Abstract
Baccharis pteronioides DC has been intermittently associated with livestock poisoning in the southwestern United States. In 2004, nearly 100 cows were reported poisoned by B. pteronioides in southern New Mexico. Initial field studies and postmortem examinations found drought conditions, evidence of B. pteronioides consumption, and a reported mortality of nearly 40%. Because postmortem materials were unsuitable for further examination, plant samples were collected for feeding trials and chemical evaluation. Forty-eight Syrian hamsters (8 weeks old) were randomly divided into 4 groups and dosed with 0, 50, 100, and 200 mg of B. pteronioides for 10 days. After dosing, the hamsters were necropsied; sera were analyzed biochemically; and tissues were collected and evaluated histologically. The hamsters treated with 200 mg and several of the 100-mg animals developed anorexia and diarrhea. These animals developed multiple hemorrhagic infarcts in the liver and kidney, with severe hemorrhagic enteritis. Histologically, the higher-dosed animals had severe necrotizing vasculitis with vascular thrombosis of hepatic and renal vessels. Many glomerular capillaries contained fibrin thrombi. The superficial intestinal and colonic mucosa was necrotic, with extensive hemorrhage and proliferation of luminal bacteria. Lower-dosed animals had mild hepatocellular swelling, with proliferation of intestinal and gastric bacteria and yeast. The findings indicate that at high doses, B. pteronioides is toxic to hamsters and produces lesions that are very similar to bacterial endotoxin–produced vasculitis and infarction. Research to purify and identify the toxin, the toxic dose, and mechanism of toxicity are ongoing.
Introduction
Baccharis spp. (family Asteraceae) are native American plants with more than 400 species and varieties in both North and South America and with 24 species in North America. 1 Many Baccharis species are used for medicinal purposes for treating aliments ranging from inflammatory disease to neoplasia. 9 An assortment of compounds, including diterpenic lactones, sesquiterpenes, flavonoids, saponins, tannins, phenolic compounds, and essential oils, have been isolated and described from many Baccharis species. 12 Some compounds and various extracts have been tested for pharmaceutical activity. 1,9,15,23 However, relatively few Baccharis species have been associated with livestock poisoning.
In South America, Baccharis cordifolia commonly poisons livestock in southern Brazil, Uruguay, Argentina, and Paraguay. 5,6,26 Experimental poisoning has also been described in cattle, sheep, horses, rabbits, and mice. 4,7,21,22,24,25,29 Signs of poisoning are variable and include anorexia, diarrhea, constipation, muscular tremors, tachypnea, tachycardia, recumbency, and death. The associated lesions are more consistent and include reddening, edema, and erosions of the stomach, with hemorrhagic gastroenteritis and widespread lymphoid necrosis. 6,21,25,29 All parts of the plant can be toxic, with higher toxin concentrations in the flowers and seeds. 15 Preliminary studies suggest there are multiple toxins, including roridin, myotoxin, miophitocen, and verrucarin. 2,15,16 Although less frequent, similar toxicity, lesions, and toxins have been identified in Brazilian Baccharis megapotamica. 8,14
In North America, Baccharis pteronioides DC (commonly known as yerba de pasmo) is common in the deserts of the southwest United States. It is a small, branched shrub 1–4 m tall. The leaves are small (0.5–2 cm), alternate, drought deciduous, variable in shape, and dark green with a sticky bottom. The flowers are dioecious, creamy white, and found in 0.5-cm racemes that bloom throughout the summer. The fruit is a small achene tipped with a feathery white bristle that ripens in the fall (Fig. 1). Baccharis pteronioides grows in clusters in gravelly soil in Texas, New Mexico, Arizona, and northern Mexico. 18 As early as 1920, North American B. pteronioides was associated with livestock poisoning, and it was reported that the lethal dose for sheep was “near one pound.” 20 The 1920 report speculated that poisoning was related to lack of alternative forage and that stockmen should learn to recognize the plant and avoid exposure when forage is limited. Other than describing the intestinal lesions as “burned with potash,” there are no good descriptions of the gross or histologic lesions. Subsequent to the 1920 report, poisonings have been sporadic. 19,20 No information is available concerning the toxin, nor are there any comparisons of this species with other toxic Baccharis species.

Baccharis spp., a multibranched shrub 1–4 m tall with an alternate, simple, and elliptic to ovate leaf. Inset, small, variably shaped leaves.

Hemorrhagic infarct in the liver of a hamster treated with 200 mg of finely ground Baccharis pteronioides for 8 days.

Photomicrograph of the liver of a hamster treated with 200 mg of finely ground Baccharis pteronioides for 8 days. Notice the vascular thrombosis (arrow) and hemorrhagic hepatic infarct with chronic inflammation and foreign body giant cells. Hematoxylin and eosin. Bar = 50 μm.

