Abstract
Mycoplasma hyopneumoniae is the causative bacterium of porcine enzootic pneumonia and one of the primary etiologic agents of the porcine respiratory disease complex. Most Brazilian commercial pig farms are positive for this pathogen. However, the prevalence of the pathogen in backyard pig farms has not been described, to our knowledge. Therefore, we aimed to determine the prevalence of M. hyopneumoniae in backyard pig farms in the state of Paraná, Brazil. In January–March 2020, we collected 585 serum samples from pigs in 187 non-vaccinated herds. We tested the sera with an indirect ELISA for anti–M. hyopneumoniae antibodies and found that 182 of 585 (31.1%) samples were positive, and were found in 109 of 187 (58.3%) herds assessed.
The increase in demand for pork has been addressed by increasing the number of farmed animals and improving the efficiency of pork production. However, larger herd sizes have led to the emergence of many enzootic diseases. Notably, the bacterium Mycoplasma hyopneumoniae stands out among pathogens causing respiratory diseases in swine. M. hyopneumoniae is the primary agent of porcine enzootic pneumonia (PEP) and contributes significantly to the porcine respiratory disease complex (PRDC). 5 PEP causes high morbidity across affected herds, is capable of widespread transmission to swine of all ages, and results in substantial losses associated with secondary infections.5,9
The key clinical sign of PEP is a mild and chronic non-productive cough. Autopsies reveal pulmonary lesions characterized by consolidation, primarily in the cranioventral areas of the lungs. 13 Antibodies induced by M. hyopneumoniae vaccination can be detected 3–4 wk after vaccination; seroconversion in natural M. hyopneumoniae infections usually occurs at 8–24 wk of age. 12 The indirect ELISA for M. hyopneumoniae antibodies is simple to perform, highly sensitive and specific, and one of the most recommended tests. 13
In Brazil, pigs on an estimated 95% of commercial farms test positive for M. hyopneumoniae, presenting a significant challenge to eradication efforts given the persistence of the bacteria within the swine respiratory tract. 16 Unlike the practices of pig farming in the developed swine industry in which pigs are raised on a large scale in many regions of the world, backyard pigs are traditionally kept in small homesteads, often to meet family needs. These farms typically operate under the radar and evade oversight by veterinarians, have low biosecurity measures, feed swill, rely on natural breeding, and engage in unregulated trade. 14 Consequently, given the unknown health status of these backyard farms, they represent a gap in the epidemiology of M. hyopneumoniae infection and other swine diseases.6,11
Paraná State plays an important role in the Brazilian swine industry, having produced an impressive 936,474 tons of pork in 2019 (= 21% of Brazil’s production) and hosting an average of 623,541 breeding sows. 8 Given the lack of research on the presence and dynamics of M. hyopneumoniae in small-scale swine operations, we aimed to identify the prevalence of M. hyopneumoniae in backyard pig farms in Paraná, Brazil. In January–March 2020, the Agricultural Defense Agency of Paraná (ADAPAR) collaborated with our research and collected 585 serum samples from swine of various ages and sex, known to be unvaccinated for M. hyopneumoniae, on 187 backyard production farms. Among these herds, 161 had their locations recorded, covering 121 cities in Paraná, Brazil (Fig. 1); the locations of the remaining 26 backyard farms were not specified.
