Abstract
Accurate staging of hepatic fibrosis (HF) is important for treatment and prognosis of canine chronic hepatitis. HF scores are used in human medicine to indirectly stage and monitor HF, decreasing the need for liver biopsy. We developed a canine HF score to screen for moderate or greater HF. We included 96 dogs in our study, including 5 healthy dogs. A liver biopsy for histologic examination and a biochemistry profile were performed on all dogs. The dogs were randomly split into a training set of 58 dogs and a validation set of 38 dogs. A HF score that included alanine aminotransferase, alkaline phosphatase, total bilirubin, potassium, and gamma-glutamyl transferase was developed in the training set. Model performance was confirmed using the internal validation set, and was similar to the performance in the training set. The overall sensitivity and specificity for the study group were 80% and 70% respectively, with an area under the curve of 0.80 (0.71–0.90). This HF score could be used for indirect diagnosis of canine HF when biochemistry panels are performed on the Konelab 30i (Thermo Scientific), using reagents as in our study. External validation is required to determine if the score is sufficiently robust to utilize biochemical results measured in other laboratories with different instruments and methodologies.
Introduction
Assessment of hepatic fibrosis (HF) is of major importance for the management and prognosis of canine chronic hepatitis. Epidemiologic data, biochemical analysis, and diagnostic imaging provide useful information, but liver biopsy remains the gold standard in the assessment of HF. Histologic examination allows assessment of the severity of disease by estimating the extent of fibrosis and degree of inflammation. However, obtaining a suitable liver biopsy from dogs with liver disease may be difficult because of anesthetic and procedural risks, and owner preferences. 12
Noninvasive approaches based on the measurement of several blood biomarkers have been validated and are widely used in human medicine.5,6,16,21,25,28,30,31,40 HF scores combine several biochemical and/or clinical parameters in a proprietary algorithm, helping to indirectly stage liver disease and thereby decrease the need for a liver biopsy. The scores were first developed to assess HF in hepatitis C virus infection and were adapted for each of the 3 main causes of chronic liver diseases in human medicine: chronic viral hepatitis, alcoholic liver disease, and non-alcoholic fatty liver disease. 35 The FibroMeter VIRUS (Echosens, Paris, France) is a clinical score used to evaluate and manage HF chronic viral hepatitis that uses an algorithm based on age, platelet count, and biochemical data (alpha 2 macroglobulin, hyaluronic acid, prothrombin index, aspartate aminotransferase [AST], urea). 16 Evaluating the HF score in people aids treatment decisions, such as whether or not to prescribe an antiviral treatment, length of treatment, monitoring for cirrhosis complications, and treatment efficacy assessment. 36
Similarly, in veterinary medicine, it would be desirable to have a simple, quick, and noninvasive method for early detection of dogs potentially affected by HF. We developed a HF score to identify dogs with moderate or marked HF, which could help clinicians rank differential diagnoses and detect patients that need further investigation such as liver biopsy.
Materials and methods
Animals
Dogs were included in our study prospectively from January 2014 to December 2016 from 5 French veterinary referral centers (Clinique vétérinaire des Cerisioz, Vetagro Sup, Centre Hospitalier Universitaire Vétérinaire d’Alfort, Centre Hospitalier Vétérinaire Frégis, and Clinique Vétérinaire Aquivet). To investigate abnormal liver enzyme activity, we included client-owned dogs ≥2 y old that had undergone a liver biopsy. Exclusion criteria were: focal liver lesion, extrahepatic cholestasis on abdominal ultrasound, glucocorticoid administration within the previous 30 d, any history of phenobarbital exposure, extrahepatic comorbidities that could have influenced serum liver enzyme activity as determined on an individual case basis, or histologic diagnosis of neoplasia on liver biopsy.
A group of 5 adult Beagles, housed at the Veterinary Small Animal Teaching Hospital Lyon at VetAgro Sup University (France), were used as a healthy control group. These dogs had unremarkable findings on physical examination, biochemistry panel, abdominal ultrasound, and liver biopsy, and had no history of medication. The study protocol was approved by an ethics committee [VetAgro Sup Ethical committee: 201504271322700 v4 (APAFIS 554)], and informed owner consent was obtained for each dog.
Blood sample collection and processing
Fasting blood samples were collected in non-additive tubes on the day of the liver biopsy. Serum was separated 30–60 min after sampling by centrifugation at 2,500 × g for 10 min at room temperature. None of the samples were hemolyzed or lipemic. Samples were immediately frozen at −20°C and shipped to the same laboratory for biochemical analysis (Laboratoire de biologie médicale, VetAgro Sup). They were stored at −80°C prior to analysis, and all biomarkers were batched and measured within 3 mo from collection.
Biochemical analysis
An automated chemistry analyzer (Konelab 30i; Thermo Scientific, Cergy-Pontoise, France) was used to measure serum alanine aminotransferase (ALT), alkaline phosphatase (ALP), AST, gamma-glutamyl transferase (GGT), glutamate dehydrogenase (GLDH), lactate dehydrogenase (LDH), total bilirubin, bile acids, urea, glucose, cholesterol, triglycerides, sodium, and potassium according to the manufacturer’s instructions and using standard methods (Table 1). The coefficient of variation for each measured analyte was calculated during in-house validation of analytical methods (Table 1). In a previous study evaluating the effects of storage time on biochemical parameters, LDH decreased >20% after 3 mo of storage at −70°C. However differences from baseline were <20% for the other enzymes (ALT, ALP, AST, GGT, GLDH) and <10% for metabolites and minerals (glucose, cholesterol, triglycerides, potassium, sodium, total bilirubin) after 3 mo of storage at −70°C. 38
Coefficient of variation (CV) calculated during in-house validation, and reagents for analytes measured in canine serum on an automated biochemical analyzer (Konelab 30i; Thermo Scientific, Cergy-Pontoise, France).
