Abstract
No prognostic tools for the prediction of COVID-19 pneumonia severity and mortality are available. We explored whether CURB-65, PSI, and APACHE-II could predict COVID-19 pneumonia severity and mortality. We included 167 patients with confirmed COVID-19 pneumonia in this retrospective study. The severity and 30-day mortality of COVID-19 pneumonia were predicted using PSI, CURB-65, and APACHE-II scales. Kappa test was performed to compare the consistency of the three scales. There was a significant difference in the distribution of the scores of the three scales (P < 0.001). Patients with PSI class ⩽III, CURB-65 ⩽1, and APACHE-II-I all survived. The ROC analysis showed the areas under the curve of the PSI, CURB-65, and APACHE-II scales were 0.83 (95% CI, 0.74–0.93), 0.80 (95% CI, 0.69–0.90), and 0.83 (95% CI, 0.75–0.92), respectively. Our findings suggest that PSI and CURB-65 might be useful to predict the severity and mortality of COVID-19 pneumonia.
Coronavirus disease 2019 (COVID-19) is caused by a novel enveloped RNA beta coronavirus. 1 COVID-19 symptoms worsen rapidly and high mortality has been reported globally. 2 The World Health Organization (WHO) has declared a public health emergency of international concern over the global outbreak of COVID-19. 3 As of 30 May 2021, there were 169,597,415 confirmed patients of COVID-19 and 3,530,582 deaths worldwide. 4
It is important to evaluate the severity of COVID-19, since the outcomes of non-severe patients were favorable compared to severe patients. A reliable tool of assessing the severity of pneumonia can help clinicians better estimate the prognosis of their patients and therefore a more suitable treatment approach can be decided. Pneumonia Severity Index (PSI) and CURB-65 (confusion, urea, respiratory rate, blood pressure, age ⩾65) are commonly used to evaluate the severity of community and hospital-acquired pneumonia.5,6 Moreover, a previous study showed the CURB-65 scale could predict the mortality of community-acquired pneumonia (CAP). 7 The Acute Physiology and Chronic Health Evaluation (APACHE) II system is widely applied in the intensive care unit (ICU). 8 Several studies have evaluated common intensive care severity scores to assess COVID-19 pneumonia severity and mortality. Artero et al. showed that performance of PSI and CURB-65 were better than quick Sequential Organ Failure Assessment (qSOFA: respiratory rate≥22/min, altered mentation, or systolic blood pressure ≤100 mmHg) at predicting mortality in patients with COVID-19 pneumonia. qSOFA score is simple, and qSOFA score≥2 predicted the severity of COVID-19, with a sensitivity of 26.7%. 9 The study by Myrstad et al. 10 showed that National Early Warning Score 2 (NEWS2) score was superior to qSOFA, Systemic Inflammatory Response Syndrome (SIRS) criteria and CRB-65 in predicting COVID-19 pneumonia severe and mortality. The finding from Ji et al. 11 suggested that the CALL (comorbidity, age, lymphocyte, and Lactate Dehydrogenase; LDH) score could predict the progression risk in patients with COVID-19 pneumonia. However, Ihle-Hansen et al. 12 found CRB-65 and qSOFA scoring systems were not suitable for evaluating the severity and mortality of COVID-19. Satici et al. 13 showed that PSI performed better than CURB-65 in assessing the mortality of COVID-19, and adding CRP levels to PSI did not improve the prediction of 30-day mortality. Thus the performance of these scoring systems in evaluating the severity and mortality of COVID-19 pneumonia is controversial. In this cross-sectional study, we aimed to test the hypothesis that PSI, CURB-65, and APACHE-II scales are associated with the severity and 30-day mortality in patients with COVID-19 pneumonia.
Methods
Study population
Patients with confirmed COVID-19 pneumonia who were admitted to Tumor Center of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology (Wuhan, China) from February 15, 2020 to March 17, 2020 were included in this retrospective study. A confirmed case of COVID-19 was defined as a positive result on real-time reverse transcription-polymerase chain reaction (RT-PCR) assay of respiratory specimens. 14 Our study was conducted in line with the Declaration of Helsinki. The Fujian Medical University Union Hospital Ethics Committee approved the study protocol (2020XGFKY002) and written informed consent was obtained from a legally authorized representative for each patient. All hospitalized patients were treated according to the COVID-19 Diagnosis and Treatment Protocol issued by National Health Commission of China. 15 Patients who were younger than 18 years, had a DNR (Do Not Resuscitate) order or were pregnant were excluded. The patient flow chart is shown in Figure 1.
