Abstract
An 8-mo-old, crossbred, heifer calf was presented to the Heeke Animal Disease Diagnostic Laboratory with a history of ataxia and altered mentation. Grossly, the liver was diffusely yellow-orange, turgid, and exuded watery, thin blood on cut section. The cortex and medulla in both kidneys were diffusely and markedly dark brown to black. The urinary bladder was filled with dark red urine. Histologically, centrilobular hepatocellular degeneration was observed, but these sections lacked necrosis. In the kidney, numerous cortical tubules contained intraluminal bright eosinophilic fluid and red-orange granular casts that stained positive for hemoglobin with the Dunn–Thompson method. The gross and histologic lesions supported a high level of suspicion for copper toxicosis. Feed and water samples from the farm were submitted for mineral analysis. The copper concentration in the feed was 118 mg/kg, and the molybdenum concentration was 0.9 mg/kg. Chronic copper toxicosis is rarely reported in cattle. The gross lesions in our case are a departure from, although similar to, previously reported cases, including lack of histologic hepatocellular necrosis. Collectively, gross and histologic lesions were compatible with copper toxicosis in this calf, and copper concentrations in the feed samples suggest a feed-mixing error.
Keywords
Ruminants require a balanced mineral diet for optimal growth. Deficiencies in essential trace minerals, including copper, zinc, iron, and selenium, can contribute to poor growth and an unthrifty animal. To avoid potential adverse effects from a poorly balanced diet, cattle feed is often supplemented with copper and other trace minerals. However, over-supplementation of copper, or an imbalance of molybdenum and other minerals that interact with copper and influence its uptake and kinetics, including iron, sulfur, and zinc, can lead to adverse effects and toxicity. 22 In ruminant species, sheep are overrepresented in cases of chronic copper toxicosis given that they are extremely sensitive to increases in copper level because of their reduced ability to increase biliary and urinary excretion of copper, coupled with the narrow margin of safety between safe levels of copper and toxic concentrations. 6 In contrast, cattle and goats are considered less susceptible to copper poisoning given their tolerance to higher concentrations of copper in feed. However, chronic copper poisoning resulting in hemolytic crisis is sporadically reported in cattle.1-4,13,17 We describe herein a case of chronic copper poisoning in a calf from southern Indiana.