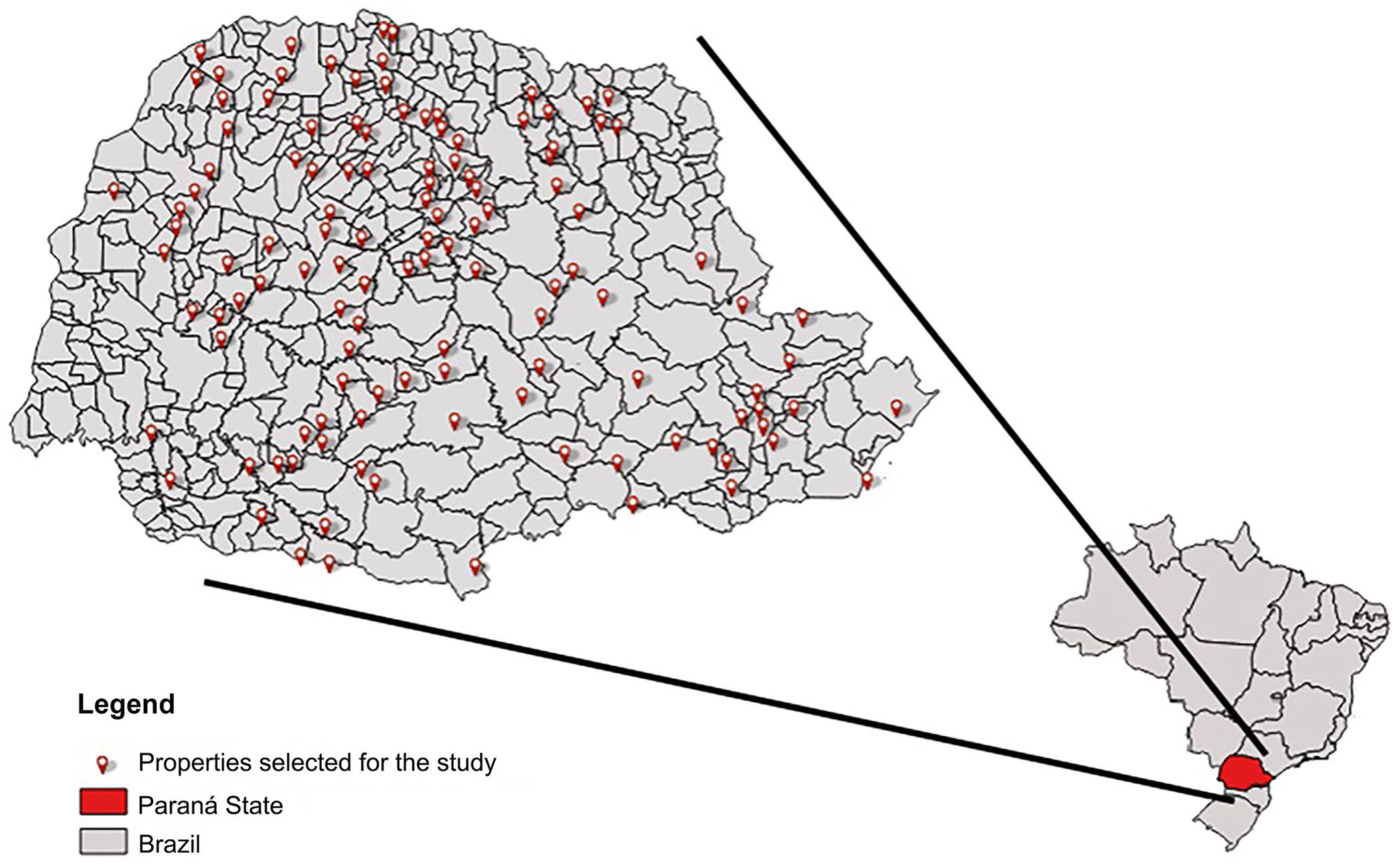
Location of the backyard pig farms sampled in the state of Paraná, Brazil.
The number of animals utilized in our study adhered to a non-probabilistic convenience sampling method. The selection criteria were confined to adult swine >8-mo-old or those in the reproductive stage, with at least 1 animal sampled per herd. The total number of samples was determined using Epi Info v.7.2.5.0 (https://www.cdc.gov/epiinfo/index.html), accounting for a total population size of 4,917 animals. The estimated expected frequency was 65.9%, based on a previous study. 15 With an anticipated 5% margin of error, a design effect of 1, and 187 clusters, the calculations for a herd confidence level of 95% amounted to a minimum of 374 animals evaluated to ensure the statistical validity of our study.