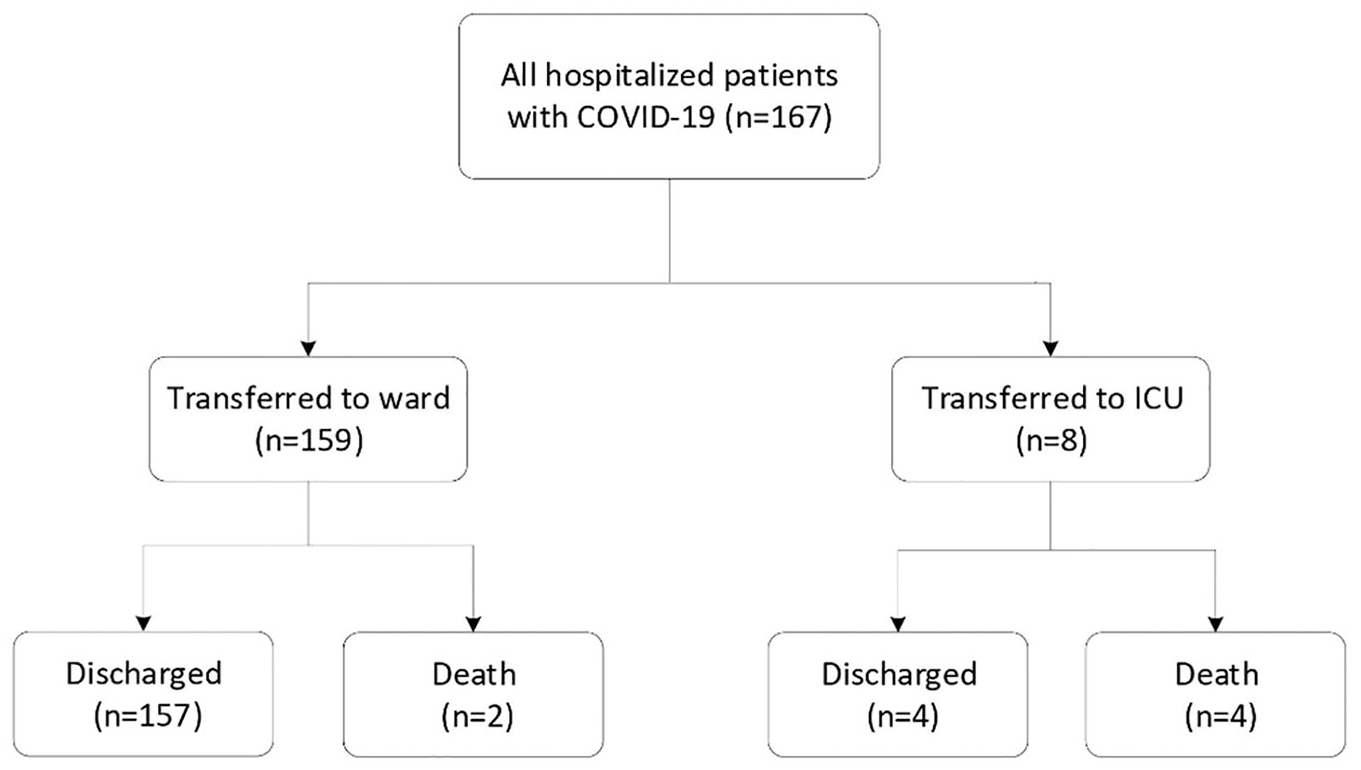
Patient flow chart.
Data collection
Clinical records were reviewed and retrospectively collected by J.C and S.R. The epidemiological, demographic, clinical, and laboratory data on admission and during treatment were extracted using a standardized data form. Epidemiological and demographic data included age, sex, profession, and comorbidities (malignant tumors, congestive heart failure, cerebrovascular disease, kidney disease, and liver disease). Clinical profile of the COVID-19 patients included mental status, heart rate, respiratory rate, blood pressure, body temperature, and radiographic signs of pleural effusion. Laboratory findings included arterial pH, blood urea nitrogen, glucose, hematocrit, and partial pressure of arterial O2 within 24 h after COVID-19 diagnosis.