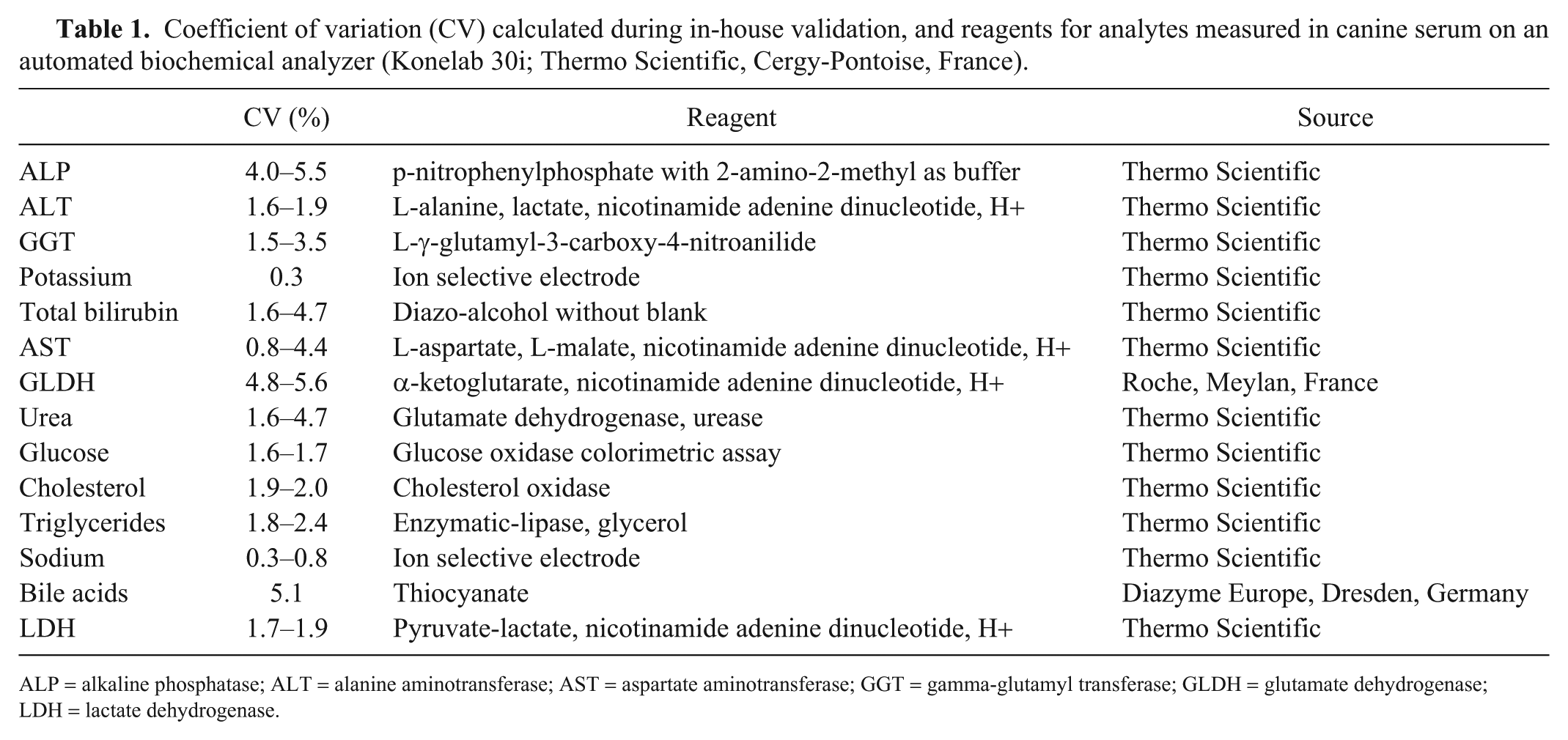
ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; GGT = gamma-glutamyl transferase; GLDH = glutamate dehydrogenase; LDH = lactate dehydrogenase.
Liver biopsy sampling
Liver biopsy samples were obtained by an ultrasound-guided needle biopsy technique using a 14- or 16-gauge disposable needle, or during laparotomy or laparoscopy. Laparotomy, laparoscopy, or ultrasound-guided biopsy were offered to owners; most chose percutaneous biopsy. Consistent with the recommendation of the World Small Animal Veterinary Association (WSAVA) liver standardization group, a minimum of 2 biopsy specimens were obtained from at least 2 different liver lobes. 32 Samples were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin for histologic examination. Picrosirius red staining, which allows for improved visualization of collagen type I and type III fibers, was used to assess HF. Biopsies were considered suitable for assessment of fibrosis if the length was >10 mm and the samples were collected at least 1 mm below the hepatic capsule.
Liver histologic analysis
To ensure diagnostic consistency, the same pathologist (M Chevallier) performed all histologic examinations. The pathologist was blinded to biochemical profile results. The extent and pattern of fibrosis was evaluated according to an established histologic scoring system. The distribution of fibrous deposits, their loose or dense character, and their intensity were assessed. Fibrosis was staged as: F0 = no fibrosis, F1 = portal fibrosis, F2 = moderate portal fibrosis with minimal bridging, F3 = bridging fibrosis present in all zones of the hepatic lobule, and F4 = cirrhosis (i.e., severe fibrosis with nodular regeneration). 2 When the HF histologic stage was between stages, the liver biopsy was classified as the higher stage.
Statistical analysis
Quantitative variables were expressed as the mean ± standard deviation, unless otherwise specified. Descriptive analysis for each blood parameter was performed relative to the diagnostic target, which was defined as a HF histologic stage ≥F2.
The dogs were randomly divided into a training set of 58 dogs and a validation set of 38 dogs. The training set was used to build the fibrosis predictive score expressed as a probability of having a HF histologic stage ≥F2. Univariate analysis using the Wilcoxon test was used to assess the association between blood parameter and outcome, which was also HF histologic stage ≥F2. For each marker studied, diagnostic performance was expressed by the area under the receiver operating characteristic curve (AUC), sensitivity, and specificity. Diagnostic cutoffs were determined according to the maximum Youden index. 17 Principal component analysis was used to identify the most explanatory parameters. Multivariate analysis using a logistic regression model was performed applying a backward procedure selection based on the Akaike information criteria. 1 This model was used to determine the fibrosis predictive score through the logit formula: 1/(1 + eα-x), wherein α is the estimate coefficient for each parameter; this score ranged from 0 to 1. The relevance of each blood parameter included in the model was evaluated by a Wald test.
