Abstract
We report a case of canine adenocarcinoma with multi-organ metastasis in which colonies of adenocarcinoma cells grew upon aerobic bacterial culture of pleural effusion. Stained agar colonies were highly similar to rare suspicious cells seen on cytologic examination of the pleural effusion, as well as rare cells seen on cytologic examination of pancreatic and gastric wall fine-needle aspirates. Cells from colonies growing on agar media were mildly immunoreactive for cytokeratin. Histologic examination of tissues obtained at autopsy revealed pancreatic adenocarcinoma with vascular invasion and nodal, gastric, pulmonary, and pleural metastasis.
A 9-y-old, spayed female Pug–Beagle cross dog was admitted to the University of Wisconsin Veterinary Care (UWVC; Madison, WI) with a 6-d history of anorexia and 1-d history of icterus that failed to improve with fluid therapy, antibiotics, and anti-nausea medication administered by her primary veterinarian. Physical examination at UWVC was unremarkable apart from mild tachypnea at 40 breaths per min, mildly reduced bronchovesicular sounds on the right lung fields, and icteric sclera and mucous membranes. Complete blood count and biochemical panel had been performed at her primary veterinarian that morning prior to presentation at UWVC. Notable abnormalities included leukocytosis (30.4 × 109/L; reference interval [RI]: 5.1–16.8); the automated differential indicated neutrophilia (25.5 × 109/L; RI: 3.0–11.6) and monocytosis (2.6 × 109/L; RI: 0.2–1.1). Biochemical abnormalities included increased total bilirubin (111 µmol/L; RI: 0–15.4), alanine aminotransferase (897 U/L; RI: 10–125), and gamma-glutamyl transferase (62 U/L; RI: 0–11). The analyzer was unable to provide a reading for alkaline phosphatase.
Point-of-care ultrasound examination detected pleural effusion, and thoracentesis was performed. Seventy mL of pleural effusion was removed and submitted for cytologic evaluation. Slides were stained with Wright–Giemsa stain. The fluid was yellow and cloudy. Total protein was 41 g/L. Nucleated cell count was 20.9 × 109/L, with 73% nondegenerate neutrophils, 23% macrophages, 2% lymphocytes, 1% eosinophils, and 1% mesothelial cells. Although free erythrocytes were minimal, macrophages were commonly erythrophagocytic, as were rare neutrophils. Given that the sample was processed promptly after collection and there was considerable erythrophagia absent hemosiderin, findings were suggestive of acute hemorrhage into the pleural cavity. There were occasional