Two physicians (G.L and R.C) reviewed the chest CT images independently. In cases of disagreement, a consensus was reached after discussion with L.C, a senior respiratory physician and H.L, a senior radiologist. We contacted attending doctors and other healthcare providers when data were missing or questionable. Two clinicians (J.C and S.R) assessed the PSI, 16 CURB-65, and APACHE-II scales independently. A consensus was reached by discussion when a disagreement occurred.
PSI is a scale containing 19 different variables, 91 points (risk classes IV–V) is the cut-off value for PSI, 17 which means a poor prognosis. We defined a cutoff value ≥91 for severe COVID-19 pneumonia. CURB-65 includes five variables (confusion, BUN >7 mmol/l, respiratory rate ≥30 bpm, systolic blood pressure <90 mmHg or diastolic blood pressure ≤60 mmHg, and age ≥65 years). Three or more positive variables indicate a poor prognosis. 18 We defined a cutoff value ≥3 for severe COVID-19 pneumonia. APACHE II score is widely applied in ICU. A score ≥15 indicates critically ill patients. In the study of Huang et al., 19 APACHE II scores at cutoff values ≥10 achieved a higher accuracy in predicting mortality. We divided the APACHE II score into three levels (<10, 10–14, and ⩾15).
Outcomes
The patients were divided into non-survivor group and survivor group. In addition, they were divided into non-severe group and severe group. The primary outcome of the study was severe COVID-19 defined as any of the following: respiratory rate >30 bpm; severe respiratory distress; or SPO2 ⩽93% on room air, which was based on the guideline of Clinical Management of Severe Acute Respiratory Infection When COVID-19 is suspected published by WHO. 20 The secondary outcome was 30-day mortality. Patients who were discharged after recovery or transferred to another hospital or treated for more than 30 days were considered as survival.
Statistical analysis
We summarized data with mean value with standard deviations or median value with interquartile range, and categorical data as counts with percentages. We used the T-test or Mann-Whitney test to compare the differences in continuous variables, and the Chi-square test or Fisher’s exact test to compare the differences in categorical variables where appropriate. To compare the consistency of the three scales, we divided the scales into three levels of mild, medium, and severe (Supplemental Table S2). Kappa test was performed to compare the consistency of the three scales. The receiver operating characteristics (ROC) and the area under the curve (AUC) were described to assess the association between the severity of COVID-19 and three scales.
Results
A total of 167 patients with confirmed COVID-19 pneumonia were included in this retrospective study. The difference in the components of three scales between non-survivors and survivors, and non-severe patients and severe patients are shown in Table 1. The overall 30-day mortality rate was 3.59% (6/167). The non-survivors were more likely to have previous cerebrovascular disease, lower systolic pressure, lower arterial PH, lower partial pressure of arterial O2, and blood urea nitrogen ⩾7 mmol/l. Compared to patients with non-severe COVID-19, patients with severe COVID-19 were more likely to have previous cerebrovascular disease, higher heart rate, higher breathing rate, lower systolic pressure, lower arterial pH, lower partial pressure of arterial O2, and blood urea nitrogen ⩾7 mmol/l.
The difference in the components of three scales between non-survivors and survivors, non-severe patients and severe patients.

Clinical characteristics and laboratory findings of patients with COVID-19 pneumonia are shown in Table 2. Primary onset symptoms were similar between survivors and non-survivors, and between patients with severe disease and non-severe disease. Compared to survivors, non-survivors were more likely to have higher blood urea nitrogen (mmol/l) (9.16 [7.35, 11.25] vs 4.30 [3.45, 5.40], P < 0.001) and lower lymphocyte count (×109) (0.30 [0.18, 0.68] vs 1.35 [0.93, 1.68], P = 0.003). Similarly, patients with severe COVID-19 were more likely to have lower blood lymphocyte count (×109) (0.85 [0.64, 1.27] vs 1.37 [1.06, 1.72], P < 0.001), higher serum interleukin-6 levels (pg/ml) (15.22 [11.19, 29.62] vs 6.41 [4.51, 13.76], P < 0.001), and higher serum interleukin-10 levels (pg/ml) (4.61 [3.90, 5.58] vs 4.20 [3.41, 4.87], P = 0.013) compared to the non-severe patients (Table 2).