To establish the binary classification, the fibrosis predictive score diagnostic cutoff was determined by setting the sensitivity at 80%. The AUC was used to assess the diagnostic performance of the score for fibrosis ≥F2. Agreement between HF score and HF histologic stage was made using the Cohen kappa coefficient. The AUC of the HF score was compared to the AUC of ALT and ALP as single markers using the Delong test. 17
The algorithm used to convert the 5 parameters into a score is not provided because it is proprietary (FibroVet Screening; Echosens, Paris, France).
All statistical analyses were performed using R v.3.0.1 (R Development Core Team; https://www.r-project.org/) and using the beanplot, Epi, caTools, and ROCR package stats. The statistical significance level was set at p ≤ 0.05.
Results
Population
From the 123 dogs enrolled during the study period, 96 met the inclusion criteria. We excluded 27 dogs because of extrahepatic cholestasis (n = 7), concomitant extrahepatic diseases that could have altered serum liver enzyme activity (hyperadrenocorticism, hypothyroidism, or diabetes mellitus; n = 6), diagnosis of neoplasia on histologic examination (n = 4), and unsuitable liver biopsy for fibrosis assessment because of small sample size or fragmentation (n = 10).
The final study group included 9 Scottish Terrier, 9 Bichons Frise, 8 Labrador Retriever, 8 Cocker Spaniel, 6 Poodle, 5 Beagle, 3 West Highland White Terrier, 2 Yorkshire Terrier, 2 Dachshund, 2 German Shepherd, 2 Cairn Terrier, 2 Chow Chow, 2 Pinscher, 2 cross-breed Griffon, 1 Korthals Griffon, 1 Vendeen Griffon, and 32 mixed-breed dogs. There were 34 male and 62 female dogs. Mean age at the time of diagnostic procedure was 8.1 ± 3.5 y. There was no significant difference in age between the training set (n = 58) and validation set (n = 38; p = 0.52).
Biochemical analysis
Significant differences in biochemical parameters were not observed between the training and validation sets (Supplementary Table 1).
Liver biopsy samples and histologic analysis
Liver samples were collected by ultrasound-guided needle biopsy in 88 dogs, by laparoscopic biopsy in 3 dogs, and by laparotomy in 5 dogs. Mean number of portal triads per biopsy (min, max) was 13.7 ± 9.7 (3–63), and mean sample size was 23.9 ± 78.7 mm2 (10–735; Figs. 1, 2).
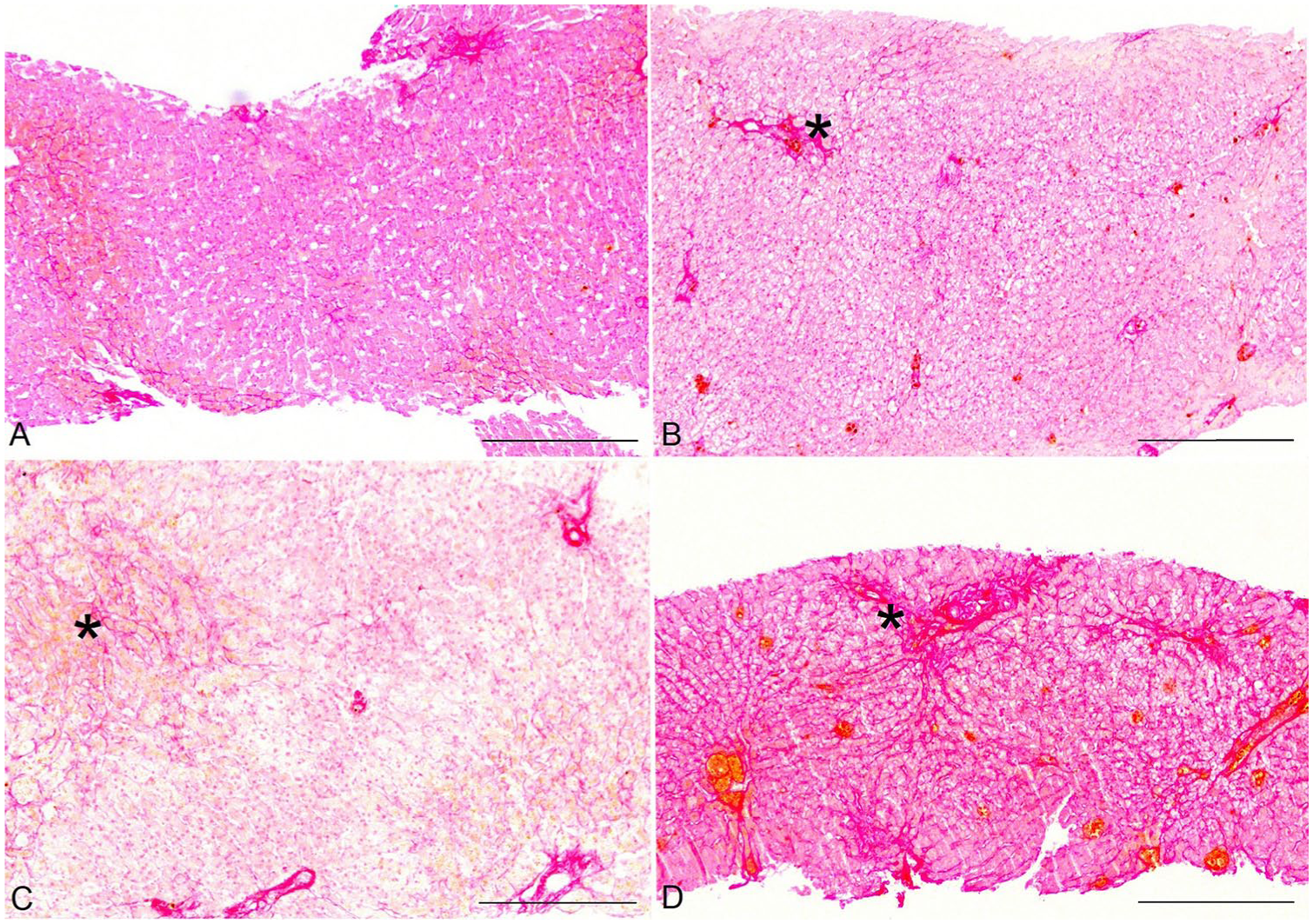
Histologic stages of canine hepatic fibrosis.