An 8-mo-old, crossbred, heifer calf was presented to the Heeke Animal Disease Diagnostic Laboratory (Dubois, IN) with a week-long history of decreased feed intake, altered behavior, and increased aggression. Additional affected animals were not reported. The animal died naturally and was submitted for postmortem evaluation. The calf was thin, with markedly sunken eyes. The subcutaneous and adipose tissues were diffusely yellow (Fig. 1). The liver was mildly enlarged, turgid, and diffusely yellow-orange. Thin, watery blood exuded from cut surfaces, and the gall bladder was moderately distended with thick bile. The kidneys, bilaterally, were diffusely and markedly discolored dark brown to black (Fig. 2). The cortex and medulla in both kidneys were similarly discolored (Fig. 3). The urinary bladder was filled with dark red urine.
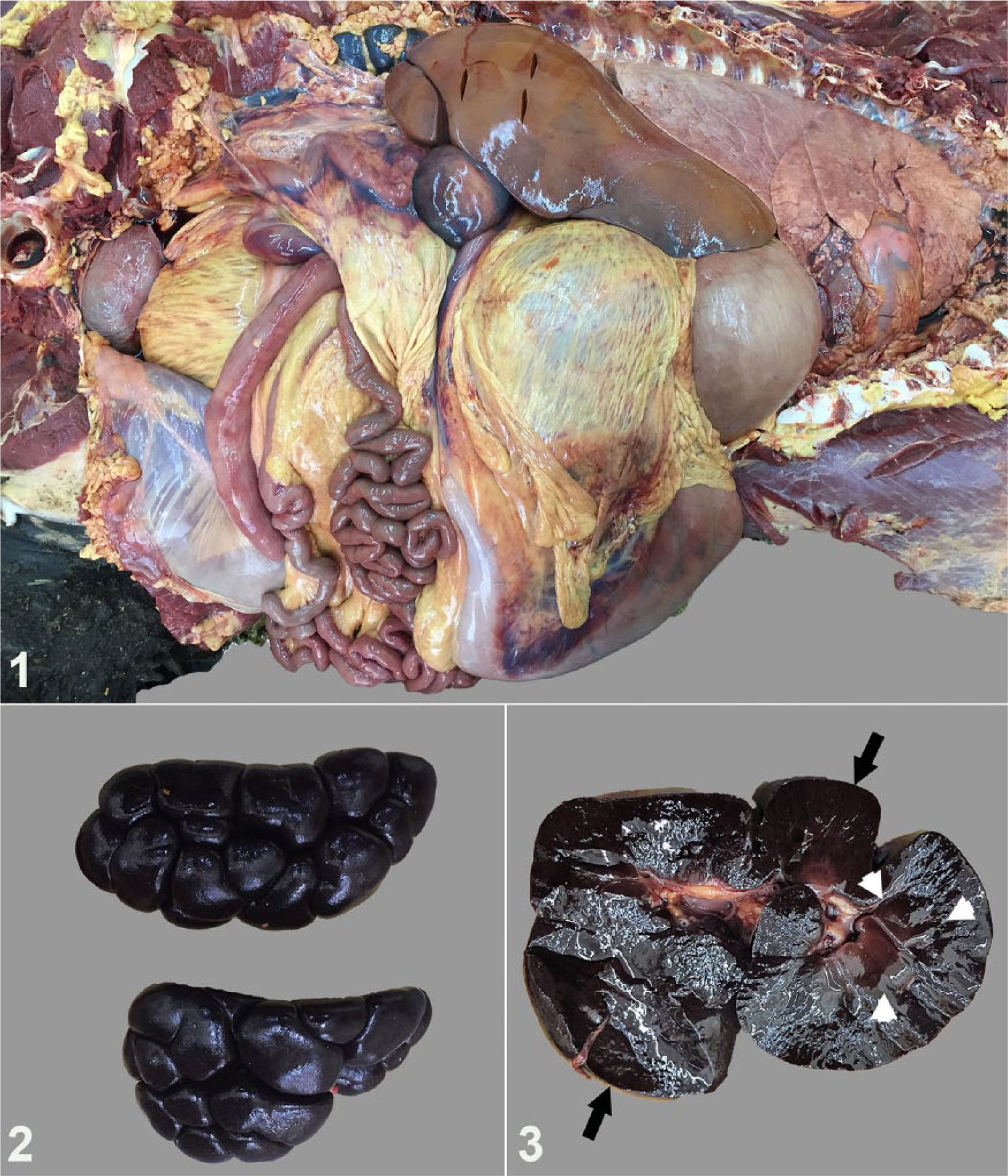
Postmortem images from an 8-mo-old crossbred calf with copper toxicosis.
Tissues examined histologically included representative sections of the heart, lungs, liver, and kidneys. Tissues were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin. Sections of the kidney were stained for hemoglobin using the Dunn–Thompson method. In the kidney, numerous cortical and medullary tubular lumina were filled with bright eosinophilic fluid and red-orange granular material, and lined by attenuated tubular epithelium (Fig. 4). Scattered tubules were lined with necrotic epithelial cells and infiltrated with neutrophils and macrophages. Other tubular epithelial cells were degenerate, with clear intracytoplasmic vacuoles. The intraluminal material stained positive for hemoglobin using the Dunn–Thompson method (Fig 5). In sections of the liver, we observed centrilobular hepatocellular degeneration consistent with microvesicular lipidosis or hydropic change and characterized by swollen hepatocytes with variably sized intracytoplasmic round, clear vacuoles (Figs. 6, 7). A few centrilobular hepatocytes were lost, with replacement by neutrophils and macrophages. Multifocally, bile canaliculi were occluded by bile plugs.