clusters of reactive mesothelial cells, but also a few tightly cohesive clusters of cells with anisokaryosis beyond that commonly seen in reactive mesothelial cells (Fig. 1). These cells had variable nuclear-to-cytoplasmic (N:C) ratios, and round-to-oval nuclei with stippled chromatin and usually a single, occasionally large, nucleolus. The cytoplasm contained moderate-to-many fine vacuoles. Binucleate and multinucleate cells were occasional. Mitotic figures were rare. These cells were of uncertain origin; reactive or neoplastic mesothelium and carcinoma were considered.
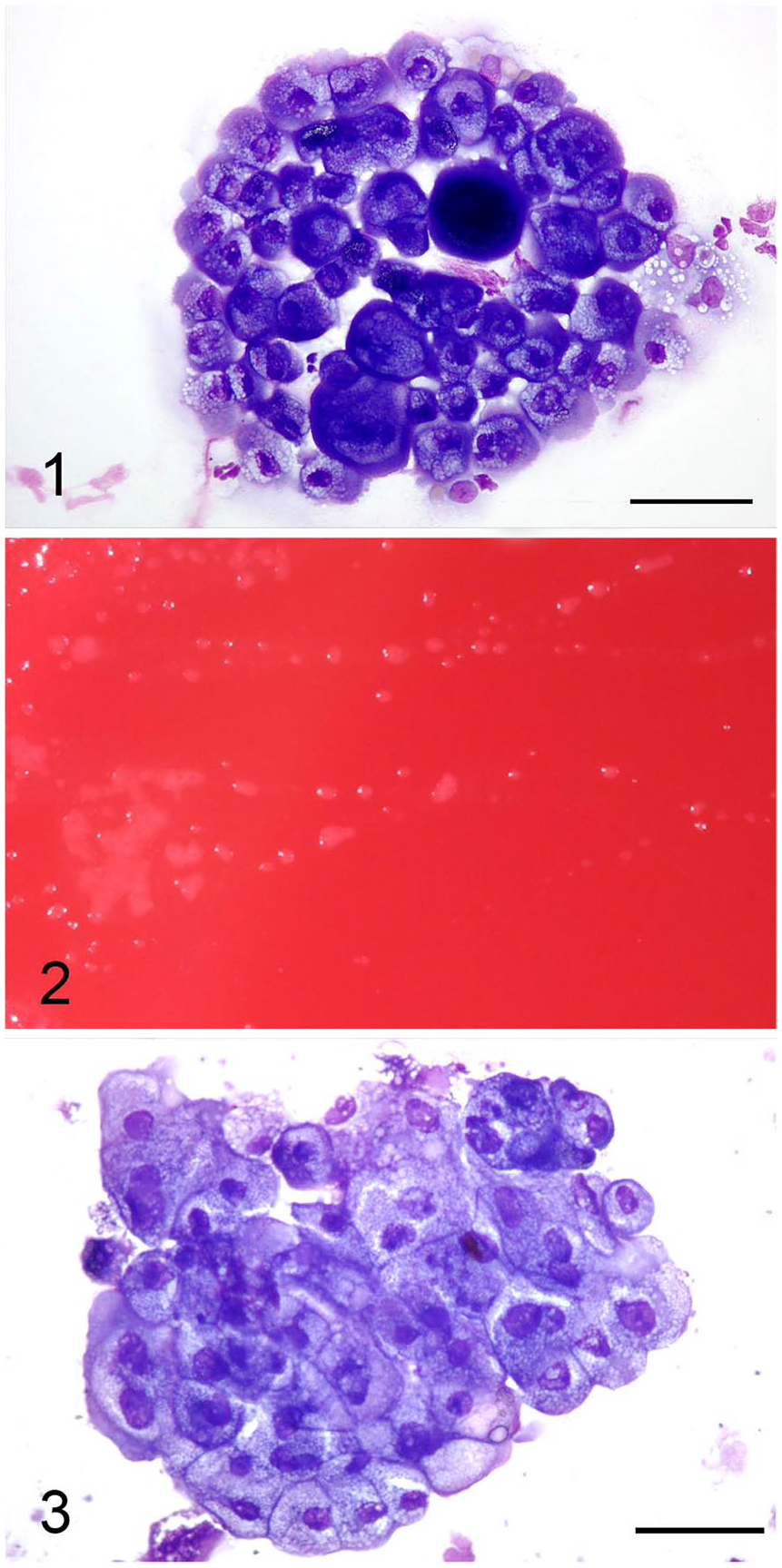
Canine pleural effusion cells, with growth on bacterial culture media.
Pleural fluid was submitted to the microbiology laboratory for aerobic culture within 30 min of collection. One drop of fluid each was inoculated onto trypticase soy agar supplemented with 5% sheep blood (Remel, Lenexa, KS), chocolate agar (Remel), Levine eosin–methylene blue agar (Remel), and colistin–nalidixic acid (CNA) agar (Remel), and 1 mL of fluid was inoculated into 10 mL of thioglycollate broth supplemented with vitamin K and hemin (Remel). Culture plates and broth were incubated at 36°C in a 5% CO2 atmosphere and examined daily for growth.
Abdominal ultrasound findings included a diffusely hypo-echoic and heterogeneous right pancreatic limb with lobulated margins and adjacent hyperechoic mesentery. The pancreaticoduodenal lymph node was enlarged and hypoechoic. There was gastric wall thickening and loss of layering as well as muscularis thickening of the proximal duodenum. Pancreatitis with secondary regional steatitis, reactive lymphadenopathy, focal duodenitis, and severe gastritis were suspected. The following day, computed tomography of the thorax also revealed a pulmonary nodule in the left cranial lung lobe. Fine-needle aspirates of pancreas revealed neutrophilic inflammation, a small amount of reactive pancreas, and rare small clusters of highly vacuolated epithelial cells similar to those seen in the pleural effusion cytology. Gastric wall fine-needle aspirates were poorly cellular with similar rare clusters of highly vacuolated epithelium. Collective findings were highly suspicious of metastatic adenocarcinoma. Clinical signs failed to improve with supportive care (intravenous fluids, antibiotics, and anti-nausea medications, and nasogastric tube for nutritional support). The owner elected to take the patient home, and the dog was euthanized by her primary veterinarian 2 d after presentation to UWVC.