Clinical characteristics and laboratory findings of patients with COVID-19 pneumonia.

We found a significant difference in the distribution of the three scales in patients with different outcomes and disease severity (P < 0.001, Table 3). No patients with PSI class ⩽III, CURB-65 ⩽1, and APACHE-II-I died, while 1 (16.7%) and 3 (83.3%) patients with PSI-IV and PSI-V died, respectively. The mortality rate in patients with a CURB-65 score of 2 and ⩾3 was 16.7% (1/6) and 83.3% (5/6), respectively. For patients with APACHE-II class II and class III the mortality rates were 16.7% (1/6) and 83.3% (5/6), respectively.
Distribution of three scales in patients with different outcomes and disease severity.

Fisher’s exact test.
the number of cases with PSI-I was 0.
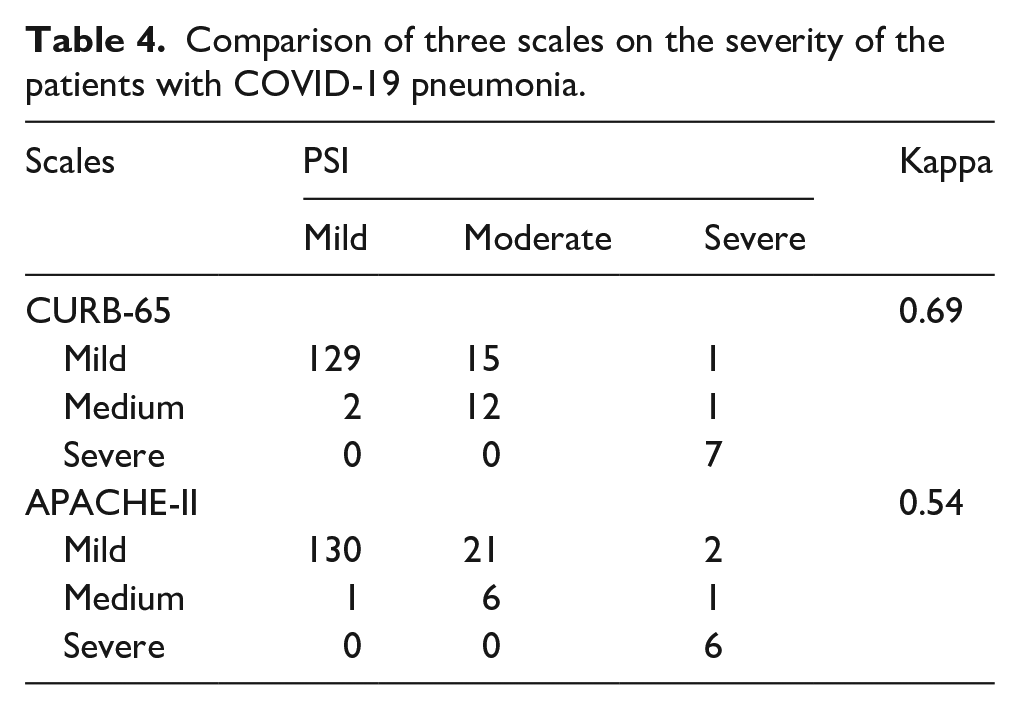
PSI and CURB-65 had substantial consistency in the evaluation of patients’ disease severity (Kappa = 0.69, P < 0.001) (Table 4). However, PSI and APACHE-II had moderate consistency (Kappa = 0.54, P < 0.001). During the matching process, the results revealed that 5 (83.3%) patients who died during hospitalization had higher scores on all three scale systems.
Comparison of three scales on the severity of the patients with COVID-19 pneumonia.
