Abstract
A 5-mo-old Bassett Hound–Labrador Retriever cross was autopsied following a bout of lethargy, inappetence, and bleeding gums. Mucous membranes were white, and the small intestine was blue-black; the colon contained black feces. The spleen was swollen, and multiple lymph nodes were enlarged and hemorrhagic. Microscopically, the small intestine had focal crypt cell necrosis and circumferential transmural vasculitis, the latter the cause of infarction and the blue-black coloration. Lymphocytes were necrotic in spleen and lymph nodes, and erythrophagocytosis was present in some nodes. Vasculitis was present in brain, meninges, lung, liver, and kidneys. Electron microscopy revealed aggregates of 15–18 nm round viral particles in damaged crypt cells and in the endothelium of small blood vessels. Electron-dense intracytoplasmic inclusions consisting of paracrystalline-arrayed virus were demonstrated in macrophages in medullary lymph node sinuses. These virions were identified as circovirus, which was confirmed by real-time PCR and sequencing.
In 2013, severe hemorrhagic gastroenteritis and multisystemic vasculitis in a young dog was reported from California. 6 The authors characterized the complete genome of a canine circovirus responsible for the lesions and then retrospectively found 3 additional cases in material retrieved from the Veterinary Medical Teaching Hospital at the University of California, Davis. A year later, a related circovirus was recovered from hemorrhagic enteritis in a young dog in Italy. 2 In 2016, dual circovirus and parvovirus infection was reported in 3 puppies from a breeding kennel in Michigan. 11 Since then, canine circovirus (CaCV) has also been identified in Germany1,4 and Thailand. 8 We document herein a case identical to the sentinel case. 6
In late September 2017, a 5-mo-old black-and-tan castrated male Bassett Hound–Labrador Retriever dog was brought for autopsy, ~12 h after its death. The dog and a female littermate had been rescued in Mississippi several months earlier and held, first at a shelter there, then at a rescue service in San Antonio, before being transferred to Connecticut. The dogs had been vaccinated against canine distemper virus, canine hepatitis virus, parainfluenza virus, parvovirus, coronavirus, Leptospira spp., Bordetella, and rabies virus at both facilities. The dog was castrated in Texas.
Two days prior to death, the dog was lethargic, inappetent, and had bleeding gums. The day before its death, it ate a small amount of dog food, drank an excessive amount of water, looked sick, and became recumbent. At 4 AM on the morning of the third day, the dog was heard to make an agonal sound, and died. Eight days after the death of this dog, its female littermate became ill with diarrhea, thrombocytopenia, and splenomegaly. That dog was treated for ehrlichiosis with doxycycline and recovered.
At the time of autopsy, the dog weighed 9 kg and was in good body condition; however, the oral mucosa and conjunctiva were white. The oral cavity and oropharynx and trachea contained brown ingesta, thought to be from agonal regurgitation. The lungs were mottled dark red, red, pink, and gray, with some emphysema of the left apical lobe. The major findings were in the abdomen, in which the small intestine was distended, and discolored deep blue (Fig. 1). The blue coloration appeared in a variegated pattern, created by numerous oval, dark maroon to blue patches with intervening normal tissue (Fig. 2). The colon was blue-gray.