After 3 d of incubation, small, opaque colonies were detected on all agar plates including the blood agar plate (Fig. 2). Turbidity with microcolonies was also detected in the thioglycollate broth. Gram stain of colony growth and fluid from the thioglycollate broth revealed no stainable microorganisms, with the presence of cells and cellular debris. Modified acid-fast stain and a wet prep were performed using colony growth from the blood agar plate to look for microorganisms that may not have taken up Gram stain. No microorganisms were observed by modified acid-fast stain. The wet prep revealed individual and cohesive clusters of somatic cells and no microorganisms. Based on the wet prep appearance of the cells, we suspected that colonies represented cellular growth. Therefore, agar colonies and fluid from the thioglycollate broth were stained with Wright–Giemsa stain. Examination of the stained slides showed cohesive clusters of somatic cells (Fig. 3). The cells were round-to-polygonal with moderate-to-low N:C ratios and deeply basophilic cytoplasm containing moderate-to-many fine vacuoles. Nuclei were round-to-oval with finely stippled chromatin and a distinct nucleolus. These cells were similar to those seen in the cytologic examination of the pleural fluid as well as pancreatic and gastric wall fine-needle aspirates. No microorganisms were present.
The limited autopsy performed by the primary veterinarian revealed a thickened, firm, mass-like area of the pancreas near the proximal duodenum, with adherent omentum. Samples of pancreas and adjacent lymph nodes, stomach, a firm area of lung, and an area of pleura with adherent white material were preserved in 10% neutral-buffered formalin. Histologic examination of these tissues at UWVC revealed exocrine pancreatic adenocarcinoma with vascular invasion and nodal, gastric, pulmonary, and pleural metastasis. Moderate necrosuppurative pancreatitis with fibrosis was also present. The neoplastic cells in histologic sections were strongly immunoreactive for cytokeratin. Smears of colonies from the blood agar plate were weakly immunoreactive for cytokeratin compared to the negative control. Although positivity was weak, the slides were air-dried and stored at room temperature for > 2 mo, which may have impacted staining negatively. 5
Definitive cytologic diagnosis of carcinoma in effusions relies on the presence of highly pleomorphic clusters of epithelial cells, but diagnosis of carcinoma can be complicated by the fact that reactive mesothelial cells in pleural effusions may also be highly pleomorphic.2,6 The rare clustered cells in the pleural fluid of this patient did exhibit moderate pleomorphism; however, the pleural fluid consisted predominantly of inflammatory cells. Inflammation has been documented to increase proliferation and reactivity of mesothelial cells,2,3 which led to uncertainty of the primary disease process underlying the effusion.
Although neoplastic cells can be cultured in soft agar, the cells traditionally must be embedded within the agar, as opposed to forming discrete colonies on top of the agar, as did the cells in our case did. 1 Growth of neoplastic cells on bacterial culture media is exceedingly rare in a clinical setting. We are aware of a single previous report of pleural effusion in an 82-y-old woman that grew colonies of adenocarcinoma cells on bacterial culture media. 4 The primary origin of that neoplasm was not determined. Although the neoplastic cells from the human case were able to be subcultured to blood agar, subcultures of our colonies taken from thioglycollate broth and chocolate and blood agar plates to chocolate and blood agar as well as thioglycollate broth did not yield growth, suggesting different growth requirements for these 2 adenocarcinomas. We speculate that additional growth factors or nutrients present in the pleural fluid were probably needed for cellular growth in our case. Colony appearance in our case did not resemble Mycoplasma spp. as was observed in the human report, given that our colonies were raised and did not have the typical “fried egg” appearance. Our colonies were originally thought to be a slow-growing fastidious microorganism or rapid growing Mycobacterium spp. until stains revealed the presence of no microorganisms.
To our knowledge, neoplastic cells growing on bacterial culture media from a veterinary patient in a clinical setting have not been reported previously. Although there is no known risk to laboratory personnel from the growth of neoplastic cells, microbiology personnel should be aware that neoplastic somatic cells may rarely grow on bacterial culture media and that traditional cytologic stains will highlight cellular features better than common microbiologic stains. Good communication between microbiology and cytology personnel can also help facilitate identification of somatic cells in these rare cases.
Footnotes
Acknowledgements
We thank Dr. Ken Fiedler for performing the autopsy, Dr. Marie Pinkerton for performing histologic examination of the autopsy tissues, and Dr. John Wojcieszyn for performing the cytokeratin immunostaining on smears of the agar colonies. This work was presented in an oral format at the ACVP & ASVCP Concurrent Annual Meeting in Vancouver, Canada in 2017.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