The performance of PSI, CURB-65, and APACHE-II scales to assess the severity of COVID-19 pneumonia are shown in Table 5. PSI had higher sensitivity but lower specificity than CURB-65. APACHE-II had the lowest sensitivity. The highest Youden index was found at the cut off of IV in PSI (0.62), ⩾2 in CURB-65 (0.55), and ⩾II in APACHE-II (0.36). The receiver operating characteristic (ROC) curves of the three scales are shown in Figure 2. The areas under the curve in the ROC analysis of PSI, CURB-65, and APACHE-II were 0.83 (95% CI, 0.74–0.93), 0.80 (95% CI, 0.69–0.90), and 0.83 (95% CI, 0.75–0.92), respectively.
The ability of three scales to evaluate the severity of COVID-19 pneumonia.

LR: positive likelihood ratio; −LR: negative likelihood ratio; YI: Youden index = (sensitivity + specificity) −1.

Receiver operator characteristic curve (ROC) and area under the curves (AUC) for the severity of COVID-19 pneumonia patient for three scales.
Discussion
In the present study, we found a significant difference in the distribution of the scores of the PSI, CURB-65, and APACHE-II scales in patients with different outcomes and disease severity. Moreover, PSI and CURB-65 both performed well in the evaluation of patients’ disease severity (Kappa = 0.69, P < 0.001). Our results are in consistency with those reported by Artero et al. 9 and Satici et al. 13 Thus our findings suggest that PSI and CURB-65 might be useful to predict the severity and mortality of COVID-19 pneumonia.
A previous study showed that patients with COVID-19 pneumonia who had compromised respiratory status on admission were at a higher risk of developing severe disease and worse outcomes. 21 Identifying the seriousness of COVID-19 is essential for clinicians to make decisions whether or not to take more intensive supportive treatment for the patients. 22 PSI, CURB-65, and APACHE-II scales have been well certified and widely applied for assessing pneumonia and general critical illness.7,19,23 In our study, we found that no patients with PSI class ⩽III (131/167, 78.4%), CURB-65 ⩽1 (145/167, 86.8%), and APACHE-II-I (153/167, 91.6%) died, the mortality rate in patients with PSI class IV, CURB-65 = 2, and APACHE-II class II is 16.7%, and the mortality rate in patients with PSI class V, CURB-65≥3, and APACHE-II class III is 83.3%. There was a significant trend of increasing mortality risk with a higher PSI, CURB-65, and APACHE-II risk class. Thus, our findings suggest that these three scales are useful tools to predict the mortality in patients of COVID-19 pneumonia, and are consistent with previous findings. For example, the PSI and CURB-65 scales could identify low-risk patients with COVID-19 pneumonia. 24 Moreover, the APACHE-II score of the survivors was significantly lower than that of non-survivors. 19 Therefore, the three scales could be reliably applied to identify low-risk and high-risk patients.
The AUCs of PSI, CURB-65, and APACHE-II were 0.83 (95% CI, 0.74–0.93), 0.80 (95% CI, 0.69–0.90), and 0.83 (95% CI, 0.75–0.92), respectively, indicating that the three scales are good predictive tools of the severity of COVID-19 pneumonia. The sensitivity of PSI was higher than CURB-65 and APACHE-II, indicating it could more correctively sort out patients who would develop into severe COVID-19 pneumonia, but the specificity was lower than CURB-65, which means that more non-high-risk patients would be falsely predicted to be severe.
The sensitivity of APACHE-II class ⩾II was only 37.9 (21.3, 57.6). However, the sensitivity of severity assessment for patients with COVID-19 pneumonia is extremely important, so APACHE-II should be used with caution. The result is different from findings reported by Cheng et al. 25 who found that the APACHE-II score was a strong predictor of COVID-19 pneumonia severity and mortality, They included a total of 53 cases. We think APACHE-II score is unsuitable for COVID-19 pneumonia for the following reasons. First, the score is complex, contains many variables and is not easy to manipulate. Second, it is no specific for respiratory system, respiratory failure is the hallmark for COVID-19 pneumonia patients, even absence of accompanying circulatory failure. 26 Third, the proportion of scores in elderly patients is high. Finally, COVID-19 patients are accompanied with various comorbidities, however, there is a lack of scoring for comorbidities in APACHE-II score. 27
Five of the six non-survivors included in this study had the highest levels in all three scales (PSI-V, CURB-65 score ⩾3, and APACHE-II-III). However, no patients with PSI class ⩽III (131/167, 78.4%), CURB-65 ⩽1 (145/167, 86.8%), and APACHE-II-I (153/167, 91.6%) died. Therefore, all three scales performed well in evaluating the mortality of mild and severe patients. A previous study showed a combination of PSI and CURB-65 scales could reliably access CAP. 28 Moreover, PSI and CURB-65 scales were almost equally sensitive and specific for predicting mortality among hospitalized patients with CAP with PSI class ⩾IV and CURB-65 score ⩾2. 29 Similarly our results showed that PSI is in good consistency with CURB-65.