Photomicrograph of a mesenteric lymph node of a hamster treated with 200 mg of finely ground Baccharis pteronioides for 8 days. Notice the extensive lymphoid necrosis, edema, and acute inflammation. Hematoxylin and eosin. Bar = 50 μm.

Photomicrograph of the kidney of a hamster treated with 200 mg of finely ground Baccharis pteronioides for 8 days. Notice the fibrin thrombi (arrow) in many glomerular capillaries. Hematoxylin and eosin. Bar = 20 μm.
In 2004, a herd of more than 100 longhorn cattle were pastured on a range in southern New Mexico that contained moderate amounts of B. pteronioides. There was a longstanding drought in the area, and few forage alternatives were available. About 80 animals became sick, anorexic, and struggled to stand; more than 40 died. Postmortem examinations performed several days later found tissues that were markedly autolytic with hemorrhagic enteritis, suggestive of a clostridial infection. As a precaution, the entire herd was revaccinated with multivalent clostridial vaccine. No postmortem samples were suitable or collected for histologic evaluation. Over the next few days, it rained, and alternative forages became available. The affected animals recovered, with no apparent residual signs of poisoning. Field investigations found that significant amounts of B. pteronioides had been eaten, but at the time of observation the livestock were eating newly abundant warm-season grasses. About 100 kg of the complete B. pteronioides was collected and frozen for future feeding trials and analysis. The purpose of the current study was to develop a small animal model of B. pteronioides toxicity, describe the clinical, gross, and histologic lesions of poisoning, and initiate chemical studies to identify the toxin and characterize and describe its toxicity.
Materials and methods
Nonflowering B. pteronioides (Intermountain Herbarium voucher no. 2828; Utah State University, Logan, UT) was collected from southern New Mexico and frozen. The leaves were stripped from the branches, freeze dried, finely ground (to pass through a 1-mm screen), thoroughly mixed, and stored at −4°C until used.
All animal work was done with the approval and under the direction of the Utah State University Institutional Animal Care and Use Committee protocol no. 1205. Forty-eight Syrian hamsters (8 weeks old) were randomly divided into 4 groups. The hamsters were housed in groups of 5 in polycarbonate cages and given food and water ad libitum. For 10 days, hamsters from 3 of the 4 groups were dosed twice daily (half dose) by oral gavage with 50, 100, and 200 mg of B. pteronioides mixed with 2 ml of peanut oil. The last group was dosed with 200 mg of finely ground alfalfa mixed with similar volumes of peanut oil as a control. All were observed every 6 hr for clinical signs of toxicity, and body weights were recorded. After 10 days of treatment, the hamsters were killed, serum was collected by cardiac puncture, and tissues were collected for histologic evaluation.
Histologic sections were prepared and stained with hematoxylin and eosin using standard methods. 3 Special stains, including periodic acid–Schiff (PAS), Gram stain, a modified Grocott methenamine silver stain, b and acid-fast stain, a were done on selected hepatic sections following the manufacturer's instructions and described techniques. 3 To better characterize the renal lesion, the kidneys were stained with Congo red and Mallory phosphotungstic acid hematoxylin (PTAH) following published techniques. 3
Serum was analyzed for sodium, potassium, chloride, carbon dioxide, anion gap, glucose, blood urea nitrogen, creatinine, calcium, magnesium, phosphorus, total protein, albumin, cholesterol, total bilirubin, alkaline phosphatase, lactate dehydrogenase, alanine aminotransferase, and aspartate aminotransferase. Mean animal weights and these biochemical parameters were compared using an analysis of variance. Significantly different means (P < 0.05) were separated using Duncan's method.c
Results
All but one hamster dosed with 200 mg developed diarrhea, became reluctant to move, and stopped eating. Because of this, the 200-mg group was necropsied after 8 days of dosing. The 200-mg group lost weight, but there were no significant differences in weight between the other groups and controls (Table 1). The 200-mg group also had significant biochemical changes. Serum cholesterol, total bilirubin, alkaline phosphatase, lactate dehydrogenase, alanine aminotransferase, and aspartate aminotransferase were all higher (P < 0.05) than controls. The 100-mg group tended to have higher lactate dehydrogenase, alanine aminotransferase, and aspartate aminotransferase activity, but the increased activities were variable and much less severe.
At necropsy, all but one of the highest-dosed animals had multiple, variably sized hemorrhagic infarctions in the liver (Fig. 2). The kidneys were enlarged and red, and the intestine and colon were red and distended with gas. The group treated with 100 mg of plant had dilated and gas-filled intestines. No gross lesions were identified in the animals in the 50-mg group or the alfalfa-treated control group.