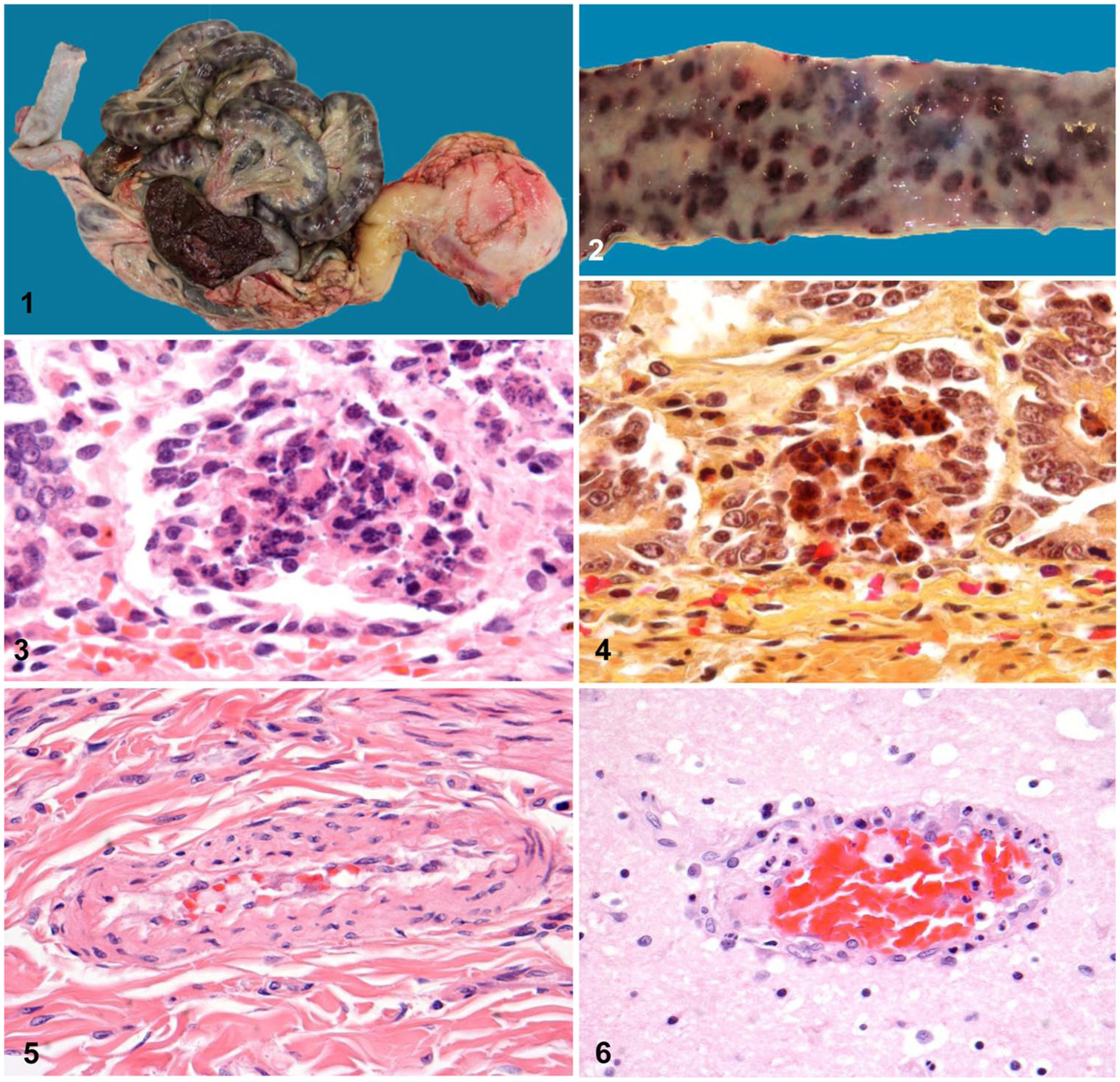
Canine circoviral enteritis.
When the gastrointestinal tract was opened, a dramatic color change was apparent between the mucosa of the stomach and the small intestine. The lesser curvature of the stomach and torus pyloricus were lightly speckled with pinpoint ulcers and the antrum was white, whereas the mucosa of the entire small intestine was variegated maroon (Fig. 2). Mucosal ridges of the colon were streaked with black petechial hemorrhages, and feces were black.
The liver was partially discolored by pseudomelanosis and had multiple flat pinpoint foci in the surface parenchyma. The spleen was swollen with rounded edges and was dark red to maroon. Lymph nodes throughout the body were enlarged and had hemorrhagic cortices; these included submandibular, cervical, tracheobronchial, hepatic, mesenteric, and internal iliac nodes.
Representative tissues were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin. Other sections were subjected to immunohistochemistry for canine parvovirus, coronavirus, and canine distemper. Other stains included Lendrum phloxine–tartrazine and Shorr–Page–Green for viral inclusion bodies, Twort Gram stain for bacteria, trichrome, and phosphotungstic acid–hematoxylin (PTAH).
For electron microscopy, tissue samples previously fixed in formalin were cut into 1-mm2 blocks and placed in 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer. Additional fixation was done in 1% osmium tetroxide. Tissue was dehydrated through a series of graded alcohols and embedded in Glauert resin containing Embed-812 (Electron Microscopy Sciences, Hatfield, PA). Ultrathin (~70 nm) sections were cut with a diamond knife, decompressed with trichloroethylene, and collected on 100-mesh copper grids. Sections were stained with 2% aqueous uranyl acetate, counterstained with 2.5% Sato lead citrate, and images were obtained (Bright-field FEI Tecnai Spirit G2 Biotwin transmission electron microscope; Thermo Fisher Scientific, Eindhoven, The Netherlands).
Microscopically, the major lesions in the gastrointestinal tract were limited to the small intestine. Foci of crypt cell necrosis (Figs. 3, 4) were scattered randomly around the circumference in every segment of the small intestine examined. Additionally, there was focal hemorrhage of the deep lamina propria, just above the muscularis mucosae and at mid-level submucosa. These foci of hemorrhage were centered on small arterioles and did not coincide with the distribution of crypt cell necrosis. The vascular changes consisted of swelling and sloughing of endothelial cells, disrupting the continuity of the endothelium. Often only 3 or 4 rounded endothelial cells remained around the circumference, exposing the basement membrane. Vasculitis was focally distributed among small arterioles of the basal mucosa and submucosa throughout the small intestine, and only rarely accompanied by thrombi. Hyaline degeneration (Fig. 5) and fibrinoid necrosis of the media of affected small vessels were only discernable occasionally. Villi and lymphatics of the intestine were unaffected.
