Abstract
Between January and October 2006, 15 Chios sheep died in a field located near a factory in Orhangazi, Bursa, Turkey. In addition, in May 2007, 2 ewes died after aborting in the same field. Clinical signs in affected animals prior to death were anorexia, hematuria, icterus, incoordination, and ptyalism. Postmortem findings included generalized icterus; yellow, friable livers; distended gallbladders with dense, dark bile; and dark, hypertrophic kidneys with hemorrhage. Copper (Cu) concentrations were measured in multiple specimens of the following: 9 sera, 3 livers, 3 kidneys, 4 plants (including 2 artichoke leaf specimens), 3 soil samples, and 1 drinking water sample. High Cu concentrations were present in the livers, kidneys, and sera of dead sheep, as well as in the vegetation and soil samples from the field. Chronic Cu toxicosis was confirmed as the cause of death attributed primarily to the use of copper sulfate as a fungicide for fruit trees within the field. In addition, factory dust containing Cu might have been an additional factor in the toxicosis.
Copper (Cu) is an essential trace element for many biological processes of plants and animals. It is required for normal iron metabolism, synthesis of elastin and collagen, melanin production, and integrity of the central nervous system. It also is essential in keratin (wool) production. Copper is one of the key trace minerals required for an effective immune response. When Cu is ingested in large amounts in the diet, it may accumulate within the liver over a period of a few weeks to more than a year without clinical signs followed by a sudden release of liver Cu stores with resultant toxicity. Many factors that alter Cu metabolism can influence chronic Cu toxicosis by enhancing the absorption or retention of Cu. Chronic Cu toxicosis may result from excessive intake of Cu; low intake of molybdenum, sulphur, zinc, or calcium; or liver damage. 8 In Cu toxicosis, blood Cu concentrations may increase suddenly, causing lipid peroxidation and intravascular hemolysis. A hemolytic crisis may be promoted by many factors including transportation, pregnancy, lactation, strenuous exercise, or a deteriorating plane of nutrition. Sheep are more susceptible to the effects of Cu toxicity than other species of farm animals because their Cu excretory mechanism is less efficient. Acute Cu toxicosis is usually seen after accidental administration of excessive amounts of soluble Cu salts, which may be present in anthelmintic drenches, mineral mixes, or improperly formulated rations. 15
Acute toxicosis may follow ingestion of 20 to 100 mg of Cu/kg body weight in sheep. Chronic toxicosis of sheep may occur with daily intake of 3.5 mg of Cu/kg body weight when grazing pastures that contain 15–20 mg of Cu/kg on a dry weight (DW) basis with concurrent low concentrations of molybdenum and sulfur. 10,15 Acute Cu toxicosis causes severe gastroenteritis with abomasal erosions and ulcerations, abdominal pain, diarrhea, anorexia, dehydration, and shock. Hemolysis and hemoglobinuria may develop after 3 days if the animal survives these gastrointestinal disturbances. Icterus usually develops in animals that survive more than 24 hr. The sudden onset of clinical signs in chronic Cu toxicosis is associated with the development of a hemolytic crisis. Affected animals exhibit depression, weakness, rumen stasis, anorexia, hematuria, hemoglobinuria, icterus, incoordination, and ptyalism. 3,15 Methemoglobinemia, hemoglobinuria, anemia, and decreased blood glutathione concentrations are usually observed during hemolytic crisis. Animals with these clinical signs and laboratory abnormalities often die within 1–2 days. Although herd morbidity is often <5%, usually >75% of affected animals die. Furthermore, losses from Cu toxicosis may continue for up to 2 months after the dietary problem has been rectified. 15
Necropsy findings in chronic Cu toxicosis include tissues that are discolored by icterus and methemoglobin. Swollen gunmetal-colored kidneys, port wine-colored urine, and an enlarged spleen with a dark brown-black parenchyma are manifestations of the hemolytic crisis. The liver is usually enlarged and friable. Histologically, hepatic centrilobular and renal tubular necrosis are observed. 1,10,15
A farmer reported the deaths of multiple sheep to the Bursa Office of the Agriculture Ministry in 2006. Between January and October, 9 ewes, 2 pregnant ewes, 2 lambs, and 2 rams from a herd of 50 sheep died in the barn or adjacent field (Table 1). In addition, 2 other ewes aborted and died in mid April 2007. The farmer claimed that deaths had increased since a nearby factory began operation. Toxicosis was suspected because this factory in Orhangazi, Bursa, Turkey, had an insufficient aeration basin collector with previous evidence of Cu and Fe contamination. The suspected toxicosis was investigated by the Bursa Office of the Agriculture Ministry and the Faculty of Veterinary Medicine of Uludag University.
Deaths of sheep in a flock of 50 animals on a farm in Orhangazi, Bursa, Turkey, during 2006.

Died after abortion.