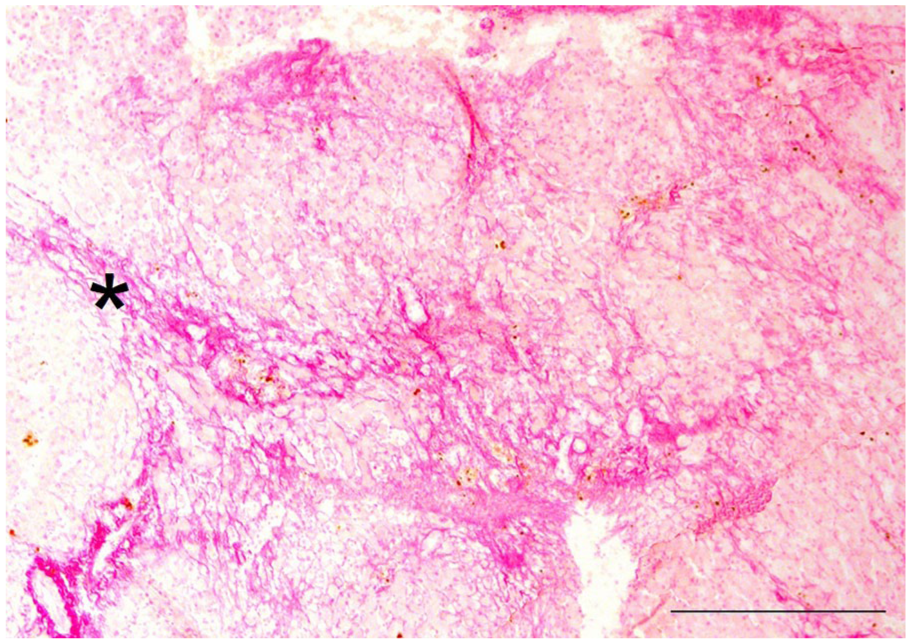
Hepatic fibrosis histologic stage F4 (cirrhosis) on histopathology, indicated by asterisk. Picrosirius red staining. Bar = 500 μm.
Final histologic diagnoses were vacuolar hepatopathy (n = 18), chronic hepatitis (n = 12), cirrhosis (n = 11), fibrosis (n = 8), cholangitis (n = 6), reactive hepatopathy (n = 6), acute hepatitis (n = 5), eosinophilic hepatitis (n = 2), granulomatous hepatitis (n = 1), vascular liver disorders (n = 3), or lobular dissecting hepatitis (n = 2). No significant histologic changes were observed in 22 cases, including the 5 healthy dogs.
Thirty-nine of the 96 dogs (41%) had HF histologic stage ≥F2. HF was absent in 24 dogs (including the 5 healthy dogs), mild (stage F1) in 33 dogs, moderate (stage F2) in 17 dogs, and severe (stage F3 and F4) in 22 dogs (Fig. 3). The prevalence of HF histologic stage ≥F2 was 36% in the training set and 47.4% in the validation set, which was not significantly different (p = 0.38).
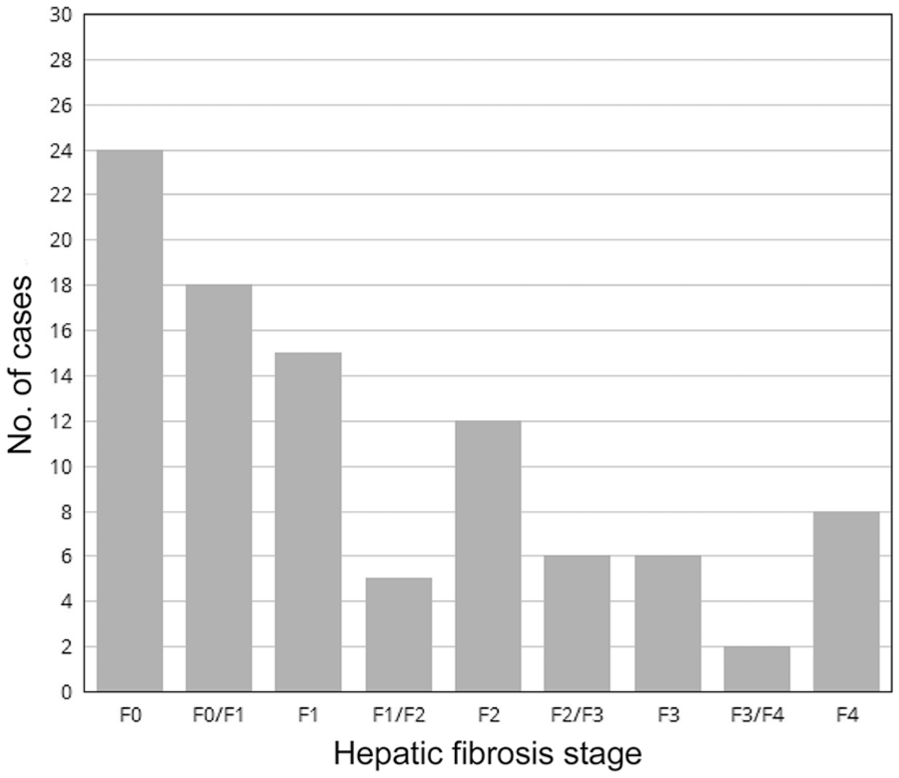
Prevalence of histologic hepatic fibrosis in the study population.