All procedures were conducted with the approval of the School of Agricultural and Veterinary Sciences’ Animals Ethics Committee (CEUA; permit 21/001469). Blood samples were collected from the jugular vein with sterile disposable syringes and needles and transferred to vacuum tubes devoid of anticoagulants and equipped with clot activators (Becton Dickinson). For serum separation, samples were centrifuged at 1,500 × g for 10 min, and sera were carefully distributed into graduated plastic microtubes (Eppendorf). Frozen sera were transported to the Swine Medicine Laboratory (School of Agricultural and Veterinary Sciences, São Paulo State University [UNESP], Jaboticabal, Brazil) and stored at –20°C until analyzed.
Anti–M. hyopneumoniae antibodies were detected with a 2-well indirect ELISA (Civitest Suis Mhyo; HIPRA), a test with high specificity and sensitivity under field conditions, 9 following the manufacturer’s instructions. One well is coated with a M. hyopneumoniae–specific antigen and the other with a generic mycoplasma antigen; the result is determined as the proportion of M. hyopneumoniae–specific antibodies in the sample.
Briefly, the validation of each plate was conducted according to the manufacturer’s parameters: the mean optical density (OD) of the positive control (CTR+) was assessed to be ≥0.6; the difference between the mean OD of the positive control and the mean OD of the negative control (CTR+ minus CTR–) was assessed as ≥0.5; the difference between the OD of the positive diluent control and the OD of the negative diluent control was evaluated. The value of index reactivity with positive control (IRPC) was calculated for each sample. An IRPC <30 indicated a negative result; values of 30–35 were considered uncertain; and values >35 were positive. The results were tabulated using Excel (v.16; Microsoft), and the frequencies were calculated with the aid of Epi Info.
Serum antibodies against M. hyopneumoniae were detected in 182 of 585 (31.1%) serum samples collected from swine, with positive sera in 109 of 187 (58.3%) evaluated backyard farms. The ELISA result can be false-positive in sera from vaccinated animals. Given that our samples were from pigs in backyard herds presumed to not have a vaccination protocol, we infer that the detection of antibodies is due to infection with the pathogen.
The transmission of M. hyopneumoniae primarily occurs through direct contact; indirect and aerosol transmission may also play a role in infection. Reinfection in farms free of the pathogen is common, as studies acknowledge the possibility of this bacterium being airborne, potentially spreading 4.7–9.2 km under favorable conditions of humidity, temperature, and wind speed.1,2,7 Pigs in a backyard farm could infect others through airborne dissemination; however, this is merely speculation, as under ideal conditions, the climate in the state of Paraná has low temperatures (~7°C).2,7 It is prudent to maintain vigilance over the risks associated with backyard swine farming and to implement rigorous biosecurity measures and disease surveillance programs to prevent the introduction and spread of M. hyopneumoniae and other pathogens.
Eradication strategies for M. hyopneumoniae are based on vaccination, antimicrobial therapy, air filtration, and movement restrictions between farms, and have shown success in geographically confined herds in Finland, Switzerland, and the United States.4,10 However, in backyard farms in Paraná, the eradication of this agent is more complex, and these herds can perpetuate the pathogen reinfection cycle. Lack of biosecurity programs, including the buying and selling of infected animals, among these rural producers can contribute to the maintenance of the pathogen.2,3,14
The detection of antibodies against M. hyopneumoniae in backyard-raised and non-vaccinated swine herds in Paraná, Brazil, demonstrates the circulation of this agent within this rearing system. This underscores the importance of including these populations in the development of strategies for the control and eradication of M. hyopneumoniae in commercial farms.
Footnotes
Acknowledgements
We thank the Agricultural Defense Agency of Paraná (ADAPAR) for the blood serum samples. We also thank CNPq for the productivity grant to Luís G. de Oliveira (CNPq Process 316447/2021-8).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
We thank the São Paulo State University Entrance Examination Foundation for the scholarship granted to Ana C. A. Lima.