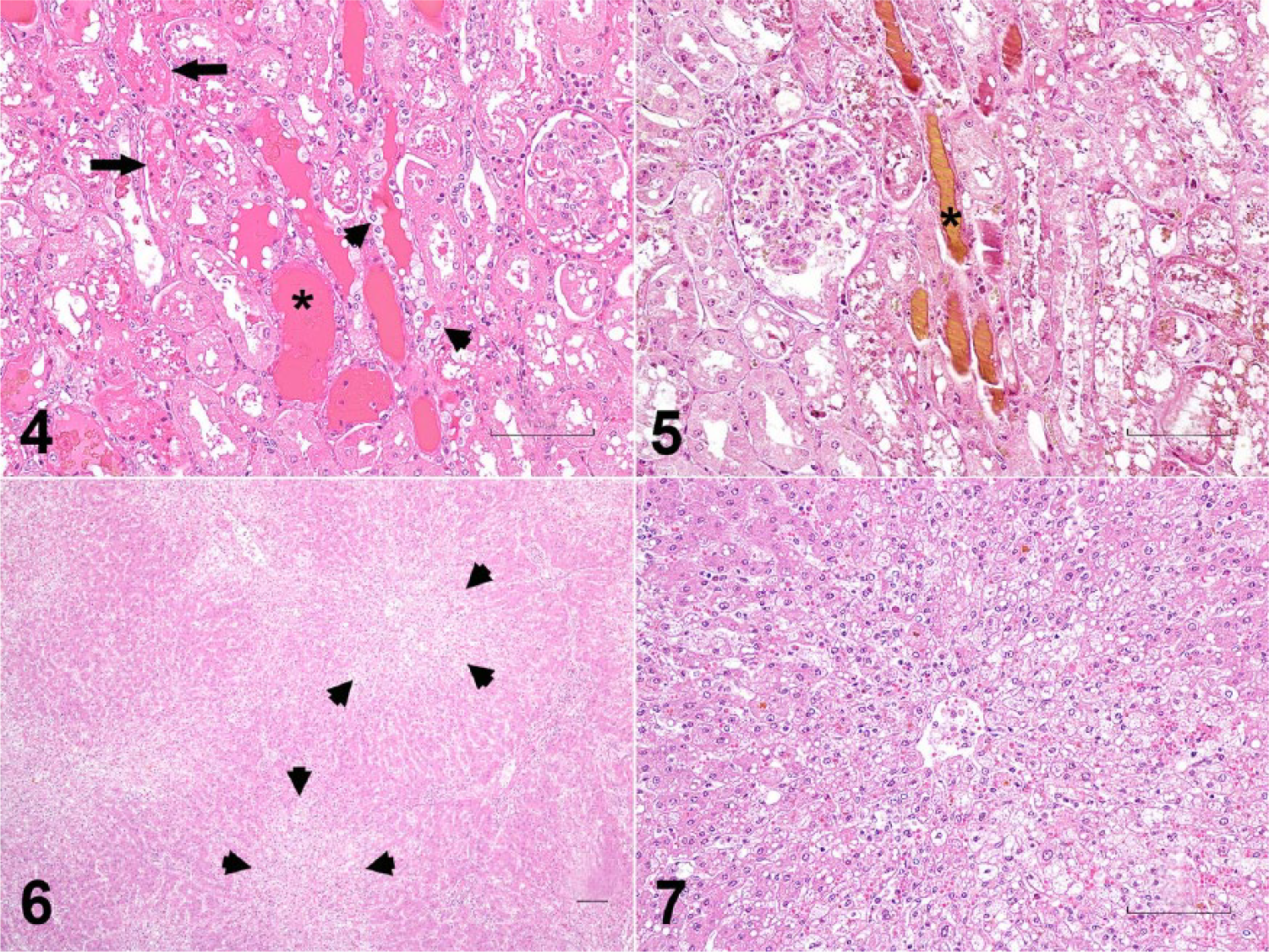
Photomicrographs of liver and kidney from a calf with copper toxicosis. Bars = 100 μm.
Based on the postmortem findings suggesting a hemolytic crisis, possible causes of death included copper toxicosis, leptospirosis, bacillary hemoglobinuria, nitrate/nitrite poisoning, and babesiosis. Representative tissue samples were submitted for PCR testing for
Mineral analyses of tissue samples and feed in a case of copper toxicosis in a calf.
