplied with the kit were used to calculate % inhibition (%I), designated bovine control ELISA (BcELISA), and this was compared to %I calculated from negative ovine sera derived from hand-raised, pathogen-free sheep, designated ovine control ELISA (OcELISA). The receiver operating characteristics area under the curve was 1.0 with a p value <0.001 regardless of the source of the control sera. The cutoff values for negative BcELISA and OcELISA were <30%I and <27%I, respectively. Our work confirmed that this Anaplasma antibody cELISA kit version 2 can be used with the serum controls supplied in the kit to test for A. ovis antibody in domestic sheep. Furthermore, this work confirmed the historically high infection prevalence (>93%) at the U.S. Sheep Experiment Station (Dubois, Idaho), in spite of efforts to reduce the possibility for iatrogenic transmission there, suggesting high levels of tick-borne transmission.
Anaplasma ovis is an obligate intraerythrocytic gram-negative bacterium vectored by ixodid ticks or transmitted mechanically by biting flies or fomites to domestic and wild sheep and goats.4,14 A. ovis infects these small ruminants in tropical, subtropical, and temperate areas worldwide, resulting in vast economic losses for sheep and goat producers as a result of anemia, abortion, decreased milk supply, weight loss, and even death of animals during acute disease.3,9,12,15 Animals that survive acute disease remain persistently infected for life at low levels that are undetectable by conventional examination of stained blood smears. 12 Most persistent infections are detectable only by sub-inoculation to a susceptible host, by PCR, or indirectly by serology. Consequently, serologic detection of A. ovis antibody is a key tool for producers to maintain healthy flocks 9 and to ensure that animals destined for movement or sale (especially for import or export) are free of infection. Given that seropositive animals are presumed to be infected, they can be isolated, treated, or culled to avoid transmission and the consequent spread of A. ovis within a flock, as well as to avoid the possible complications of acute disease.
Rapid, accurate, and inexpensive detection of A. ovis antibody is critical if producers are to minimize the prevalence of A. ovis in their flocks. Indirect fluorescent antibody (IFA) test and complement fixation were used historically for serologic detection of A. ovis. However, in our experience, these tests are complex, expensive, and produce subjective results, making them cumbersome to use outside of research settings. These tests also produce relatively high rates of false-positive or false-negative results. 7 Because A. ovis shares the major surface protein 5 (MSP5) epitope with Anaplasma marginale, a commercial competitive ELISA (cELISA) kit (Anaplasma antibody test kit, version 1, VMRD, Pullman, WA) used in the detection of A. marginale antibody in cattle was validated for the detection of A. ovis antibody in sheep in 2008. 10 This cELISA was relatively inexpensive and simple to run, but was reported to result in cross-reactivity with Ehrlichia spp.1,6 Also, because it had many steps, it has been our experience that this assay is challenging to perform with consistency. In 2015, a modified version of this cELISA, designated cELISAv2 (Anaplasma antibody test kit, version 2, VMRD), was developed, gained approval from the U.S. Department of Agriculture, and is now commercially available for use in detecting A. marginale antibody in cattle. Both the cELISA version 1 (v1) and v2 kits use recombinant MSP5 (rMSP5) to detect antibody to A. marginale. In the cELISAv1, the rMSP5 for coating the cELISA plates was expressed as a fusion with the maltose-binding protein (MBP), and, to reduce background reactivity, a MBP adsorption step was required. In the cELISAv2, the MBP was replaced with a glutathione S-transferase fusion protein to reduce the possibility for cross-reactivity with bacterial proteins and to avoid the necessity for the MBP adsorption step. These improvements produce fewer false-positive results 2 and, because it eliminates an entire step, this new kit is simpler and less time-consuming to use.
In order to validate this new version of the cELISA kit for detection of A. ovis, we tested domestic sheep from the flock at the U.S. National Sheep Experiment Station (USSES) in Dubois, Idaho. One EDTA and 1 coagulant tube of blood were drawn from each of 262 sheep in September 2014 and 125 sheep in February 2015 (387 samples in total). These 387 field samples were domestic Rambouillet, Texel cross, Suffolk, or Polypay ewes from this extensively managed range (Washington State University IACUC approval 04594 and USSES IACUC approvals 04-14 and 10-07). This location had been found to have a high prevalence of A. ovis infection in 2003. 10
In order to establish true-negative and true-positive sera for validation of the cELISAv2, a sample of USSES sheep were tested using nested PCR (nPCR) targeting the MSP5 gene. 11 Eighty-four of the sheep that tested nPCR positive for A. ovis were used as true-positives and 6 that tested nPCR negative were used as true-negatives. The remainder of the true-negative samples came from sera that had been obtained in 2006 from 105 specific-pathogen–free sheep at the Agriculture Agri-Food Canada Research Center (Lethbridge, Alberta, Canada), where A. ovis infections had never been detected. 10 DNA extracted from blood of these 105 sheep was tested at that time for A. ovis using the same nPCR assay 11 and at the same time were also confirmed to be negative by IFA test using the Idaho isolate of A. ovis. 13 All 105 Lethbridge samples tested negative for A. ovis.