Hepatic fibrosis score
Among the measured parameters, only serum ALP (p = 0.011), total bilirubin (p = 0.013), GGT (p = 0.027), bile acids (p = 0.009), and potassium concentration (p = 0.035) were able to discriminate between no or mild fibrosis (<F2) and moderate-to-marked fibrosis or cirrhosis (≥F2). ALT and GLDH were not significantly associated with HF (Table 2). ALP, bilirubin, bile acids, and GGT were positively associated with fibrosis, whereas potassium was negatively associated (Supplementary Figs. 1–5). ALT activity with a cutoff value of 270 IU/L (approximately twice the upper normal limit) appeared to be a poor fibrosis marker, with an AUC of 0.59 (Table 3). The biochemical parameters were combined using logistic regression to predict the presence of HF histologic stage ≥F2. The HF score was composed of 5 parameters with p ≤ 0.05 (ALT, ALP, total bilirubin, GGT, and potassium). The AUC of the derived score probability on the whole database was 0.80 (Se = 80%; Sp = 70%). Validation set performances were not significantly different, with an AUC of 0.83 (Se = 83%; Sp = 70%). Agreement between commercial HF score and HF histologic stage was moderate (kappa coefficient = 0.46; 95% confidence interval = 0.39–0.54; Table 4).
Comparison of biochemical values in 96 adult dogs with and without hepatic fibrosis.
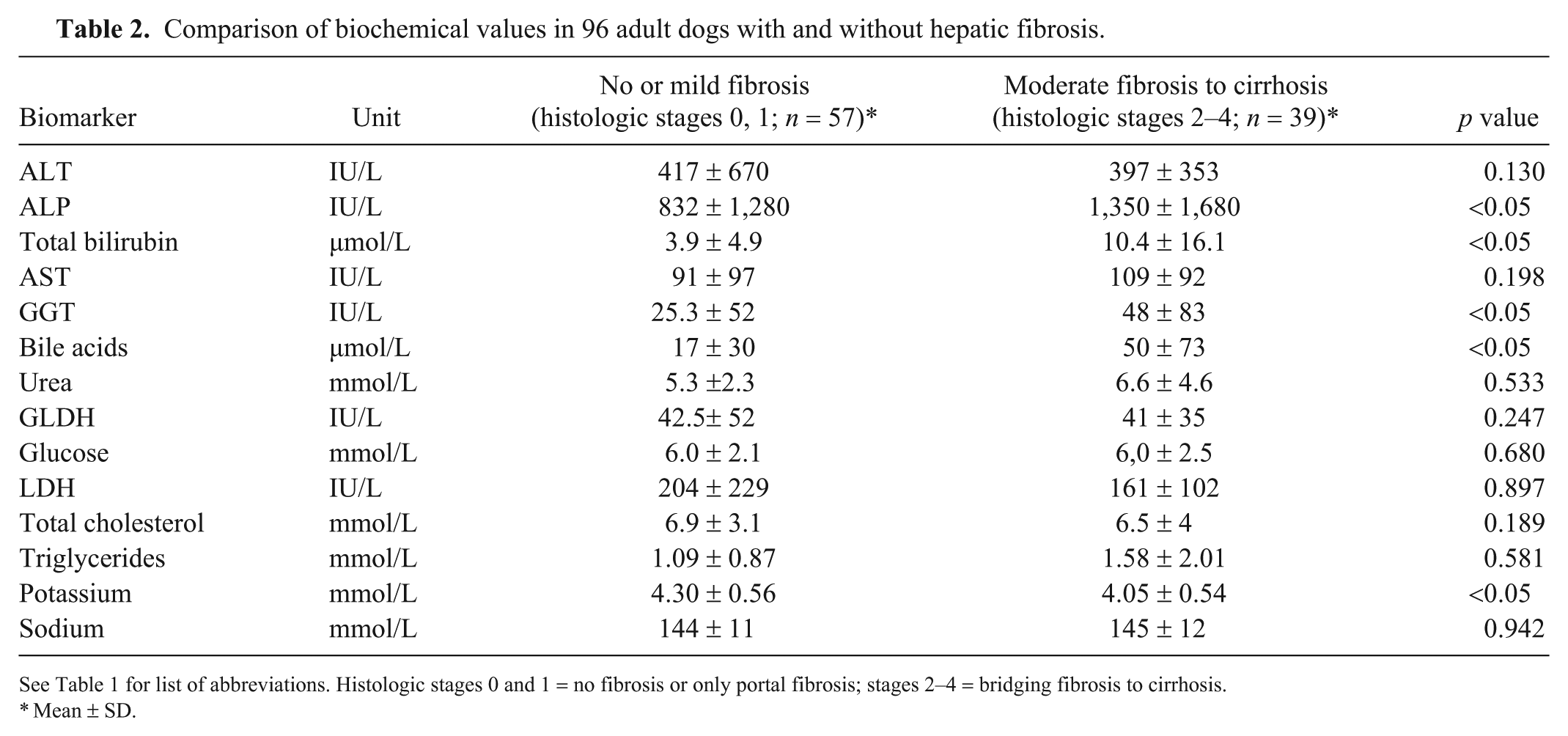
See Table 1 for list of abbreviations. Histologic stages 0 and 1 = no fibrosis or only portal fibrosis; stages 2–4 = bridging fibrosis to cirrhosis.
Mean ± SD.
Diagnostic performance of biochemical values in 96 adult dogs used to create a proprietary hepatic fibrosis score (n = 96).