Due to a very small sample of death in our study, the sensitivity and specificity of the three scales can’t be further compared to predict the mortality of patients with COVID-19 pneumonia. Notably, one non-survivor who was 85 years old, with a previous cerebrovascular disease and abnormal mental state during hospitalization, was rated as medium (PSI-IV, CURB-65 of 2, and APACHE-II-II) on all three scales. This example suggests we should pay more attention and vigilance to old COVID-19 patients with underlying chronic diseases.
There were some limitations in the present study. This is a single-center, retrospective study with small sample size. Our findings need to be validated in further multicenter larger studies. Due to retrospective nature of the present study, we did not have a predefined statistics protocol to calculate the sample size. The parameters of these scales were only a binary variable. They did not fully reflect the severity of COVID-19 pneumonia. Also, we should consider other factors for COVID-19, such as D-dimer, body mass index, IL-6 levels, which are not included in PSI, CURB-65, APACHE-II. In the study by Zhou et al., 30 D-dimer levels >1 μg/ml were shown to be associated with higher mortality (OR 18.42 (95% CI 2.64–128.55)). In a retrospective study by Nadkarni et al., 31 which included 4389 hospitalized patients with COVID-19, anticoagulantion resulted in lower mortality and intubation. Finally, the effects of treatments on mortality were not analyzed.
Conclusion
In conclusion, our study provided a preliminary assessment tool of the severity of COVID-19 pneumonia patients using PSI, CURB-65, APACHE-II scales. Our findings suggest that PSI and CURB-65 are useful to assess the severity and mortality of COVID-19 pneumonia; these scales might be used by clinicians to better manage their patients with COVID-19 pneumonia. More external validation studies are needed to verify our findings.
Supplemental Material
sj-docx-1-eji-10.1177_20587392211027083 – Supplemental material for Performance of CURB-65, PSI, and APACHE-II for predicting COVID-19 pneumonia severity and mortality
Supplemental material, sj-docx-1-eji-10.1177_20587392211027083 for Performance of CURB-65, PSI, and APACHE-II for predicting COVID-19 pneumonia severity and mortality by Junnian Chen, Bang Liu, Houwei Du, Hailong Lin, Cunrong Chen, Shanshan Rao, Ranjie Yu, Jingjing Wang, Zhiqiang Xue, Yixian Zhang and Yanghuang Xie in European Journal of Inflammation
Footnotes
Acknowledgements
We thank all the patients who consented to donate their data for analysis and the medical staff members who are on the front line of caring for patients. We thank Dr. Lianming Liao at the Center of Laboratory Medicine, Union Hospital and Dr. Jin Wei from School of Health Sciences, College of Health and Medicine, University of Tasmania, Australia, for their help in language improvement.
Author contributions
J.C, B.L, and H.D wrote original draft and collected the references. L.H, C.C, S.R, R.Y, J.W, Z.X, Y.Z, and Y.X provided clinical support. H.D and C.C helped to revise the manuscript. Dr. H Du was listed as a co-first author for his substantial contribution to manuscript critical revision, data analysis, interpretation, and drafting responses to reviewers. All co-authors agreed and approved Dr Du’s contribution and authorship. Dr. H Du approved the version to be published.
Availability of data and material
The data in the present study is not publicly accessible. Qualified researchers who are interested in obtaining the data can contact the corresponding author (Email:
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Fujian, China (grant numbers.2018J01309).
Ethical approval
Our study was conducted in line with the Declaration of Helsinki. The Fujian Medical University Union Hospital Ethics Committee approved the study protocol (2020XGFKY002), and written informed consent was obtained from the patients for their anonymized information to be published in this article.
Code availability Statistical
Analysis of data was performed on R software, version 3.6.3 (R Foundation for Statistical Computing, AT&T Bell Laboratories).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
