Abstract
We report herein a fatal case of acute human orthopneumovirus (formerly respiratory syncytial virus) infection in a captive white-handed gibbon (Hylobates lar). Other members of the housing group had mild respiratory signs. Gross examination revealed bilateral pulmonary congestion and froth in the bronchi. Microscopically, the lungs had lymphocytic, neutrophilic infiltration of the interstitium and alveolar walls. There was necrosis of terminal bronchiolar epithelium and terminal bronchioles, and surrounding alveoli contained necrotic and exfoliated epithelial cells admixed with histiocytes and syncytial cells. Additional lesions included nonsuppurative meningoencephalitis, and epidermal hyperkeratosis and hyperplasia with syncytial cell formation. PCR screening for 12 human respiratory viruses was positive for orthopneumovirus in multiple tissues, including lung, and immunohistochemical staining for human orthopneumovirus detected viral antigen within bronchial epithelial cells. IHC and PCR for measles virus on preserved sections were negative. White-handed gibbons have not been previously reported as hosts for human orthopneumovirus, an important respiratory pathogen of both primates and humans.
A 16-mo-old male, white-handed gibbon (Hylobates lar) was presented with acute onset of depression, crusty nose, and labored respiration. This gibbon was housed with a 23-y-old male, 17-y-old female, and 4-y-old female. Keepers noted respiratory distress in the 16-mo-old male after exercise on the date of presentation; attitude and behavior had been normal the previous day. The troop of white-handed gibbons lived in an open indoor enclosure with 3 m distance to the public walkway. Approximately 10 mo prior to this respiratory event, the gibbon had a traumatic fall, resulting in a subdural hematoma; a computed tomography scan identified left-sided mastoiditis. Supportive care was provided on recommendation from a pediatric neurologist, and the gibbon made a full recovery with no lasting deficits. Otherwise, this animal had been apparently healthy. The gibbon and its social group were all immobilized for a complete physical exam, vaccinations, tuberculosis, blood testing, and microchipping 2 mo prior to the onset of respiratory signs. Nearly all values in the complete blood count (CBC) and serum chemistry analytes for the gibbon of this report were within reference intervals (RIs) for this species. Alkaline phosphatase (1,131 U/L; RI: 36–261 U/L) and phosphorus (2.26 mmol/L; RI:0.26–2.20 mmol/L) were elevated but considered normal values for an animal of this age (ZIMS expected test results for Hylobates lar, Species360 Zoological Information Management System, http://zims.Species360.org). No testing for exposure to viral pathogens was performed during this procedure.
Historically, laboratory testing for several viruses had been performed on blood from this group of gibbons during routine examinations, looking for a variety of human viral pathogens. The 23-y-old male and 4-y-old female tested positive for human orthopneumovirus (formerly known as respiratory syncytial virus;) on every titer obtained.1,7 The 17-y-old female tested negative for orthopneumovirus 7 y prior but seroconverted before titers were obtained 2 y later.
The gibbon of our report was manually restrained for examination, and loud auscultable rales were evident in all quadrants of the lungs. The gibbon also had a low-grade fever of 39.7°C (103.5°F). The animal was treated with meloxicam (0.2 mg/kg, route unknown; Metacam; Boehringer Ingelheim Vetmedica, St. Joseph, MO) and enrofloxacin (10 mg/kg, route unknown; Baytril; Bayer HealthCare, Shawnee Mission, KS) but was found dead 1 h after the examination.