Sera from both the Lethbridge flock and the USSES flock (195 in total) were tested using cELISAv2. Testing was completed using the manufacturer’s protocol included with the kits. Each test plate was run with both bovine and ovine sera negative controls, and samples were run in duplicate. For the purpose of our study, the cELISA calculations based on the bovine serum negative controls supplied with the kit are designated as the bovine control cELISA (BcELISA), and the calculations based upon the ovine serum negative controls are designated as ovine control cELISA (OcELISA). Ovine serum negative controls were bulk sera banked from specific pathogen–free sheep that were hand raised on our own research farm. 5
All analyses were performed utilizing GraphPad Prism 6 v.6.07 (GraphPad Software, La Jolla, CA) with significance levels set at 0.05. A receiver operating characteristic curve (ROC) was generated to determine sensitivity, specificity, and parametric cutoff points for samples run with either the bovine or the ovine negative control sera tested using the cELISAv2 kit.
The % inhibition (%I) cutoffs for both the BcELISA (<30%) and the OcELISA (<27%) classified true-positive and true-negative test samples with 100% specificity (95% CI: 96.7–100%) and 100% sensitivity (95% CI: 95.7–100%), respectively, giving perfect separation of samples (Fig. 1). The ROC curve confirmed these results for both assays with the area under the curve equal to 1.0 and p < 0.001. The %I cutoff value for calculations using bovine (BcELISA, kit-supplied) versus ovine (OcELISA, USDA-produced) negative controls differed by only a narrow margin, and both %I cutoffs (bovine control <30% and ovine control <27%) classified A. ovis samples as negative with high sensitivity and specificity.
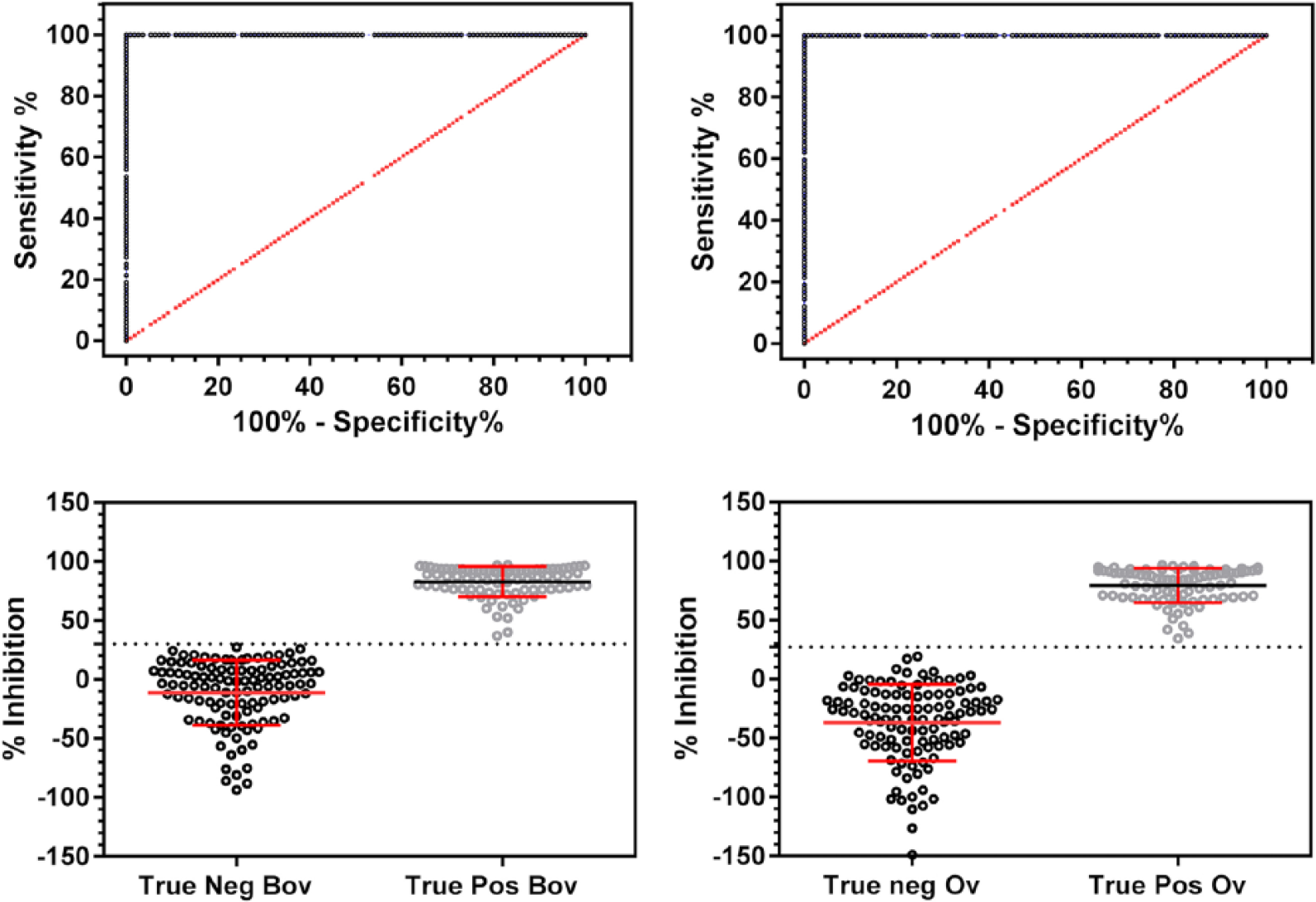
Receiver operating characteristic curves using 111 true-negative samples and 84 true-positive samples. We assessed the agreement between the test run with bovine negative control sera (BcELISA) supplied with the kit (left) and ovine negative control sera (OcELISA) from specific-pathogen–free sheep (right). Plots on the left indicate ELISA results calculated as described in the kit-supplied protocol using kit-supplied BcELISA, and plots on the right are calculated using OcELISA in place of the bovine serum controls. The line of no discrimination is shown as a broken red line at 45° angle. Frequency distributions show cutoffs for negative samples at <30% inhibition (%I) for BcELISA and <27%I for OcELISA with clear separation of samples.
We used 111 true-negative sheep samples, 84 true-positive sheep samples, and 387 field samples to validate the cELISAv2 kit for the detection of A. ovis in domestic sheep. Complete separation between the positives and negatives for both the BcELISA and the OcELISA occurred using a cutoff of 30%I for the former, and 27%I for the latter, a finding that clearly demonstrates the accuracy and usefulness of this improved test for A. ovis in domestic sheep for research or field applications.