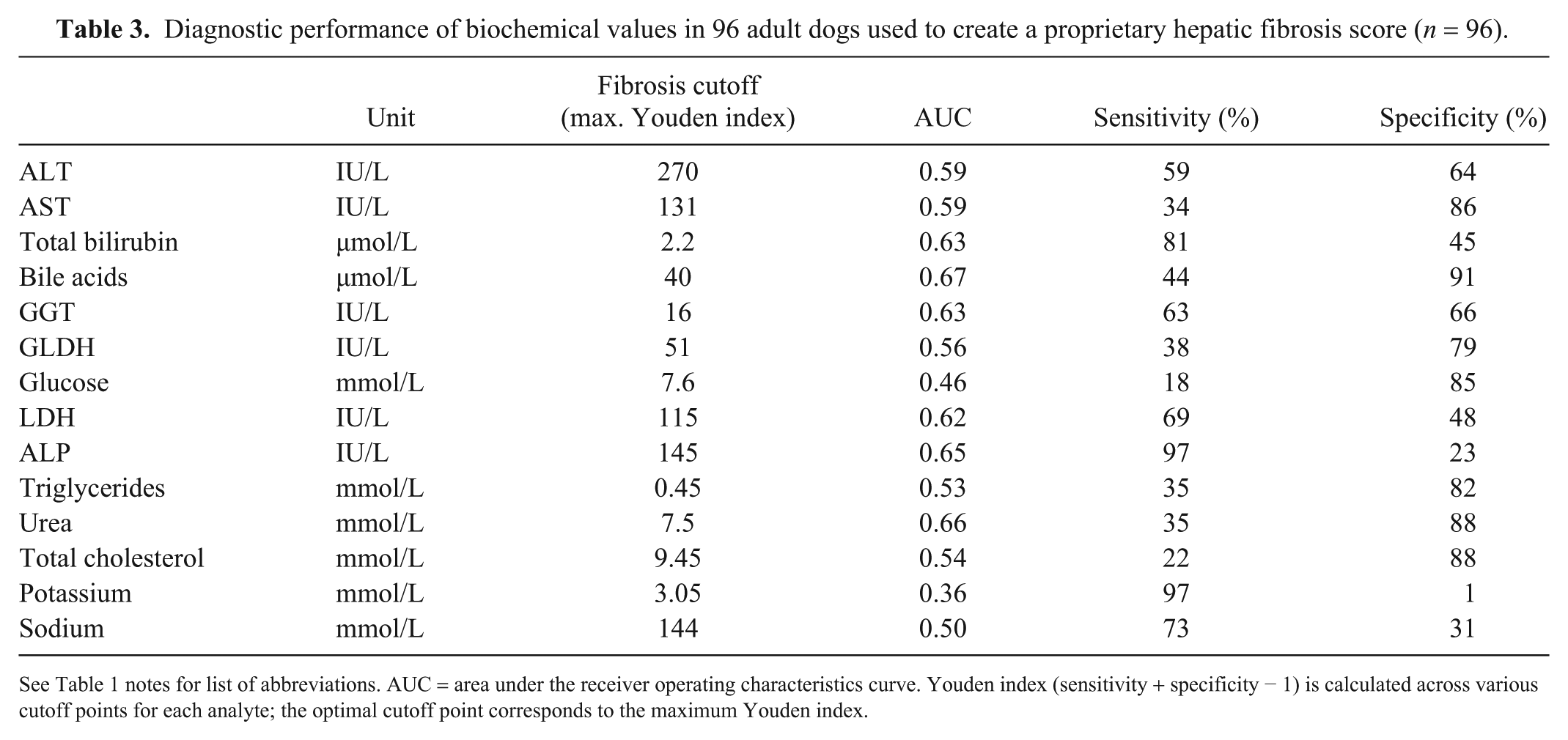
See Table 1 notes for list of abbreviations. AUC = area under the receiver operating characteristics curve. Youden index (sensitivity + specificity − 1) is calculated across various cutoff points for each analyte; the optimal cutoff point corresponds to the maximum Youden index.
Agreement between hepatic fibrosis (HF) score and HF histologic stage.

Kappa coefficient = 0.46 (95% confidence interval = 0.39–0.54).
HF score AUC is significantly different from ALP and ALT AUC (Fig. 4). HF score was significantly higher in dogs with HF histologic stage ≥F2 than in dogs with HF histologic stage <F2 (p < 0.001; Fig. 5).
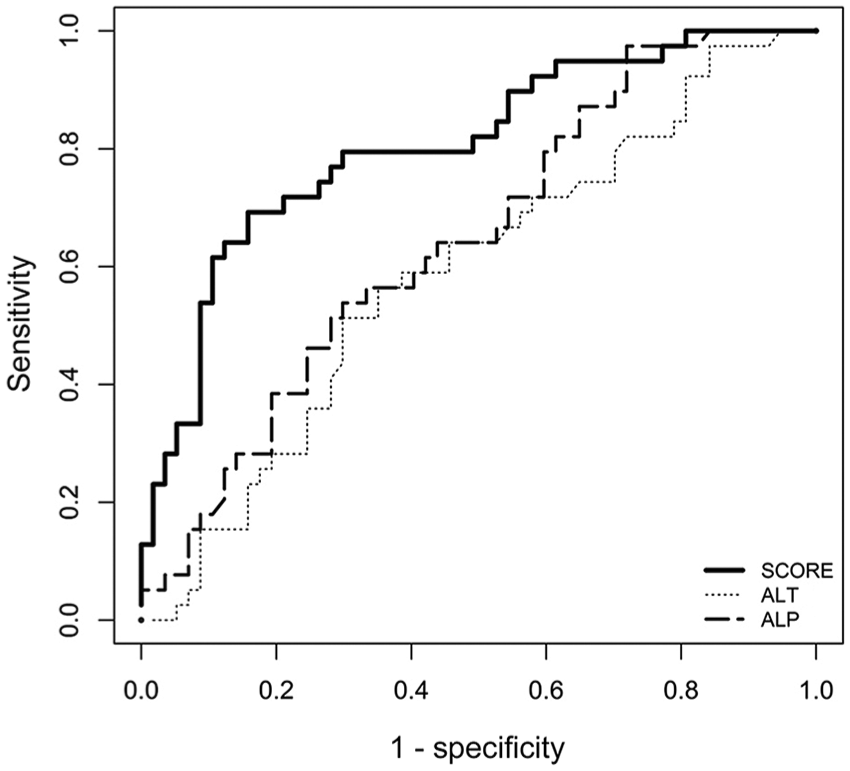
Receiver operator characteristic curve for the diagnosis of the hepatic fibrosis (HF) score, alanine aminotransferase (ALT), and alkaline phosphatase (ALP) for the diagnosis of hepatic fibrosis stage ≥F2 in 96 adult dogs. The prevalence of HF histologic stage ≥F2 was 40.6%, and the area under the curve was 0.804 (0.714–0.895) for the HF score, 0.588 (0.475–0.706) for ALT, and 0.646 (0.543–0.761) for ALP.