The stomach had only focal mucosal hemorrhage, whereas the ileum, cecum, and colon had necrosis of lymphocytes at the centers of submucosal lymphoid follicles, including those of Peyer patches. Centers of some of the damaged lymphoid follicles were hemorrhagic. Vasculitis and microhemorrhages were present in the colon, but were much less severe than in the small intestine.
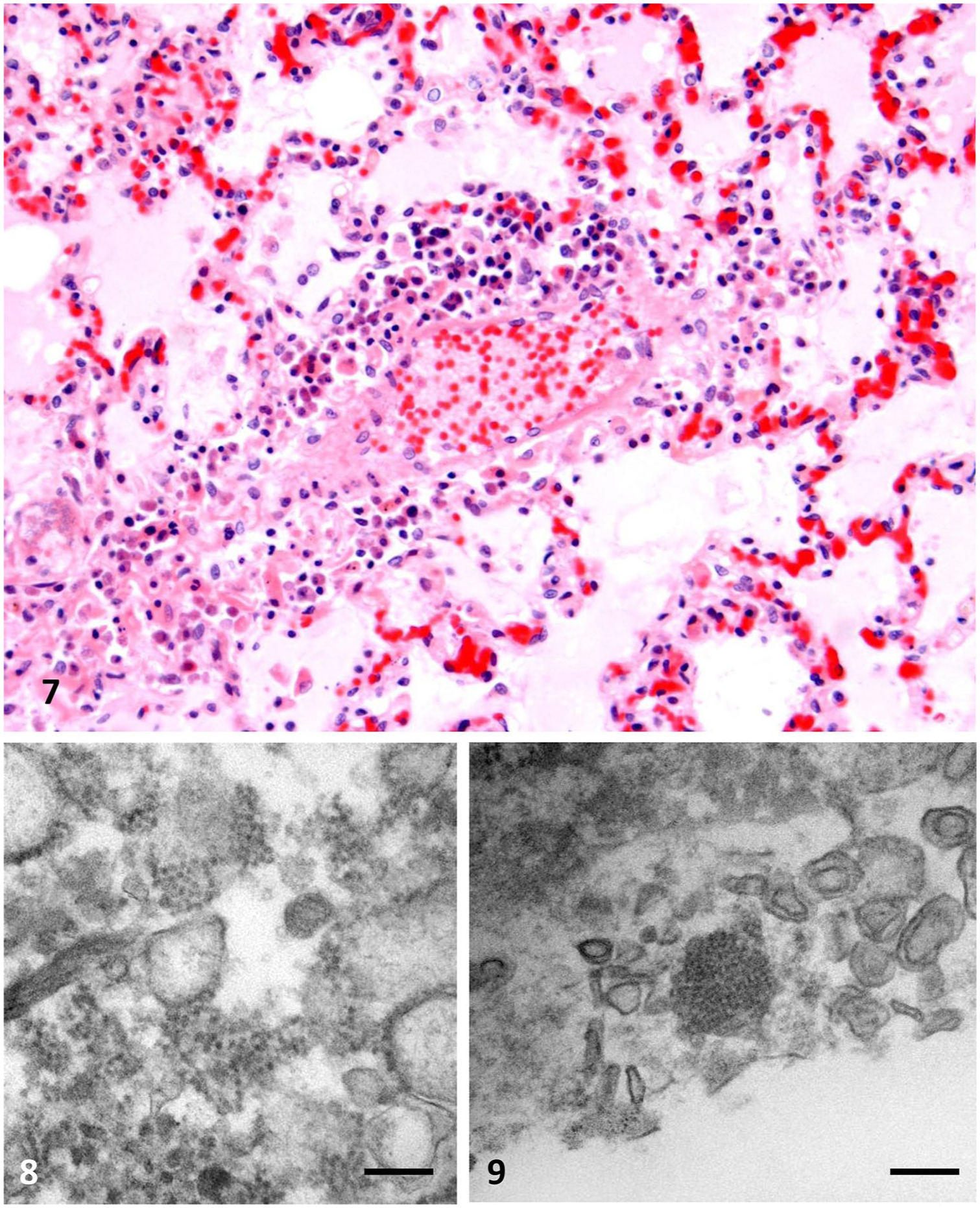
Severe focal vasculitis occurred in the brain, meninges, myocardium, lung, liver, and kidney (Figs. 6, 7). In the liver, the distribution was both central and periportal, creating a variable pattern of necrosis. In the kidney, arterioles within the cortex were affected. In each of these tissues, vasculitis was accompanied by mononuclear cell perivascular inflammation, often eccentric, and consisting of plasma cells and histiocytes. In the meninges, this constituted mononuclear cell meningitis. In the spleen and lymph nodes, there was marked lymphocyte necrosis, hemorrhagic in the latter, some accompanied by erythrophagocytosis. The nodes had marked sinus histiocytosis, and the bone marrow lacked megakaryocytes.
