Abstract
Cryptococcosis, caused by the
Introduction
Cryptococcosis, an uncommon but important fungal disease of a wide variety of human and animal hosts, is caused by yeasts in the
Ferrets (
A domestic ferret residing in western Sydney, Australia, was diagnosed with sinonasal cryptococcosis. The affected animal was communally housed with 8 asymptomatic ferrets. This case prompted concerns from the owner and attending veterinarian regarding whether any of the 8 companion ferrets and the owners exposed to the same environment could be at risk of developing cryptococcosis from a localized, identifiable, heavily contaminated source. The ferrets had access to a confined outdoor area, which included some soil and a large, hollow eucalypt log provided for shelter and environmental enrichment. Our aims were therefore to: 1) determine, via culture of nasal swabs (to assess for colonization) and serologic testing (for subclinical disease) where possible, if exposure of the other ferrets to
Materials and methods
Case background
A 5-y-old, albino, castrated male (or gib) domestic ferret residing in western Sydney, Australia, had a 4-wk history of violent sneezing, with a bilateral bloody purulent nasal discharge. The ferret had access to a confined outdoor area, which included some soil and a large hollow eucalypt log provided for environmental enrichment and as a place of refuge. On physical examination, marked facial asymmetry was noted, with swelling of the nasal bridge on the right side (Fig. 1). Cytology was performed on the nasal discharge, revealing numerous encapsulated yeasts displaying narrow-necked budding and degenerate inflammatory cells (largely macrophages and some neutrophils). In addition, there were some raised circular (0.5-cm diameter) alopecic skin nodules over the neck, face, and body. Cytologic examination of smears from fine-needle aspirates of the skin lesions also revealed encapsulated yeasts with an inflammatory infiltrate dominated by macrophages (Fig. 2). Based on the characteristic cytology, a preliminary diagnosis of cryptococcosis was made. The infection presumably started in the sinonasal cavity, with invasion of the adjacent bone and extension to subcutaneous tissues of the nasal bridge, and hematogenous dissemination giving rise to skin nodules.
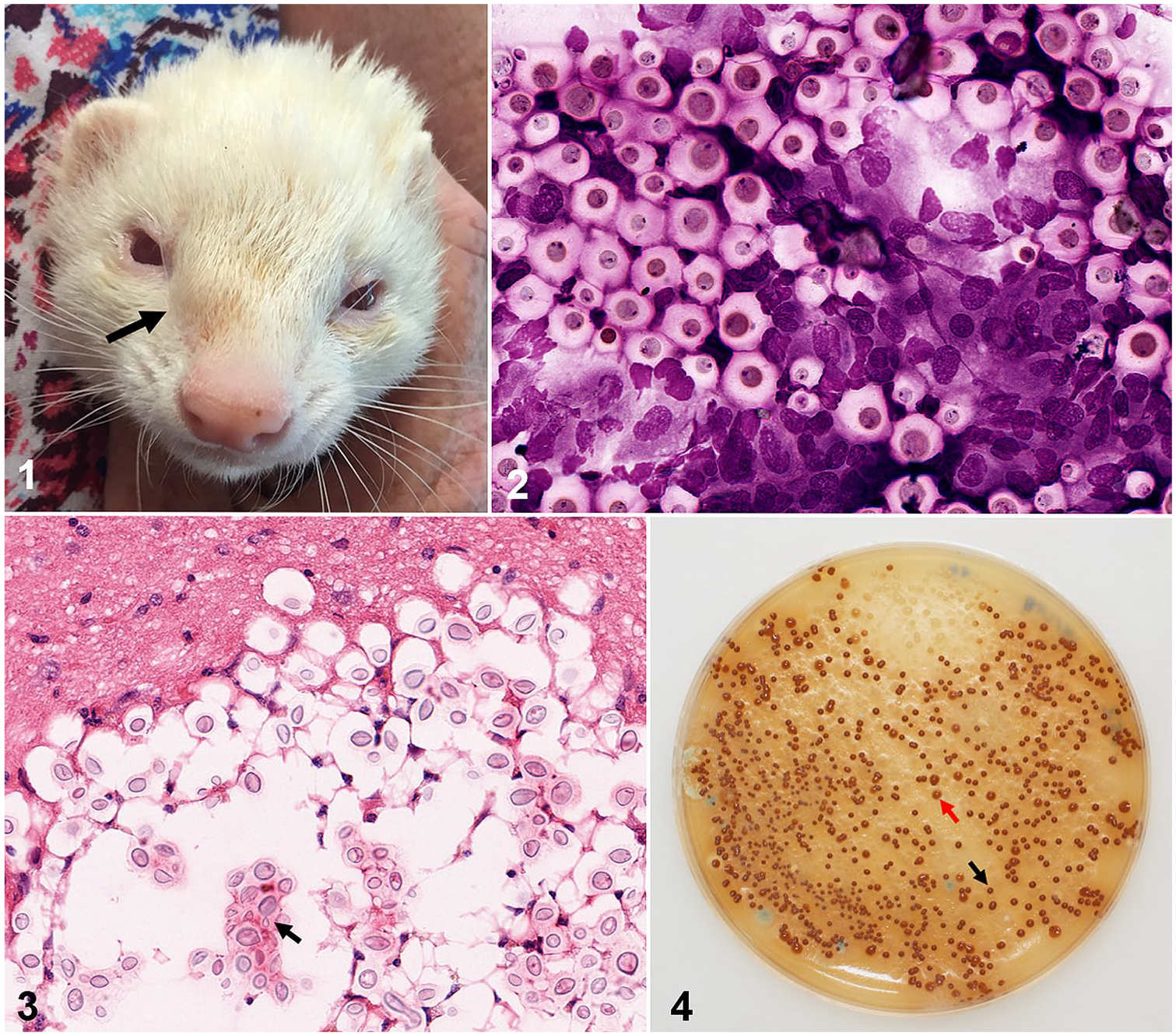
Cryptococcosis in a ferret.