Using this new cELISAv2, we determined that the prevalence of infection was 94.8% among the 387 sheep tested from USSES during late 2014 and early 2015. In 2003, 402 domestic sheep from the extensive grazing range of the USSES had a 93% prevalence of A. ovis when tested using the cELISAv1 that was commercially available at that time. 10 Starting in 2008, a pneumatic injection device was adopted for use in delivering vaccinations at USSES instead of the traditional practice of using a needle. 8 It was hoped that this new practice would reduce the opportunity for iatrogenic transmission, thus reducing prevalence. As of 2016, the last of the ewes that would have received a vaccination by needle at the station would have been removed from the flock.
Comparing the 2003 data with the 2014–2015 data shows that there has been no significant change in the prevalence of this infection in the intervening 11–12 y (Fisher exact test, p = 0.316). It appears that the force of transmission is sufficiently high at this location that efforts to reduce opportunities for iatrogenic transmission have had no effect on the prevalence of A. ovis, which has remained at an endemically stable high level. This suggests that there may be a high level of tick-borne transmission.
Our study also confirms that the bovine controls supplied with this commercial cELISAv2 kit for the detection of Anaplasma, which has been validated for use with cattle, can be used with confidence to determine %I and to confirm presence or absence of A. ovis antibody in sheep. Now that the cELISAv2 kit has been validated for detection of A. ovis antibody in sheep, this new detection tool, which is simpler and more accurate than the previously available test, will benefit sheep producers worldwide, allowing them to confidently evaluate their flocks for infection with ovine anaplasma, and thus market A. ovis test–negative animals as breeding and replacement stock.
Footnotes
Acknowledgements
VMRD Inc. in Pullman, WA provided cELISA kits for this work. We thank the following personnel: USSES—Mark Williams and Natalie Pierce for sheep sample-collection coordination and management; Boyd Leonard, Ella Ybarlucea, Harley Carpenter, Nicole Strong, and Tonya Thelen for sample collection and processing; ADRU—James Reynolds, Nic Durfee, and Sara Davis for laboratory technical support; James Allison, Lori Fuller, and Ralph Horn for traveling to help bleed sheep, and transport and ship samples; Maggie Highland for providing serum and blood for this project; and Lynn Herrmann for supplying serum from hand-reared, pathogen-free sheep. Lethbridge sheep samples were acquired with the assistance of Dr. Timothy Lysyk (Agriculture Canada, retired).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: At the time of the study, C Chung was an employee of VMRD Inc., Pullman, WA, which manufactures the cELISA kits tested in this study. However, C Chung did not participate in data acquisition, measurements, results, or interpretation of data. The use of product or firm names in this publication does not constitute an official endorsement by the USDA Agricultural Research Service. The USDA-ARS makes no warranties as to the fitness of the products or methodologies described on these pages for any particular purpose.
Funding
This work was supported by USDA ARS CRIS Projects 2090-32000-033-00D and 2090-32000-031-00D.