Canine circoviral enteritis.
At the electron microscopic level, enlarged damaged crypt cells with granular, globular contents were found to contain variably sized aggregates of 15–18 nm round virus particles. These were sometimes arranged in rows (Fig. 8). The virions were uniform in size with dense centers and lighter collars. Inclusion bodies were rare. Endothelial cells of affected vessels were rounded or sloughed. Electron microscopy of macrophages of an affected lymph node identified electron-dense inclusions composed of virus particles arranged in paracrystalline arrays (Fig 9).
Immunohistochemistry for parvovirus, coronavirus, and canine distemper virus was negative. The Lendrum and Shorr stains for virus inclusions only rarely disclosed small round, intracytoplasmic inclusions. The Gram stain revealed postmortem bacteria in some tissues. Trichrome and PTAH revealed only rare thrombi in damaged vessels.
To identify CaCV in tissues, we performed a TaqMan real-time PCR reaction following methods and using primers and probe described previously. 6 DNA was extracted from intestine, liver, and spleen (DNeasy extraction kit; Qiagen, Germantown, MD) following the manufacturer’s instructions. The PCR reaction mixture contained TaqMan Fast advanced master mix (Applied Biosystems, Austin, Texas), forward primer (5’-CTGTTGTGAAACTGAAAGAGACGA-3’), reverse primer (5’-TGACGTAGGTCTCCGGATACG-3’), FAM-AGCCTTGCCGCTGTCGCGTC-BHQ1 probe for CaCV-2 capsid, DNA template, and nuclease-free water. The reaction was run (CFX96 C1000 real time system; Bio-Rad, Hercules, CA) at 95°C for 5 min, followed by 40 cycles at 95°C for 15 s, and 60°C for 1 min. The DNA extracted from intestine, liver, and spleen yielded amplicons of the predicted size. Additionally, DNA extracted from the spleen was also amplified with the same CaCV capsid primers by conventional PCR, and the amplicon (68 bases) was separated in, and extracted from, agarose gel, and subjected to Sanger sequencing. The sequence in this region was identical to CaCV strain OH19098-1 (GenBank MF457592.1).
Severe hemorrhagic canine circoviral disease has not been reported previously in New England, to our knowledge. We do not know if the virus was acquired in Mississippi (where the dog lived for 53 d), in Texas (for ~3 mo), or in Connecticut (33 d), and why, albeit rare, this condition is reported in shelter dogs. Prevalence studies, employing molecular methods on feces, have shown that CaCV was carried by 6.9% of healthy dogs in California (n = 204), 6 4.6% in Germany (n = 66), 1 11.9% in Taiwan (n = 19), 5 and 28.5% in Italy (n = 67). 3
The crypt cell necrosis is reminiscent of coronaviral crypt damage in winter dysentery of cattle and coronaviral enteritis of calves. 13 The blue discoloration of the intestine was a very striking gross autopsy finding that has been reported in porcine circovirus 2 (PCV-2) disease of swine. 7 At the time this dog was recumbent and had oral hemorrhage, the owner, a nurse, reported that the dog had the odor of a “GI bleed.” She had recognized the characteristic odor, a finding consistent with the appearance of the intestinal contents at autopsy. A similar odor of a GI bleed occurs in winter dysentery of cattle, in which the feces are similar. 12 The gastrointestinal changes in this disease belie the very severe damage that occurs as a consequence of virus-induced vasculitis in many other organ systems. The viruses and viral inclusion bodies we demonstrate herein are identical to those described in the California dog 6 and to those of PCV in infected PK-15 cells, 10 and in the lymph nodes of pigs with postweaning multisystemic wasting syndrome. 9 Although there is some controversy regarding the need for a co-pathogen in canine circoviral gastroenteritis, 3 we saw no evidence of participation by other viruses in our case.
Footnotes
Acknowledgements
We thank Denise Long for histology and special stains, and Maritza Abril for technical support with electron microscopy. We thank Dr. Cara Spagnoletti of Cromwell, CT for clinical details, Dr. Guillermo Risatti for help with the sequencing of the amplicon, and Dr. Mazhar Khan for help with several images. Jeremy Doucette helped with manuscript preparation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