The ferret was anesthetized by the attending veterinarian using isoflurane (IsoFlo Inhalation anesthetic; Abbott Australasia, Sydney, New South Wales, Australia) in 100% oxygen facilitating collection of blood by jugular venipuncture. After centrifugation to harvest serum, cryptococcal antigen testing was performed using both a lateral flow assay (LFA; CrAg LFA; IMMY, Norman, OK) and a latex cryptococcal antigen agglutination test (LCAT; CALAS; Meridian Bioscience, Cincinnati, OH) by Veterinary Pathology Diagnostic Services (VPDS) at The University of Sydney. Serologic testing was positive using both the LFA and LCAT, with the latter recording a reciprocal titer of 1,024, consistent with clinical cryptococcosis in a ferret. 22 Therapy was commenced with itraconazole (Sporanox 100 mg capsules; Janssen, North Ryde, New South Wales, Australia) at a dose of 10 mg/kg once daily orally (mixed in with the ferret’s normal food). No further diagnostic work-up was performed to assess possible underlying causes of immunosuppression. No possible causes of immunosuppression (e.g., lymphoma) 6 were indicated by the history or physical examination, although further investigations were precluded because of financial constraints.
By 4 wk after commencing therapy, nasal discharge and sneezing had resolved, but nasal bridge swelling was still evident. The skin lesions resolved slowly over the next few months. Although the medication was generally well-tolerated, intermittent periods of anorexia resolved when itraconazole therapy was interrupted for 2–3 d. By 12 mo after commencement of therapy, the facial asymmetry had disappeared. The ferret was maintained on itraconazole therapy because complete resolution of the cryptococcal antigenemia was considered unlikely at this stage and further serologic testing was not possible because of financial constraints.
The ferret had been treated with itraconazole for 18 mo when it developed generalized weakness, inappetence, polydipsia, an unkempt coat, and became acutely dyspneic. On physical examination, cutaneous bruising and subcutaneous ecchymoses on the ventrum were evident, accompanied by marked dyspnea and tachypnea. The ferret was in poor body condition, with dull mentation and pale mucous membranes. The owner opted for euthanasia without further investigation. The body was submitted to VPDS for postmortem examination, where hemoabdomen and a ruptured splenic hemangiosarcoma with hepatic metastases were identified. Histologic examination of tissues revealed microscopic cryptococcal lesions in the cerebrum (Fig. 3) and nasal turbinates. Other age-related abnormalities observed incidentally on gross and microscopic examination included chronic renal disease and dilated cardiomyopathy.
Sample collection
Pooled soil samples (3) from pot plants and garden beds to which ferrets had access, and a log within the enclosure, were submitted by the attending veterinarian. Samples from the log were obtained by running moistened sterile cotton swabs thoroughly over its surface.