In all but two of the highest-dosed hamsters, the stomach and intestine were dilated and filled with debris histologically containing numerous bacteria and yeast. The intestinal mucosa was multifocally necrotic, with extensive hemorrhage. The ileum and colon were filled with numerous bacteria, and there was goblet cell depletion. The mesenteric vessels were congested, and there was extensive necrosis of the mucosal lymphoid tissue. The hepatocytes were swollen with open, enlarged nuclei and abundant nuclear pseudoinclusions. Many hepatic vessels were dilated, with multifocal vasculitis, fibrinous vascular degeneration, and fibrin thrombi. Scattered randomly in multiple hepatic lobules were large zones of hemorrhagic and coagulative necrosis (infarctions; Fig. 3). These infarcts were surrounded by fibrin and infiltrates of neutrophils, with smaller numbers of macrophages, multinucleated giant cells, and lymphocytes. Although many inflammatory cells contained PAS-positive debris, no microorganisms were identified. Vasculitis and thrombosis similar to those seen in the liver were also present in small numbers of splenic and mesenteric vessels. There was multifocal necrosis of lymphoid tissue in many of the follicles in the spleen and mesenteric lymph nodes (Fig. 4). The kidneys of many of the highest-dosed hamsters were congested, and many of the glomerular capillaries were dilated and contained fibrin thrombi (PTAH positive and Congo red negative; Fig. 5). There was also multifocal coagulative necrosis of the proximal convoluted tubules. Two affected hamsters had several fibrinous thrombi in small bronchial arteries, and there was extensive peribronchial edema. The 2 hamsters that did not develop hepatic infarcts were less severely affected, with diffuse, central, lobular, hepatocellular swelling.
Clinical and serum biochemical findings in hamsters that were dosed with freeze-dried, finely ground Baccharis pteronioides for 10 days.*
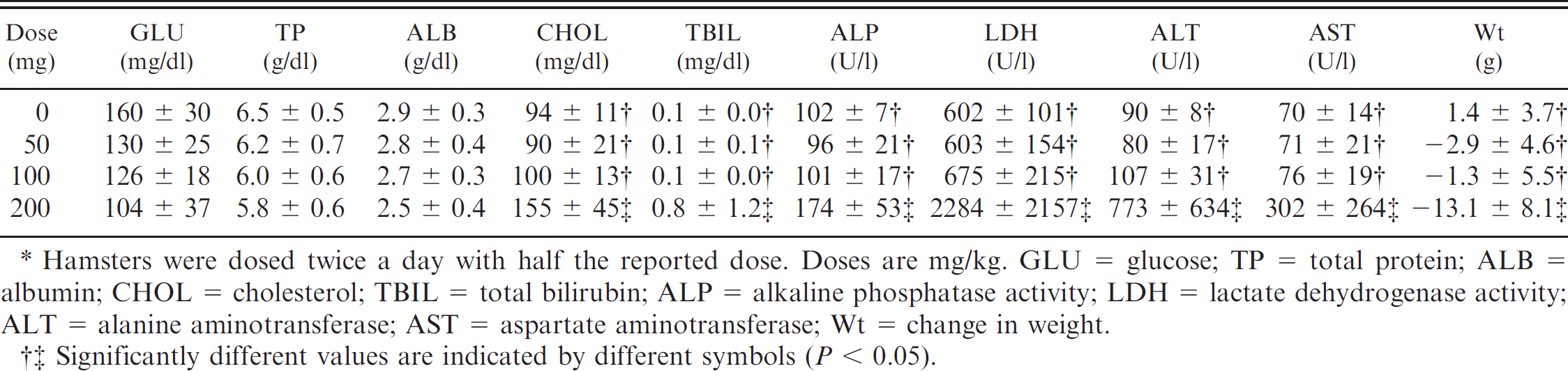
Hamsters were dosed twice a day with half the reported dose. Doses are mg/kg. GLU = glucose; TP = total protein; ALB = albumin; CHOL = cholesterol; TBIL = total bilirubin; ALP = alkaline phosphatase activity; LDH = lactate dehydrogenase activity; ALT = alanine aminotransferase; AST = aspartate aminotransferase; Wt = change in weight.
Significantly different values are indicated by different symbols (P < 0.05).
The hamsters dosed with 100 mg of B. pteronioides exhibited prominent, centrilobular, hepatocellular swelling. The swollen hepatocytes had vacuolated cytoplasm with prominent nuclear pseudoinclusions. The gastric mucosa often contained numerous yeast organisms. No significant histologic lesions were identified in the controls or hamsters treated with 50 mg.