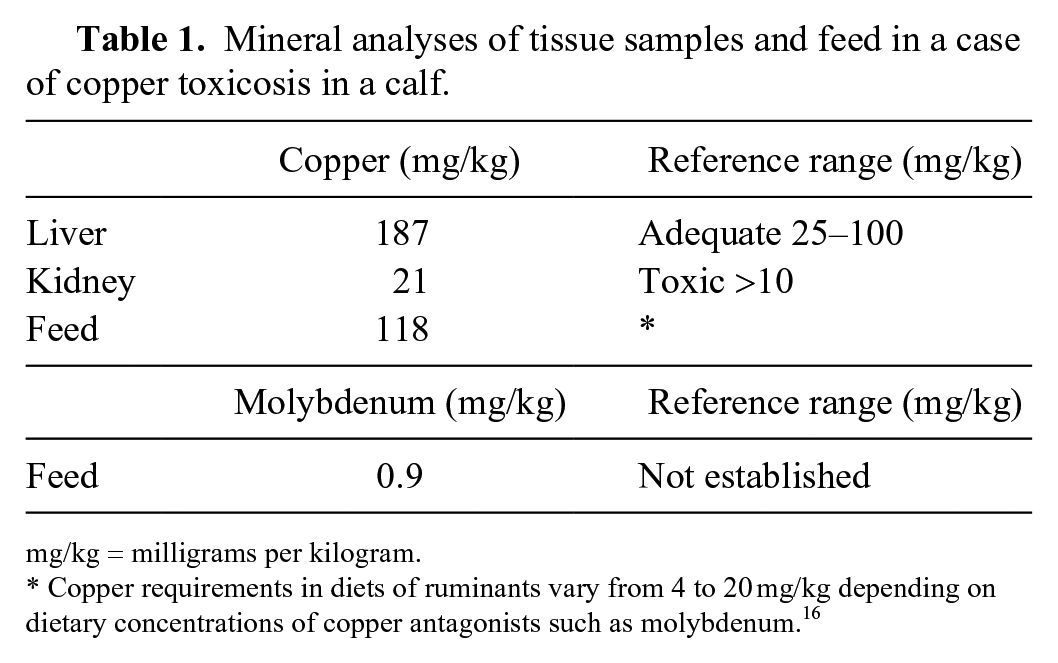
mg/kg = milligrams per kilogram.
Copper requirements in diets of ruminants vary from 4 to 20 mg/kg depending on dietary concentrations of copper antagonists such as molybdenum. 16
Two forms of copper toxicosis, acute and chronic, are recognized and reported in ruminants.6,22 The acute form of copper toxicosis is often secondary to iatrogenic administration of copper. Normally, free copper ions from the diet are absorbed by the small intestine, bound to transcuprein and albumin in the blood, and delivered to the liver. 22 The mechanism of acute toxicity involves saturation of copper-binding proteins, such as metallothionein, glutathione, and ceruloplasmin, leading to free copper ions in the cytoplasm and bloodstream.6,15,22 Unbound copper is a strong oxidant and can form reactive oxygen species leading to membrane damage by lipid peroxidation and damage to nucleic acids and cellular proteins, ultimately causing cell death. 22 Two previously reported cases of acute copper toxicosis in calves were associated with supplementation of large boluses of injectable forms of copper.8,9 Histologic lesions of acute copper poisoning were variable and ranged from hepatocellular degeneration to necrosis in reported cases.6,8,9
Numerous reports of chronic copper toxicosis are described in sheep but infrequently reported in other ruminants.6,11,21 The mechanism of chronic copper toxicosis involves long-term ingestion of dietary copper above the recommended daily amount but below the level that would cause immediate toxicosis. This chronic accumulation leads to saturation of copper-binding proteins and the slow buildup of copper within lysosomes of hepatocytes. Once copper stores in the liver are overloaded, and with a preceding stressful event, such as transport, calving, or ingestion of hepatotoxic plants, copper is released from necrotic or degenerate hepatocytes into the bloodstream. Unbound copper acts as an oxidizing agent resulting in oxidative damage to the cellular membrane of erythrocytes, leading to hemolytic crisis.1,6,22 In sheep, an intrinsic inability to increase biliary excretion of copper in response to dietary increases predisposes this species to copper poisoning even with mild increases in dietary copper.6,22 Cattle and goats are reportedly more resistant to chronic copper toxicosis, possibly because of their capacity to increase biliary excretion of copper. 6 Deficiencies in upregulating biliary excretion are not identified in current literature in cattle. Although cattle and goats are described as resistant to copper poisoning, cases of chronic toxicosis in cattle and goats have been reported.2,3,5,13,18 In most reports, the underlying cause of chronic copper poisoning in cattle is often associated with diet/feed supplementation errors, but other factors, such as breed, have been suggested to predispose cattle to copper poisoning.2,4,13,17,18,22
In reported cases of chronic copper toxicity in cattle, over-supplementation or a formulation error contributed to toxicosis in dairy herds4,13,17 and in 2 separate groups of beef calves.18,20 Unlike acute copper poisoning, gross lesions of “gun-metal blue” kidneys secondary to hemolytic crisis, icterus, and histologic lesions of hepatocellular degeneration and necrosis were observed in cases with chronic copper toxicosis. The gross and histologic kidney lesions observed in our case are consistent with other reports of chronic copper toxicosis in cattle. In our case, the feed, liver, and kidney copper concentrations were 118 mg/kg, 187 mg/kg, and 21 mg/kg, respectively (Table 1). The copper concentrations in the liver and kidney were above reference ranges used in our laboratory. In comparison to most reports of copper toxicosis, the liver copper concentration in our case was lower compared to copper levels in previously described affected animals.13,18,20 The highest liver copper concentration from previously reported copper-intoxicated calves was 935 mg/kg, which is roughly 5-fold greater than the liver copper value in our case. The kidney copper concentrations from previous reports ranged from 1.1 to 964 mg/kg.4,18,20 Unlike other reports, histologic lesions of hepatocellular necrosis were not observed in our case, and the discrepancy is possibly the result of variable progression of the disease and the regenerative capacity of the liver. Sporadic centrilobular hepatocytes were lost in several sections of liver with a few infiltrates of neutrophils, suggesting low levels of hepatocellular necrosis, but we did not observe overt hepatocellular necrosis. Hemolysis-inducing copper release likely occurred with sublethal hepatocellular injury, which may explain the lower liver copper concentrations compared to previous reports of copper poisoning in cattle.