Nasal swabs from 8 asymptomatic ferrets, co-habiting the same enclosure, were collected by the attending veterinarian at the request of their owner, by inserting a sterile, moistened, thin-tipped cotton urethral swab into the nares and rotating gently while under gentle manual restraint. One nasal swab was obtained from each individual over several weeks after the diagnosis of the clinical case as part of the disease investigation. A convenience blood sample from one asymptomatic ferret was collected through jugular venipuncture while it was under general anesthesia to investigate an intra-abdominal mass (later determined to be intestinal adenocarcinoma), and an aliquot was available for serologic testing. A sample of serosanguineous peritoneal fluid, collected from the clinical case at postmortem, was also available for serologic testing.
A swab of the nasal exudate from the clinical case, also collected by the attending veterinarian at the time of diagnosis using a similar method to that described above, was available for mycologic culture. All samples were submitted to VPDS. The body of the ferret was also submitted after euthanasia, and several tissue samples were collected for mycologic culture during the postmortem examination (nasal turbinate, lung, and brain).
Serology
Serum antigenemia was determined using the LFA (CrAg LFA; IMMY) and LCAT (CALAS; Meridian Bioscience) kits according to the manufacturers’ instructions (both including negative and positive controls) on a sample from one asymptomatic ferret. Serosanguineous abdominal fluid from the clinical case was also obtained at postmortem for LFA and LCAT testing. Samples with a positive LCAT result progressed to a titration procedure (performed using the same LCAT kit and according to the manufacturer’s instructions) to determine the antigen titer.
Culture
All swabs (environmental and nasal) were inoculated onto bird seed agar (containing penicillin G [40 units/L] and gentamicin [80 mg/L]) by gently rolling the swabs across the entire surface of the plate. Soil samples were inoculated by first agitating ~5–10 g of the sample in ~50 mL of sterile saline, allowing the mixture to settle for 10 min and then spreading 1 mL of supernatant across the surface of a bird seed agar plate.
17
Tissue samples were inoculated directly onto the surface of the bird seed agar plates. All plates were incubated at 27°C and checked daily. A positive control plate (inoculated with known
Antifungal susceptibility testing
Two disease isolates, one obtained at the time of diagnosis (from nasal discharge) and the other at postmortem (from brain tissue), were retrospectively submitted to the Clinical Mycology Reference Laboratory at Westmead Hospital in Westmead, New South Wales, Australia for antifungal susceptibility testing using a commercial kit (Sensititre YeastOne; Thermo Scientific, Waltham, MA). Both isolates were tested against amphotericin B, fluconazole, flucytosine, itraconazole, and voriconazole, and results were interpreted using known epidemiologic cutoff values for members of the
Molecular and mating type determination
DNA was extracted from all isolates using a previously established protocol for fungi.
8
Restriction-fragment length polymorphism (RFLP) analysis of a PCR product of the
PCR amplification of the
Multilocus sequence typing and phylogenetic analysis
All isolates underwent multilocus sequence typing (MLST) according to the consensus scheme for
Concatenated MLST sequences were aligned by MUSCLE and underwent maximum-likelihood phylogenetic analysis, using the Kimura 2-parameter model
15
with gamma distribution, to construct a phylogenetic tree. Four standard strains were included for comparison and out-grouping (
Results
Serology
A serum sample from one asymptomatic ferret tested negative for cryptococcal antigen using both the LFA and LCAT. The serosanguineous abdominal fluid, collected postmortem from the clinical case, returned positive LFA and LCAT results (the latter test recording a reciprocal titer of 32, consistent with unresolved cryptococcosis). 22
Culture
All 3 swabs collected from the hollow log were heavily positive (>100 CFUs on the isolation plate) for
Antifungal susceptibility testing
Both isolates appeared to be susceptible to all antifungals tested.