Two of the remaining 3 white-handed gibbons (4-y-old female and 23-y-old male) had minor respiratory distress, nasal discharge, inappetence, and hyporexia within 48 h of this gibbon’s death. These animals were active and did not have labored breathing. The 4-y-old female was immobilized for a repeat pregnancy evaluation examination and to investigate a cough that had developed 48 h after the juvenile gibbon’s death. Induction was performed in a squeeze cage via hand injection of ketamine hydrochloride (9.84 mg/kg IM; ZooPharm, Windsor, CO) and dexmedetomidine (0.033 mg/kg IM; Dexdomitor; Orion, Espoo, Finland). She was intubated and maintained under anesthesia with sevoflurane gas (SevoFlo; Zoetis, Parsippany, NJ). All vital parameters were within normal limits and remained so throughout the procedure. Physical examination, thoracic radiographs, brief ultrasound, CBC, and serum chemistry panel were unremarkable. She was treated supportively with ceftiofur crystalline free acid (6.8 mg/kg SQ; Excede; Zoetis), meloxicam (0.16 mg/kg SQ; Metacam; Boehringer Ingelheim), and enrofloxacin (9.6 mg/kg PO q24h × 10 d; Baytril; Bayer HealthCare). Methylprednisolone acetate (2.5 mg/kg SQ; Depo-Medrol; Pfizer, New York, NY) was administered to treat possible airway inflammation that was suspected because of the cough.
The 23-y-old male was presented with a crusty nose and no other clinical signs 8 d after the juvenile gibbon died. Oral enrofloxacin and meloxicam were prescribed, but neither medication was reportedly consumed. The 17-y-old female did not receive any treatments during this period. Fifteen days after the death of the juvenile gibbon, the remaining members of the group had complete resolution of respiratory signs.
A postmortem examination of the 16-mo-old male gibbon was performed within 3 h of death. The gibbon was in good body condition. External examination revealed a 1-cm wound on the caudal aspect of the head and no other evidence of external trauma, with hemorrhage related to this wound. Internal gross examination revealed congested lungs and mild froth in the bronchi. The adrenal glands were slightly enlarged subjectively, but no measurements were performed. No other gross abnormalities were seen. Differential diagnoses were acute pneumonia or anaphylaxis.
Tissue samples were fixed in 10% neutral-buffered formalin, processed routinely, and 5-µm sections stained with hematoxylin and eosin. Histologically, the lungs had focally extensive interstitial pneumonia with necrotizing bronchiolitis. Lesions were characterized by infiltration of the interstitium and alveolar walls by lymphocytes and a few neutrophils. Terminal bronchioles and surrounding alveoli contained necrotic and exfoliated epithelial cells admixed with histiocytes and syncytial cells, leaving a denuded epithelial lining (Fig. 1). Rare intranuclear or intracytoplasmic eosinophilic-to-amphophilic inclusions were noted in epithelial cells, and hyaline membrane formation was prominent along the alveolar surfaces. Pneumocytes were frequently hypertrophied.
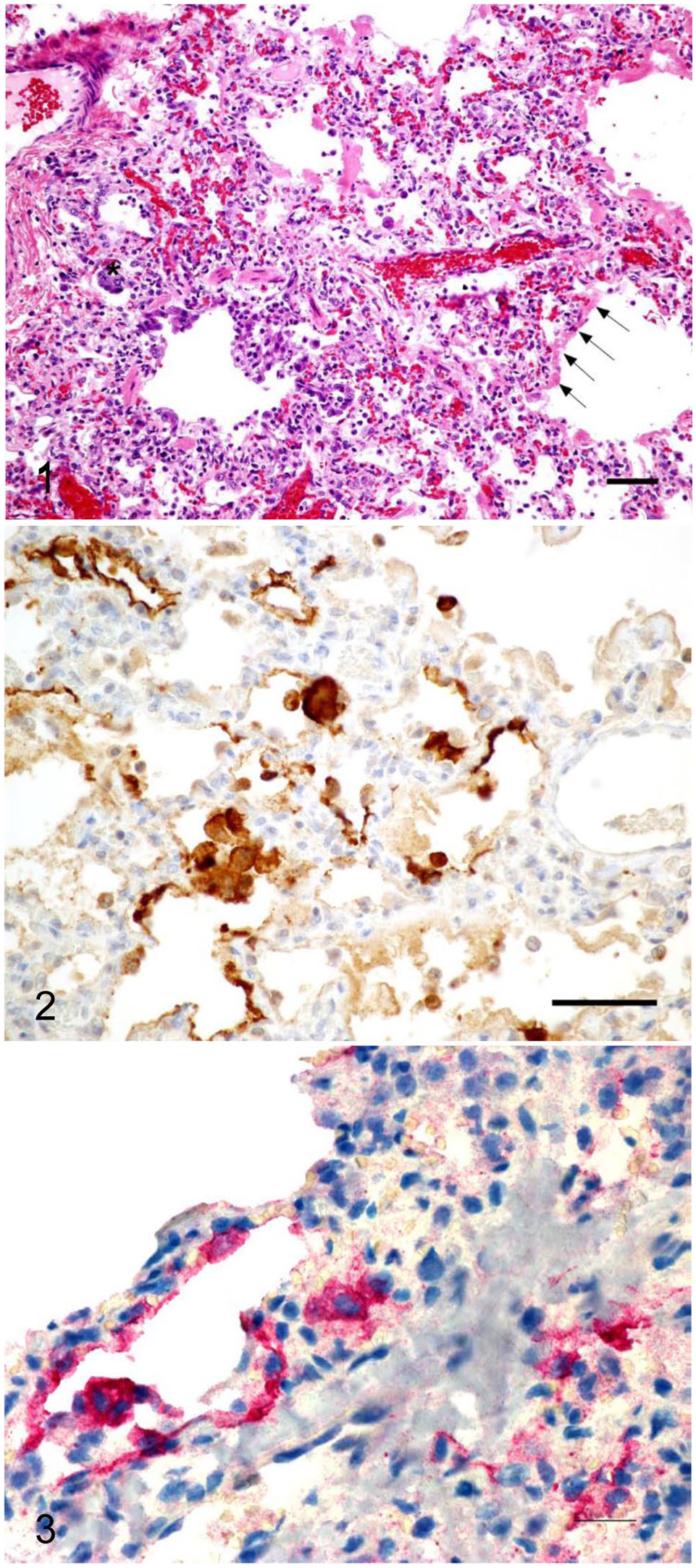