The balance between copper deficiency and toxicosis is complicated by other trace minerals that affect copper bioavailability. Molybdenum, sulfur, iron, and zinc at various ratios to copper can affect the intestinal absorption of copper.15,22 Sheep are known to be highly sensitive to copper toxicity and/or molybdenum deficiency. The copper-to-molybdenum feed ratio recommended for sheep is 6:1 and any values approaching and exceeding 20:1 are considered a risk for developing copper toxicosis in sheep. 22 Although a specific copper-to-molybdenum ratio for cattle is not firmly established, the National Research Council has established the maximum tolerable level of copper in the feed of cattle as 40 mg/kg, assuming normal concentrations of molybdenum (1–2 mg/kg diet) and noting that, at molybdenum concentrations in the feed below this, copper may become toxic at lower levels. 16 In this case, the concentrations of copper and molybdenum in the feed were 118.4 mg/kg and 0.9 mg/kg, respectively, resulting in a copper-to-molybdenum ratio of approximately 131:1. Comparison of the feed copper-to-molybdenum ratios with other chronic copper poisoning cases was difficult given the absence of feed copper and/or molybdenum concentration measurements in other reports. Although feed formulation analysis was not pursued by the owner, a feed-mixing error seems to be the most likely cause of copper poisoning in this case. Interestingly, other animals in the herd that were presumably on the same diet were not reportedly affected, raising consideration of additional factors or infectious comorbidities that predisposed this calf in the herd to chronic copper toxicosis. An infectious process was not observed on gross and histologic evaluations of the representative tissue sections. A limitation in our case is the lack of hematology and clinical chemistry data on this calf to further characterize the hematologic parameters and differentiate the renal tubular cast as hemoglobin from myoglobin. The histochemical stain used to detect hemoglobin can presumably stain myoglobin, so skeletal muscle damage as a cause of renal cast formation cannot be excluded. Despite these limitations, copper levels in the submitted liver, kidney, and feed indicate excessive exposure and support copper poisoning. Other factors may have led to toxicosis in this calf and no other herdmates; however, the logistical and fiscal constraints of a diagnostic postmortem examination will leave some of these relatively minor questions unanswered.
Several suggested risk factors for chronic copper toxicosis have been proposed in cattle.1,2,7,12 As reported in the Ronaldsay sheep breed, the possibility of cattle breed susceptibility to copper toxicosis, particularly in Jersey cattle, was suspected in multiple studies.1,2,6,7,10,12 In one study investigating chronic copper poisoning in a dairy herd comprised of Jersey and Holstein cows, 6 of the 8 cows died within the first 15 d of the dry period, suggesting that the initial 2-wk dry period may be a predisposing factor to copper toxicosis. 2 Other factors contributing to chronic copper toxicosis during the drying period in the reported case included a copper-enriched diet during lactation and an abrupt withdrawal of concentrates prior to the dry period. 2 Additional studies on copper poisoning in cattle can aid in refining recommended dietary copper levels, especially because copper loading is considered an emerging concern in parts of the world.1,14,19 Our case of chronic copper toxicosis in a calf had lesions consistent with other reports in cattle and sheep, but a departure from the other reported cases included a lack of hepatocellular necrosis, which presents a knowledge gap in the mechanism of copper toxicosis in cattle.
Footnotes
Acknowledgements
We thank the technical staff of the Histology and Toxicology departments of the Animal Disease Diagnostic Laboratory. This work was presented at the Am Assoc Vet Lab Diagn Annual Meeting, October 18–22, 2018, Kansas City, MO.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