The farmer and local veterinarian had noticed that all ill sheep had depression, anorexia, hematuria, icterus, incoordination, and ptyalism prior to death. In the dead sheep, the blood appeared darker than expected. Three dead sheep (2 females and 1 male) were necropsied, and observed changes included enlarged, yellow, and friable livers; distended gallbladders containing dense, dark bile; dark, enlarged kidneys; and icteric adipose tissues. Portions of the liver and kidneys from each animal were collected for toxicological analysis, and portions of liver were obtained for histopathological examination. Histologic lesions in the liver included hepatocellular cytoplasmic vacuolation and necrosis with infiltrates of macrophages in sinusoids and around some blood vessels.
Researchers visited the farm 3 times, during which random blood samples were collected from 9 live sheep, including a ewe in hemolytic crisis. The distance from the factory to the fruit trees and vegetation sample areas varied from 30 m to 300 m, while the distance from the factory to the sheep barn was approximately 20–30 m. Environmental samples of vegetation, soil, and water were collected for toxicologic analysis. Four vegetation samples were collected, including 2 samples of dried vegetation under the fruit trees and 2 specimens of artichoke leaf located 25 m and 250 m from the factory but near the barn. Three soil samples were collected, including 1 mixed-soil sample from various areas of the field and 2 soil samples from areas located 25 m and 250 m from the factory but close to the barn. A drinking water sample was obtained from a depth of 10 m. Serum samples were analyzed spectrophotometrically using a commercial clinical chemistry analyzer a according to the manufacturer's instructions. The other samples were analyzed for Cu by flame atomic absorption spectrometer b following the manufacturer's instructions. A calibration curve was constructed using appropriate standard solutions c of Cu; the detection limit of Cu was 0.05 mg/l.
The Cu concentrations in the biological and environmental samples, in relation to those reported in the veterinary literature, are presented in Table 2. In the present study, the Cu concentrations of serum (8–108 μmol/l), liver (103–614 mg/kg, wet weight [WW]), kidney (26–61 mg/kg, WW), and vegetation (114–170 mg/kg, dry basis [DB]) were consistent with chronic Cu toxicosis as reported in the veterinary literature. 1,3,6,11,12,14,16,17 The Cu concentrations of the vegetation and diet in the present report were also higher than previously reported physiological Cu concentrations. 4,9,15 Although the Cu-contaminated vegetation (114 and 170 mg Cu/kg, DB; Table 2) was withheld from the sheep starting in October 2006, deaths did not cease until mid April 2007.
Sheep are very susceptible to Cu overdose, and chronic Cu toxicosis occurs when animals are fed diets over weeks or months that are marginally high in copper content (15–20 mg/kg, DW) with low concentrations of molybdenum. 10,17 Reported clinical signs of chronic Cu toxicosis in sheep are depression, weakness, rumen stasis, anorexia, hematuria, hemoglobinuria, icterus, incoordination, and ptyalism. 3,10,15 Clinical signs in the sheep of the present report were similar. Furthermore, the necropsy lesions in the liver, gallbladder, kidneys, and adipose tissue and microscopic findings in the liver of affected sheep also could be attributed to Cu toxicosis. 7,10,15
The source of Cu toxicosis in the present report could be attributed to copper sulfate application on the fruit trees as a fungicide and/or the factory dust containing Cu. Copper sulfate had been used twice in 2006 as a fungicide on the apple, peach, and plum trees in the field, and the vegetation under the trees had been harvested and used as a food source for the sheep. Copper concentrations in the artichoke leaf samples collected 25 m and 250 m from the factory were 13 mg/kg (WW) and 81 mg/kg (WW), respectively. For the soil samples, the copper concentrations were 165 mg/kg (DW) and 480 mg/kg (DW) for samples collected 25 m and 250 m from the factory, respectively. These findings suggest that the use of copper sulfate as a fungicide on the fruit trees was primarily responsible for the chronic Cu toxicosis in these sheep. The factory dust, which had previously been shown to contain copper and could not be collected in the factory's aeration basin, might have been a contributing factor in the development of chronic Cu toxicosis. Cu-containing dust could have been blown onto the field by wind, where it subsequently was ingested and/or inhaled by the sheep. Previous studies have shown that breathing air contaminated with Cu may cause metal accumulation in animals such as sheep. 5,13 For example, chronic Cu toxicosis was reported in an impala and buffalo in the Palaborwa area of Kruger National Park, 5 where the source of Cu was determined to be a Cu mine. In the current report, the sheep might have inhaled Cu dust in the vicinity of the barn (approximately 20–30 m from the factory). Two pregnant ewes that aborted and died in the same barn in April 2007 could have accumulated Cu in their livers by ingesting Cu-contaminated vegetation over a 5-month period. In addition, stressors such as pregnancy could have attributed to the hemolytic crisis in these 2 individuals.