Molecular and mating type determination
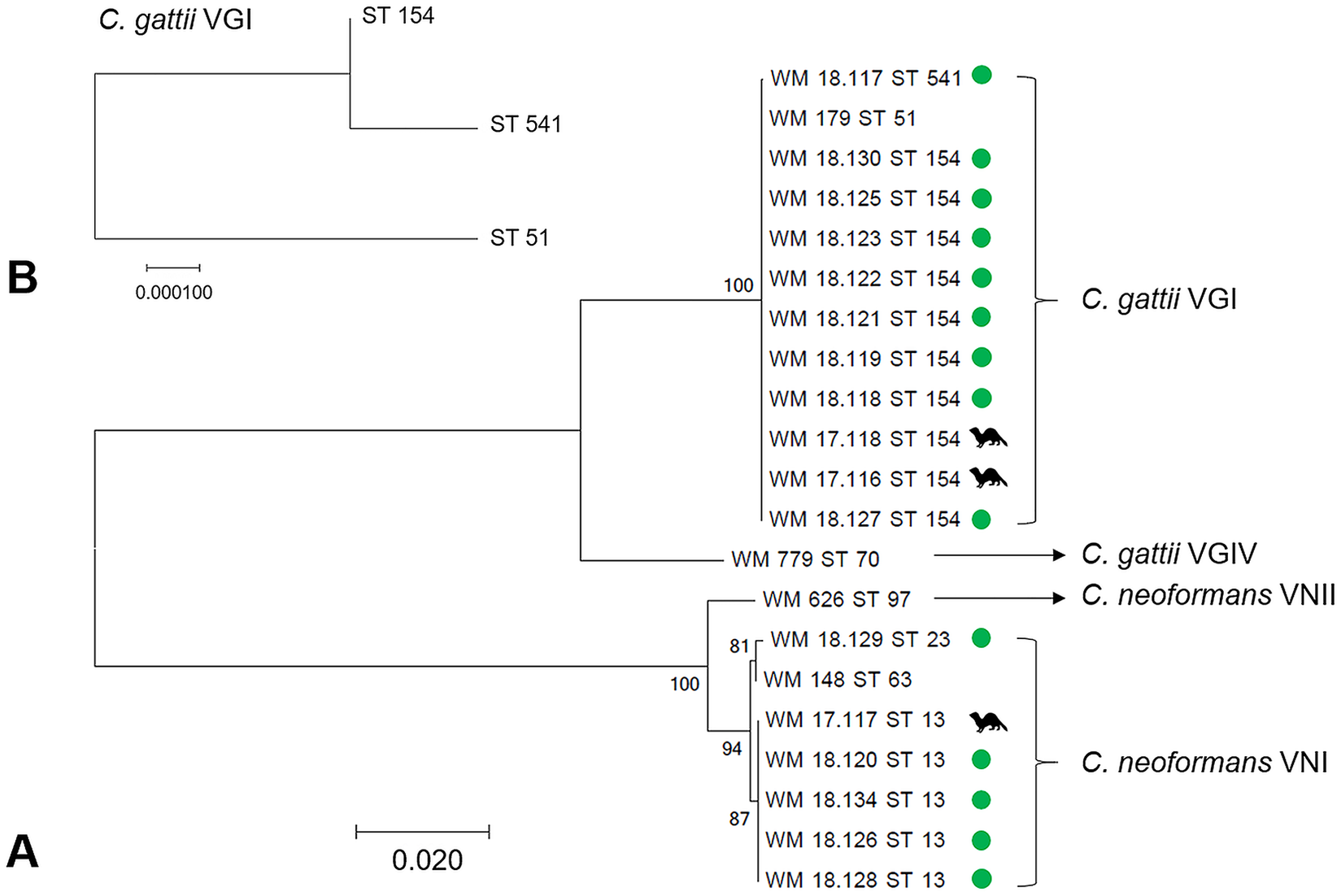
A total of 17 isolates proceeded to DNA extraction and molecular typing, including 14 environmental isolates collected from the positive log, a colonizing isolate from the nasal swab of an asymptomatic ferret, and 2 disease isolates from the clinical case (1 from the nasal exudate at the time of diagnosis, 1 from the brain at postmortem).
A total of 15 isolates were mating type α, whereas 2 isolates (both environmental
MLST and phylogenetic analysis
Among the 11
Discussion
We identified a hollow eucalypt log, used for shelter and environmental enrichment, as the likely point source of infection for
The 18-mo survival of a ferret with disseminated cryptococcosis is a good clinical outcome, considering the patient had a good quality of life before developing a terminal malignancy, with 2 concurrent age-related comorbidities (dilated cardiomyopathy and chronic renal disease). The prognosis for disseminated cryptococcosis in ferrets is not well-established but could reasonably said to be guarded, based on a review of prior case reports.