Interstitial pneumonia caused by acute human orthopneumovirus infection in a captive white-handed gibbon.
The liver had mild cholestasis and vacuolar change in the centrilobular regions. The skin had multifocal epidermal hyperkeratosis and hyperplasia with dysplastic changes in the follicular epithelium, with occasional intracytoplasmic eosinophilic inclusions and individual cell necrosis. Syncytial cells were present in one follicle. Sections of the brain at various levels had reactive gliosis and occasional glial cell necrosis. Rare intracytoplasmic and intranuclear inclusions were noted in glial cells. The small intestine had prominent enterochromaffin cells in the crypts with congested blood vessels. The colon had slightly increased numbers of lymphocytes and plasma cells in the lamina propria. All other examined tissues were within normal limits.
Histologic findings in the lung, skin, and brain were consistent with a viral infection. Samples of frozen spleen, liver, lung, and heart were submitted for testing by a real-time PCR (rtPCR) panel of 12 human respiratory viruses. All submitted samples tested positive for human orthopneumovirus by rtPCR. Other rtPCR tests for human metapneumovirus, influenza A and B viruses, human parainfluenza viruses 1–4 (Human respirovirus 1; Human orthorubulavirus 2, Human respirovirus 3, Human orthorubulavirus 4), human rhinoviruses A–C (Rhinovirus A–C), human coronaviruses, human adenoviruses (Human mastadenovirus A–G), and bocaparvovirus were negative. Samples of lung and heart contained abundant human orthopneumovirus; spleen and liver contained moderate amounts. Immunohistochemistry (IHC) used a goat polyclonal antibody to detect bovine orthopneumovirus antigen within bronchial epithelial cells (Fig. 2). 5 IHC was performed with a polymer-based, multi-step labeling protocol. The positive control was bovine lung tissue with histomorphologic and molecular evidence of bovine orthopneumovirus.
Given concerns for measles virus because of the reported skin lesion, additional testing was performed through multiple branches of the Centers for Disease Control and Prevention (Atlanta, GA). The Infectious Diseases Pathology Branch (IDPB) performed measles IHC on stomach, lung, and intestines; the test was negative. RNA was extracted from a formalin-fixed, paraffin-embedded tissue (FFPE) block containing sections of stomach, intestines, and lung at IDPB and shared with the Viral Vaccine Preventable Diseases Branch for measles PCR. 9 All sections of stomach, intestines, and lung tested negative for measles virus. Additional testing at IDPB included IHC and PCR for human orthopneumovirus on the tissue block containing lung, stomach, and intestine. IHC utilized goat polyclonal antibodies raised against human orthopneumovirus and a polymer-based detection system; the positive control was human orthopneumovirus in cell culture. PCR for orthopneumovirus was performed targeting the M gene. 6 Both IHC and PCR were positive, which further confirmed the diagnosis of acute human orthopneumovirus infection (Fig. 3).