Copper (Cu) concentrations in ovine serum, liver, and kidney samples; dietary vegetation and concentrate additives; drinking water; and environmental plant and soil samples in cases of chronic Cu toxicosis reported in the veterinary literature and in the current study.*
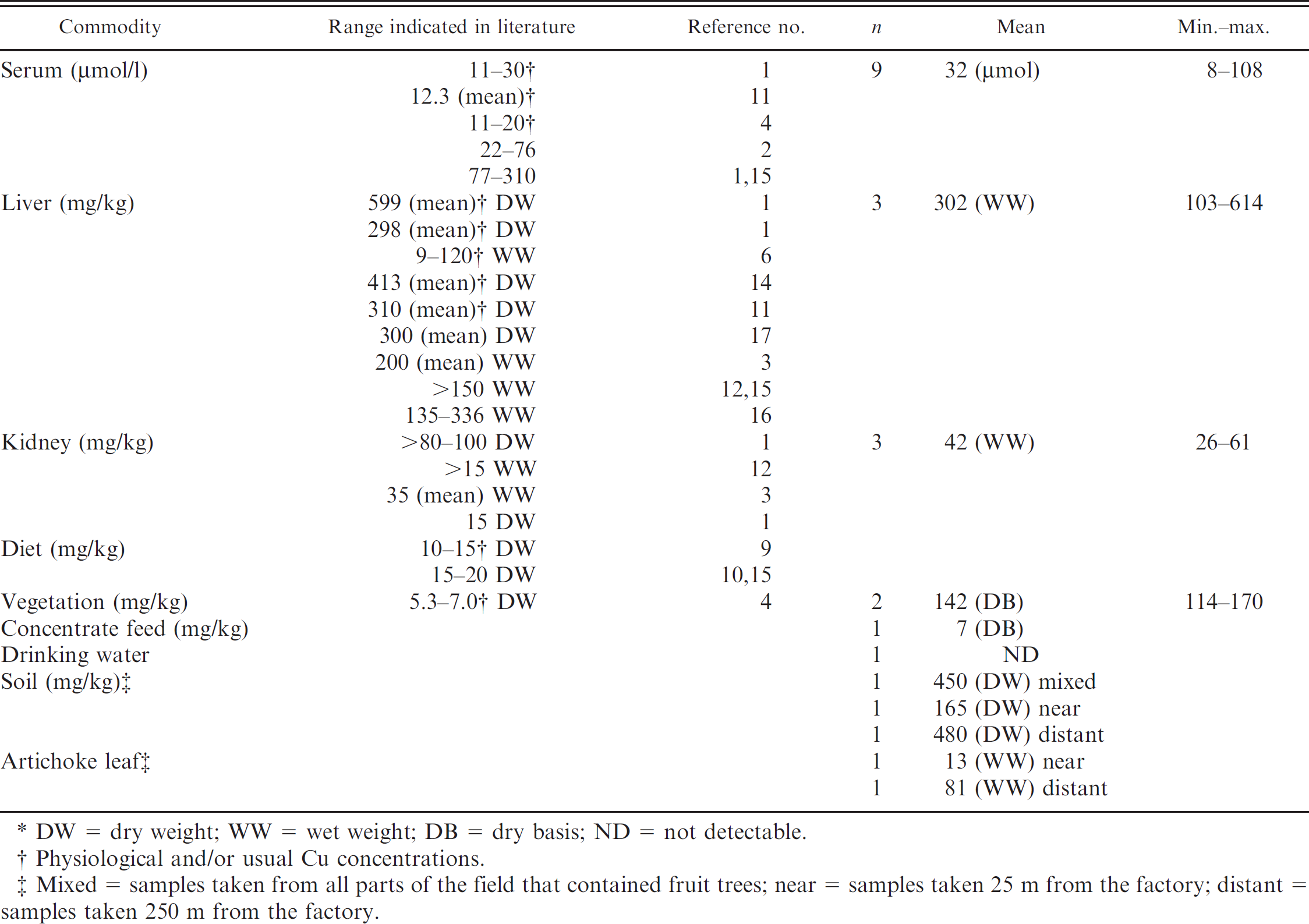
DW = dry weight; WW = wet weight; DB = dry basis; ND = not detectable.
Physiological and/or usual Cu concentrations.
Mixed = samples taken from all parts of the field that contained fruit trees; near = samples taken 25 m from the factory; distant = samples taken 250 m from the factory.
In summary, the diagnosis of chronic Cu toxicosis was based on toxicological analysis of biological, plant, and soil samples as well as the supportive medical history, clinical signs, necropsy lesions, and microscopic findings in sections of liver. The most likely source of Cu contamination was attributed to the use of copper sulfate as a fungicide for nearby fruit trees, but Cu-containing factory dust may have been a contributing factor.
Acknowledgements. The authors would like to thank the Management of the Veterinary Faculty of Uludag University for funding this research, and Dr. Figen Kahraman, from the Bursa Office of the Agriculture Ministry, for the necropsy and histopathologic examinations of these animals.
Footnotes
a.
Abbot Aeroset®, Toshiba Corporation for Abbot Laboratories, Tokyo, Japan.
b.
Analyst 800, Perkin Elmer Inc., Waltham, MS.
c.
Custom Grade Standards, Inorganic Ventures Inc., Lakewood, NJ.