36
The decision to use itraconazole was based largely on the ease with which itraconazole capsules can be opened and mixed with palatable food. Ideally, antifungal susceptibility testing should have been undertaken prior to treatment to determine minimum inhibitory concentrations for itraconazole and fluconazole, but financial constraints made this impossible. Retrospective testing of disease isolates suggested that they were susceptible to all antifungals tested (including itraconazole and fluconazole). This is consistent with the expectation that cases of cryptococcosis on the east coast of Australia are usually susceptible to both agents, given that the
With the benefit of hindsight, it seems likely that this ferret had asymptomatic CNS disease at the time of initial diagnosis, but that long-term itraconazole therapy was enough to prevent this developing further. Indeed, the substantial clinical improvement suggested efficacy of therapy. Microscopic CNS lesions have been detected through postmortem histologic examination of tissues in cases of cryptococcosis (in the absence of CNS-specific clinical signs), including in koalas and ferrets.32,36 Given the full clinical resolution with itraconazole monotherapy, alterations to the treatment regimen were not considered necessary, but other options could have included a higher dose of itraconazole, 22 the substitution of itraconazole by another triazole antifungal with better CNS penetration (such as fluconazole), 7 or the addition of amphotericin B (administered subcutaneously or intravenously).21,31 Our findings of microscopic foci of ongoing infection (despite full resolution of clinical signs) also emphasize the value of sequential antigen titers. Unfortunately, serial LCAT titer determinations were beyond the financial resources of the owner. Therapeutic drug monitoring would have been ideal to ensure that dosing was producing effective itraconazole levels in blood, given that the pharmacokinetics have not been determined for antifungal drugs in ferrets.
Most of the environmental
MLST results, along with absence of positive cultures from any other environmental samples, provide high support for the notion that this single eucalypt log was the point source of disease and nasal colonization. The log was therefore removed from the enclosure. Aerosolization of
Our findings are similar to a study in which a likely environmental source of infection for
Our findings raise a question similar to prior studies: why did only 1 of 9 ferrets in contact with this source of infection develop clinical cryptococcosis? In our case, the answer remains unclear. Genetic susceptibilities could play a role, as suggested by the report of cryptococcosis in 2 sibling ferrets. 22 In the avian case mentioned previously, a high-stress period was considered the likely cause of increased susceptibility. 33 In one ferret case, concurrent lymphoma was reported as a likely cause of immunosuppression, leading to cryptococcosis. 6 It is also possible that more of the in-contact asymptomatic ferrets were colonized but this remained unidentified because of either transient colonization or the poor performance of the relatively superficial swabs able to be collected safely while under manual restraint. General anesthesia could have allowed for the collection of potentially more representative nasal samples (either deeper swabs or nasal flushes), along with blood samples from all asymptomatic ferrets to test for subclinical cryptococcosis, but this was not possible for logistical reasons. It may also have been of interest to test the human owners for cryptococcal antigenemia and nasal colonization, given that colonized humans were identified in association with another case of ferret cryptococcosis, 28 but this was also not possible.
The mixed mating type population (9 type α and 2 type
Based on our findings, and our knowledge of the environmental niches of pathogenic
We successfully identified an environmental point source of infection for
Supplemental Material
Supplemental_material – Supplemental material for Identification of the environmental source of infection for a domestic ferret with cryptococcosis
Supplemental material, Supplemental_material for Identification of the environmental source of infection for a domestic ferret with cryptococcosis by Laura J. Schmertmann, Alison Wardman, Laura Setyo, Alex Kan, Wieland Meyer, Richard Malik and Mark B. Krockenberger in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Suzanne Taylor for her assistance and willingness to allow this case and the subsequent investigation to be published. We also thank Patricia Martin and Christine Black at the Veterinary Pathology Diagnostic Services, The University of Sydney, for their advice and assistance in performing part of the culturing and serologic testing. We thank Veronica Ventura at Sydney School of Veterinary Science, The University of Sydney, for providing the bird seed agar and Krystyna Maszewska at The Westmead Institute for Medical Research, for storing the isolates in the Medical Mycology Culture Collection. We also thank Catriona Halliday of the Clinical Mycology Reference Laboratory at Westmead Hospital for arranging the antifungal susceptibility testing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
R Malik’s position is supported by the Valentine Charlton Bequest. Our research was supported by the Betty Rosalie Richards Bequest at The University of Sydney.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