Fresh lung tissue was submitted for aerobic and anaerobic bacterial culture. Aerobic culture grew 1+ Escherichia coli and Enterococcus spp., which were likely contaminants and not the cause of the reported clinical signs. Anaerobic culture did not grow any bacteria.
Species Human orthopneumovirus (order Mononegavirales, family Pneumoviridae) is a single-stranded RNA virus and the most commonly reported cause of bronchiolitis and viral pneumonia in human infants, but disease can occur at any age. 13 Transmission between humans occurs via large aerosol droplets or secretions. 13 Natural infections of human orthopneumovirus have been reported in chimpanzees, southern muriqui, and western lowland gorillas, resulting in peracute illness and, in some cases, mortality, especially in young animals.2,3,8,10–12,14 Experimental infection in rhesus macaques, African green monkeys, and owl monkeys with large doses of virus have resulted in few to no clinical signs of disease, and reports of natural clinical infection of human orthopneumovirus in Asian primates have not been identified.10,13 It has been reported previously that Old World primates, such as captive chimpanzees, gorillas, and orangutans, have a high seroprevalence for human respiratory pathogens. 2 However, gibbons have not been reported previously to become infected with orthopneumovirus.
Histologic findings in the gibbon of our report were consistent with those seen in human infection with human orthopneumovirus. These findings include bronchitis, bronchiolitis, interstitial pneumonia, syncytial cells, and intracytoplasmic and intranuclear inclusion bodies. 12 Extrapulmonary orthopneumovirus infection has also been reported in human infants but not great apes, most frequently seen as myocardial disease or hepatitis. 4 An infection with human orthopneumovirus was confirmed by PCR at multiple institutions in various tissues. IHC using an antibody raised against human orthopneumovirus revealed antigen in the lungs associated with epithelial cell necrosis. Antemortem samples required to attempt a diagnosis of orthopneumovirus can be obtained from the upper respiratory tract (nasal aspirate or swab) or from the lower respiratory tract (bronchoalveolar lavage, bronchoaspirate, tracheal aspirate). 7 One report gave conscious chimpanzees sterile gauze to chew on which was returned for analysis. 14 These samples should be analyzed with PCR or immunofluorescent antigen detection of orthopneumovirus. 7 The IHC results in combination with the clinical signs, microscopic lesions, and the PCR data strongly support the conclusion that the gibbon group in our case became acutely ill as a result of human orthopneumovirus infection.
We have described acute fatal anthroponotic respiratory syncytial virus infection in a white-handed gibbon. The rest of the group had respiratory signs, but no other fatalities were reported. The peracute nature of the infection in the individual that is the focus of our report precluded successful treatment, which suggests a more aggressive and systemic disease process than previously reported in other ape species. To our knowledge, natural human orthopneumovirus infection has not been reported in white-handed gibbons. A specific source of the virus could not be identified given the unlikelihood of identifying a human host of human orthopneumovirus during active infection.
Our case exemplifies the balance that zoologic institutions must achieve in providing for the safety of animals in their collection and accessibility to the public. It also informs the clinician that human orthopneumovirus infection should be a differential diagnosis for white-handed gibbons with acute respiratory signs.
Footnotes
Acknowledgements
We thank multiple individuals from the Centers for Disease Control and Prevention: Adam K. Wharton of the Viral Vaccine Preventable Diseases Branch performed the measles virus RT-rtPCR; Amy M. Denison of the IDPB performed molecular testing for orthopneumovirus and parainfluenza; Luciana Silva-Flannery, also of IDPB, performed IHC for human orthopneumovirus. We also thank Amanda Hand for her help in the editing of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