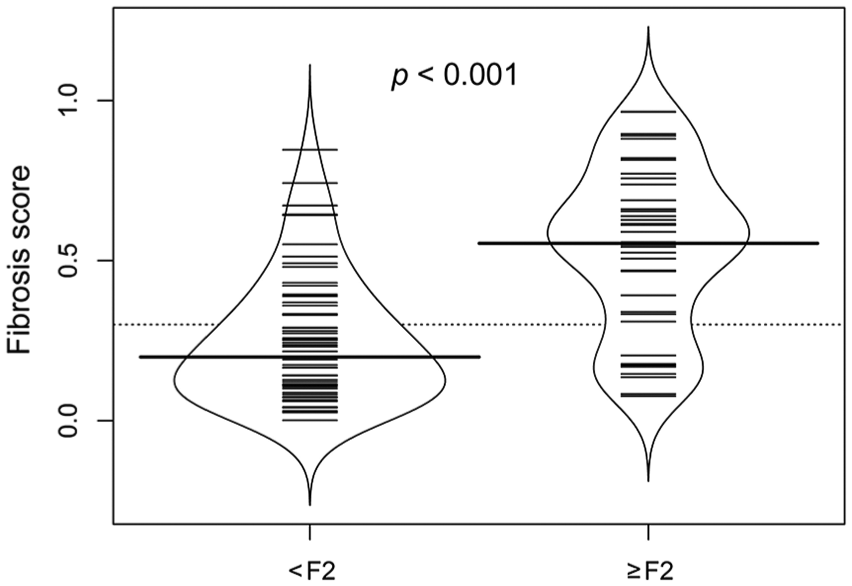
Bean plots representing the hepatic fibrosis (HF) score in 96 adult dogs with and without HF histologic stage ≥F2 (p < 0.001). HF score was significantly higher in dogs with HF histologic stage ≥F2 than in dogs with HF histologic stage <F2 (p < 0.001). Bold bar = median; small bars = individual data points (if there are “n” identical values, the line is drawn “n” times as long as the number of identical values); blue line = density shape.
Discussion
Increased serum liver enzyme activity might be noticed when a dog is presented for a health checkup or because there are clinical signs consistent with liver disease. The HF score described in our study was developed to detect dogs that need further investigation, such as liver biopsy. A HF score consistent with moderate or greater fibrosis may be an argument for performing a liver biopsy. Conversely, the absence of HF may allow the clinician to monitor liver enzyme activity before performing more invasive diagnostic tests, especially if clinical signs are absent.
Our results suggest that a commercial HF score based on common biomarkers (ALT, ALP, total bilirubin, GGT, and serum potassium concentration) could be used as a screening test for moderate or greater HF, with sensitivity and specificity of 83% and 70%, respectively. There was no significant difference for the HF score performances between the training and validation sets.
In practice, extrahepatic disease or concurrent drug administration should be ruled out before using the HF score. In fact, none of the biomarkers used are individually specific for HF detection. However, the combination of several markers in a score allowed HF to be detected noninvasively. We excluded from our study young animals, animals with comorbidities such as hyperadrenocorticism, and animals receiving treatment that may increase serum liver enzyme activity, or with extrahepatic cholestasis. Only samples without hemolysis or lipemia should be used when using the HF score because they can interfere with many assays. 3
A tool to calculate the HF score is available online. Veterinarians can theoretically measure all 5 biochemical parameters in their clinical practice and then enter the results via a website. According to the score, the score is positive (high risk of moderate or greater HF) or negative (low risk of HF). However, we performed analyte measurements in only one laboratory on the Konelab 30i using specific reagents, and the HF score cannot be generalized to biochemical values evaluated at another laboratory, on another instrument, or with other reagents. External validation is required to know if these findings hold true when measurement of analytes is performed in other laboratories with other instruments and reagents. Intrinsic biological heterogeneity of blood parameters 34 and variability of assay or laboratory techniques will affect the accuracy of HF scores. We performed biochemical analyses using the same automated analyzer in the same laboratory to avoid analytical variability. In human medicine, several HF scores are used to assess HF, and their robustness against the variability of composite blood variable is inconsistent (Calès P, et al. Meta-analysis of blood scores for liver fibrosis in chronic hepatitis C [Abstract 239]. Hepatology 2006;44(4 Suppl 1):188A–700A). 4
The effects of freeze–thaw cycles and storage time are poorly documented in canine serum. In a previous study including clinically healthy dogs, most of the enzyme activities (ALT, ALP, AST, GGT, GLDH) and analytes (glucose, cholesterol, triglycerides, potassium, sodium, total bilirubin) had minor differences (e.g., differences from baseline <20% for the enzymes and <10% for metabolites and minerals) after 3-mo storage at −70°C. 38 We could not rule out that those differences affected our results. LDH values decreased by >20% after 3 mo of storage at −70°C in a previous study 38 ; potential effects of freeze–thaw cycles and storage time are not documented for bile acids, to our knowledge. However, we did not include those analytes in our final HF score.
There are several other limitations of our study. The dogs recruited for our study were from referral centers and may not be fully representative of the wider population of dogs with increased liver enzymes and undergoing liver biopsy. We did not include dogs from first-opinion practices, which may have led to inclusion of dogs with a more severe increase in serum liver enzyme activity. An independent external validation protocol is needed for further assessment of the reliability of our scoring system.