Discussion
The clinical signs and serum biochemical and enzymatic changes were consistent with the histologic changes present in the hamsters. Treated animals developed extensive hepatocellular swelling and degeneration that progressed to vasculitis and hemorrhagic infarction in the highest-dosed group. Because the low- and medium-dosed animals were similar to controls, the biochemical changes were dependent on the development of the extensive vasculitis and infarctions that characterized the 200 mg/day group. These hepatic and vascular changes may well explain the clinical and reported gross findings in the affected cattle from New Mexico. Clostridial infections, especially those that produce vascular α and β toxins (Clostridium novyi, or black disease) resulting in hepatic vasculitis, thrombosis, and infarction, could easily be confused with Baccharis-induced disease. 17
Brazilian B. cordifolia and B. megapotamica poisoning in livestock produce necrosis of the gastrointestinal mucosa and lymphoid tissues. 5,8,27,29 Similar necrotic lesions involving secondary lymphoid germinal centers, the lymph nodes, spleen, intestine, and thymus have been described in rodents and rabbits gavaged with ground plant. 13,29 Although less severe, the authors observed similar lesions in many of the highest-dosed hamsters. Figures 3 and 4 demonstrate hemorrhagic gastroenteritis and mild submucosal lymph node necrosis, as observed in these animals. Similar lymphoid necrosis is associated with stress and endogenous corticosteroid release, and recent work has shown that certain trichothecenes exacerbate lymphoid cytotoxicity or apoptosis induced by a variety of toxins and cellular messengers. 28 In the current study, the lymphoid necrosis was only seen in animals with extensive hepatic necrosis and hemorrhagic enteritis. Further work is needed to determine whether B. pteronioides toxins are directly involved in producing lymphoid necrosis in these hamsters.
In the current study, hamsters treated with relatively high doses of B. pteronioides developed extensive vascular lesions. The plant material used in the current study was probably less toxic than the plants that poisoned the cattle in New Mexico. The plants used were collected in early spring in the vegetative stage. One study of B. cordifolia has shown that female plants are most toxic and that the flowering plant is 4 to 8 times more toxic than other plant phenotypes. 29 That study has indicated that the dried plant remains toxic, but that drying significantly reduces its toxicity. 29 If B. pteronioides toxicity is similar, the dried material used in the current study may have been 10 to 20 times less toxic than the flowering plant consumed by the grazing cattle that were reportedly poisoned.
The extensive vasculitis, thrombosis, and infarctions seen in the hamsters in the current study have not been observed in toxicity studies using other Baccharis spp. This may be related to the relatively high doses that were given to these hamsters. Alternatively, B. pteronioides could have a novel vascular toxin that has not yet been identified. It is also possible that the vasculitis is a secondary lesion. Baccharis pteronioides treatment consistently produced extensive proliferation of bacteria and yeast in the stomach and intestine of these hamsters. The vasculitis may be a result of the growth of these bacteria and subsequent endotoxin production. Both α and β toxins associated with C. novyi and Clostridium haemolyticum are related to extensive extraintestinal vascular changes and hepatic infarction. 17 This hypothesis is supported by the finding that some trichothecenes and Baccharis toxins have been shown to alter gastrointestinal motility and function. 10,11
Studies are currently underway to isolate and identify the B. pteronioides toxins. If the toxins are similar to many of the trichothecenes isolated from other Baccharis plants, they are likely to be similar to the roridins, myotoxins, miophytocens, and verrucarol that are produced by the soil fungi, Myrothecium spp., which are absorbed in the root and translocated to the vegetative plant parts. 5,13 Although effects and toxicity of mycotoxins are fairly well understood, such preharvest fungal–plant interactions are relatively unexplored and present new research challenges to explain the conditions and interactions that result in poisonous plant problems.
In summary, clinical and histologic lesions of B. pteronioides poisoning in hamsters that were similar to those reported in previous field cases and feeding trials were reproduced. The findings indicate that at high doses, B. pteronioides is toxic and produces lesions that may be similar to bacterial endotoxemia–produced vasculitis and infarction. Research to purify and identify the toxin, the toxic dose, and mechanism of toxicity is ongoing.
Acknowledgements
The authors thank Ed Knoppel and Joseph Jacobson for their assistance in animal care and laboratory technical expertise, and Drs. Larry D. Stuart, John C. Reagor, and Anthony P. Knight for their constructive review of this manuscript. This research was partially supported by the Utah Agriculture Experiment Station (Utah State University, Logan, UT), and approved as journal paper number 7997.
Footnotes
a.
Accustain, Sigma-Aldrich, St. Louis, MO.
b.
Accustain modified Grocott methenamine silver, Sigma-Aldrich, St. Louis, MO.