Second, many different breeds were included, and the HF score accuracy may vary according to breed. For instance, Scottish Terriers have higher serum ALP activity than other breeds, 26 and ALP was positively associated with HF, which may in turn increase the sensitivity and decrease specificity of the tests for these dogs. Ideally, a HF score would be built or validated for each breed, which is not feasible given the large number of dog breeds. However, breed-specific HF scores for the breeds commonly seen in veterinary practice with chronic hepatitis, such as Labrador Retrievers and Cocker Spaniels, might be clinically useful. 39
Other potential limitations of our study included a potential inaccurate assessment of HF as a result of sampling error, small sample size, nonhomogeneous distribution of lesions, and subjective evaluation of morphologic changes. Inaccurate evaluation as a result of nonhomogeneous distribution of lesions within the liver could not be ruled out. A liver biopsy only evaluates a small sample of tissue, and there can be site-to-site variation even within a liver lobe. 20 In our study, at least 2 biopsies from 2 different lobes were taken, and we excluded specimens unsuitable for fibrosis assessment to minimize pre-analytical error. According to the WSAVA liver standardization group, 2 well-taken needle biopsies are usually sufficient to permit reliable evaluation, 32 and biopsy of at least 2 liver lobes identified the predominant histologic abnormality in 98.6% of cases. 20 Liver biopsy by laparotomy allows larger biopsies to be sampled than do percutaneous needle or laparoscopic biopsy. 23
However, according to a previous study, histologic interpretation of a liver biopsy specimen is unlikely to vary if it contains at least 3–12 portal triads. 19 The mean number of portal triads per biopsy in our study was 13.7 ± 9.7 (3–63), which should allow accurate histologic assessment.
The reliability of liver biopsies to assess HF may also be questioned because of inter-observer variability. In a previous veterinary study, 3 pathologists agreed on the morphologic diagnosis assigned to needle (18 gauge) and wedge biopsy for 44% and 65% of the samples, respectively. 7 However, the use of an 18-gauge needle did not fit the current WSAVA liver standardization group recommendations, which recommends 14-gauge needle samples in most dogs and 16-gauge needle samples for small patients. This inter-observer variability is also present in human medicine.33,35 In our study, histologic examination was performed by a single human hepatopathologist specialized in comparative pathology to ensure diagnostic consistency. Moreover, we used a grading scheme to decrease the variability of assessment. In a previous study, there was fair agreement between pathologists when a histologic scoring scheme was used. 24
Finally, the use of HF scores to monitor disease progress and potential response to treatment remains to be investigated. In fact, repeated liver biopsies are often recommended to evaluate the efficacy of treatment, but are not usually performed in a clinical setting. A HF score allowing assessment of treatment efficacy would be of interest. However, as glucocorticoids are frequently used in the therapy of idiopathic chronic hepatitis,13,37 this may modify the results of the score given that the drug increases serum activity of 3 of the 5 analytes used to calculate the HF, namely ALT, ALP, and GGT. 11
In human medicine, the accuracy of these HF scores depends on the underlying process (e.g., viral hepatitis vs. nonalcoholic fatty liver disease). 8 Growing understanding of the pathogenesis and etiology of canine liver disease will potentially improve the accuracy of noninvasive blood biomarkers of HF. The main reason for liver biopsy in humans is to assess the degree of fibrosis, whereas in dogs the goal is often to establish a specific diagnosis. Many noninvasive tests are available in humans to identify the cause of hepatic disease (e.g., serologic testing for viral hepatitis); liver biopsy also allows for the collection of samples to diagnose an underlying hepatic disorder leading to fibrosis in dogs (e.g., copper-associated hepatitis). Research of biomarkers is also ongoing for copper storage disease. For instance, in a pilot study in Labrador Retrievers with copper-associated hepatitis, evaluation of copper/zinc superoxide dismutase 1 and its chaperone show promising results for future clinical use. 9 Research on a microRNA panel is also ongoing and seems to be a promising tool to help diagnose various hepatobiliary diseases in dogs. 10 A combination of noninvasive tests may provide sufficient information to avoid hepatic biopsy in the future.
Other noninvasive approaches are available for the assessment of HF in people. For instance, transient elastography (TE) uses a small transducer on the end of an ultrasound probe that evaluates HF by measuring shear wave velocity. This measurement is then converted into a liver stiffness measurement.8,40 TE was found to be reproducible in healthy dogs. 30 However, no significant correlation was demonstrated between liver stiffness and HF histologic stage in another study (Oudry et al. Evaluation of hepatic diseases in in vivo dogs with Fibroscan device: a feasibility study. Proc 21st ECVIM-CA Congress; Sept 2011; Sevilla, Spain).
Based on our findings, the HF score cannot replace liver biopsies. However, the score developed in our study was designed to help clinicians screen dogs for HF. The diagnostic target was HF histologic stage ≥F2 in order to identify dogs at risk of clinically relevant fibrosis. Further larger cohort studies may allow a more precise cutoff value of the HF score between each histologic stage of fibrosis from F0 to F4.
Single blood biomarkers such as type III procollagen aminopeptide (PIIINP), 15 transforming growth factor β1, 27 and hyaluronic acid14,18 seem to be promising assessors of HF in dogs in some studies. However, in another study, PIIINP and hyaluronic acid were not found to be reliable in assessing HF. 22 In a previous study, median serum interleukin 6 was observed to be twice as high in dogs with low HF scores compared to dogs with high fibrosis score. 29 However, there was substantial overlap between groups, and significance was borderline. 29 Further research on a larger cohort is needed, and analysis of these biomarkers is not widely available in clinical practice. The HF score developed in our study may be improved with the use of new or additional biomarkers. The utility of this HF score in monitoring disease progression and response to treatment should be investigated.
Supplemental Material
DS1_JVDI_10.1177_1040638719866881 – Supplemental material for Use of serum biomarkers in staging of canine hepatic fibrosis
Supplemental material, DS1_JVDI_10.1177_1040638719866881 for Use of serum biomarkers in staging of canine hepatic fibrosis by Maud Menard, Alexis Lecoindre, Jean-Luc Cadoré, Michèle Chevallier, Aurélie Pagnon, Juan Hernandez, Rodolpho Oliveira Leal, Marine Hugonnard, Véronique Miette, Marie Destro, Benoît Rannou, Ghita Benchekroun and Patrick Lecoindre in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the clinical staff members involved in recruiting cases, the referral veterinarians, and the clinicians at the Aquivet Veterinary Clinic (Bordeaux, France).
Declaration of conflicting interests
The authors declare a potential conflict of interest with the company Echosens (Paris, France). Echosens is a vendor of the hepatic fibrosis score (FibroVet Screening) and financially supported the residency of one of the authors (M Menard). Two of the authors are employees of Echosens (V Miette and M Destro).
Funding
The company Echosens provided partial funding for expenses related to some biochemical measurements in the clinical trial.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
