Abstract
Four turkeys from a commercial flock with acutely elevated mortality levels were submitted for postmortem examination and diagnostic workup. No clinical signs had been observed before death. On gross examination, hemorrhage and necrosis were present throughout the intestinal tracts, and the spleens were markedly enlarged and speckled. Microscopically, numerous, large basophilic-to-amphophilic intranuclear inclusion bodies were observed in mononuclear cells of the spleen and the lamina propria of the small intestine. In addition, there were lesions of diffuse villus blunting and necrosis of the small intestine, with large numbers of rod-shaped bacteria adhered to the epithelium and in the intestinal lumen. Hemorrhagic enteritis virus (HEV) infection was confirmed via PCR on the spleen. Clostridium perfringens was demonstrated in the small intestine by anaerobic culture and immunohistochemistry. The C. perfringens isolate was type F by PCR and, to our knowledge, necrotic enteritis in turkeys has not been described in association with C. perfringens type F infection.
Necrotic enteritis (NE) continues to be a serious health problem for the commercial chicken and turkey industry globally, resulting in economic losses as a result of elevated mortality levels, poor feed conversion, decreased weight gain, and treatment costs.3,6,19 The etiologic agent of NE is Clostridium perfringens, a gram-positive anaerobic bacterium that is ubiquitous in the poultry house environment and a common commensal of the avian gastrointestinal tract.14,22 Transmission and spread of the bacterium is via the fecal–oral route, contaminated fomites, and mechanical vectors. 13 Most cases of NE are produced by C. perfringens type G, which encodes alpha (CPA) toxin and NE-like B (NetB) toxin, but several cases have been reported in turkeys and other birds from which this toxinotype was not isolated, suggesting that other types of C. perfringens may be involved in the pathogenesis of NE in poultry. 19
NE prevention strategies focus on improving overall gastrointestinal health, promoting establishment of a healthy gut microbiota, and preventing overgrowth of C. perfringens by understanding and mitigating predisposing factors.1,18 Pathogens that increase susceptibility of turkeys to NE include coccidia, ascarids, and immunosuppressive agents such as HEV. 13 Non-infectious predisposing factors are related to diet, environment, and genetics and can include diets high in cereal grains and fish meal, high stocking density, and high litter moisture content.6,7 Furthermore, the incidence of NE has increased in some “No Antibiotic Ever” production operations and following the regulatory ban on in-feed antibiotics, including anticoccidials such as ionophores, in commercial poultry. 14
Clinical signs of NE in chickens include unexpected deaths, severe depression, diarrhea, ruffled feathers, and decreased feed and water intake.2,14 There are limited published studies on NE in turkeys; publications have focused on the association with coccidiosis in poults and on toxinotyping of C. perfringens strains.9,12,19 Gross lesions of NE in poultry are found primarily in the intestine; however, the liver may also be affected.2,8 In chickens, intestinal lesions occur most often in the jejunum, whereas in turkeys the duodenum is the most frequently affected segment of intestine, although lesions can extend into the colon. 2 Gross intestinal changes observed in both species include friability, distension with green-to-red fluid, pseudomembrane formation, multifocal-to-coalescent ulcers, and hemorrhage. 8 Microscopic findings include extensive mucosal necrosis with hemorrhage. Necrosis starts at the villus tips. Necrotic and viable tissue are clearly demarcated, and inflammatory cells accumulate. Pseudomembrane formation and congestion are noted. Large numbers of gram-positive rods, rarely sporulated, are commonly seen in areas of necrosis.2,8
Immunosuppressive agents such as viruses and mycotoxins can predispose poultry to NE outbreaks. In turkeys, hemorrhagic enteritis virus (HEV; Adenoviridae, Turkey siadenovirus A) is an important immunosuppressive virus. 4 In turkey flocks, HEV infection is observed mainly at 4–12-wk-old, and virulence can be mild to severe. Subclinical-to-acute infection can result in depression, intestinal damage, and hemorrhage, followed by death.5,17 Transmission occurs via the fecal–oral route; the virus persists in the environment and is easily recovered from contaminated litter. 5 Vertical transmission does not occur with HEV, unlike other adenoviruses, such as fowl adenovirus (Aviadenovirus, the cause of inclusion body hepatitis) and egg drop syndrome virus (Duck atadenovirus A). 5
Gross changes of HEV include enlarged mottled spleens, dilated intestine with bloody content, and congested lungs.4,5 Microscopic changes are most evident in the lymphoreticular system and gastrointestinal tract, particularly the spleen and small intestine. In the spleen, changes include white pulp hyperplasia, lymphoid necrosis, apoptosis, and intranuclear inclusion bodies (INIBs) in macrophages and lymphocytes.5,17 Lymphoid depletion may be evident in the bursa and thymus. 5 Intestinal tract lesions are more prominent in the duodenum and include mucosal congestion, villus tip hemorrhage and infiltration of heterophils, and plasma cells in the lamina propria. 5 INIBs may be seen in mononuclear cells of the intestine. 5 Heterophilic infiltration may occur in the intestine, liver, lung, kidney, pancreas, and brain. 5
We describe here the clinical history, gross lesions, and microscopic lesions of concurrent NE and HEV infection in a commercial turkey flock in California. Four, 10-wk-old, female, commercial meat turkeys were submitted to the Turlock branch of the California Animal Health and Food Safety Laboratory System (CAHFS), for autopsy and diagnostic workup. Three live and 1 dead bird were submitted from a single curtain-sided, dirt-floor house of 8,600 turkeys on a grow-out facility with 60,000 turkeys. The client reported sudden, increased mortality in the affected house with no clinical signs observed the day before submission. The birds submitted alive appeared depressed. Euthanasia was performed in a carbon dioxide chamber. Autopsies were performed on all 4 carcasses. The birds were in good nutritional condition and were well-muscled with adequate adipose tissue stores. Diffuse hemorrhage and necrosis were present throughout the small intestine of all birds (Fig. 1). One of the birds submitted alive had hyperemia and petechiation throughout the mucosa of the small intestine (Fig. 2). All spleens were markedly enlarged and speckled (Fig. 3). No other significant gross changes were observed in the carcasses.
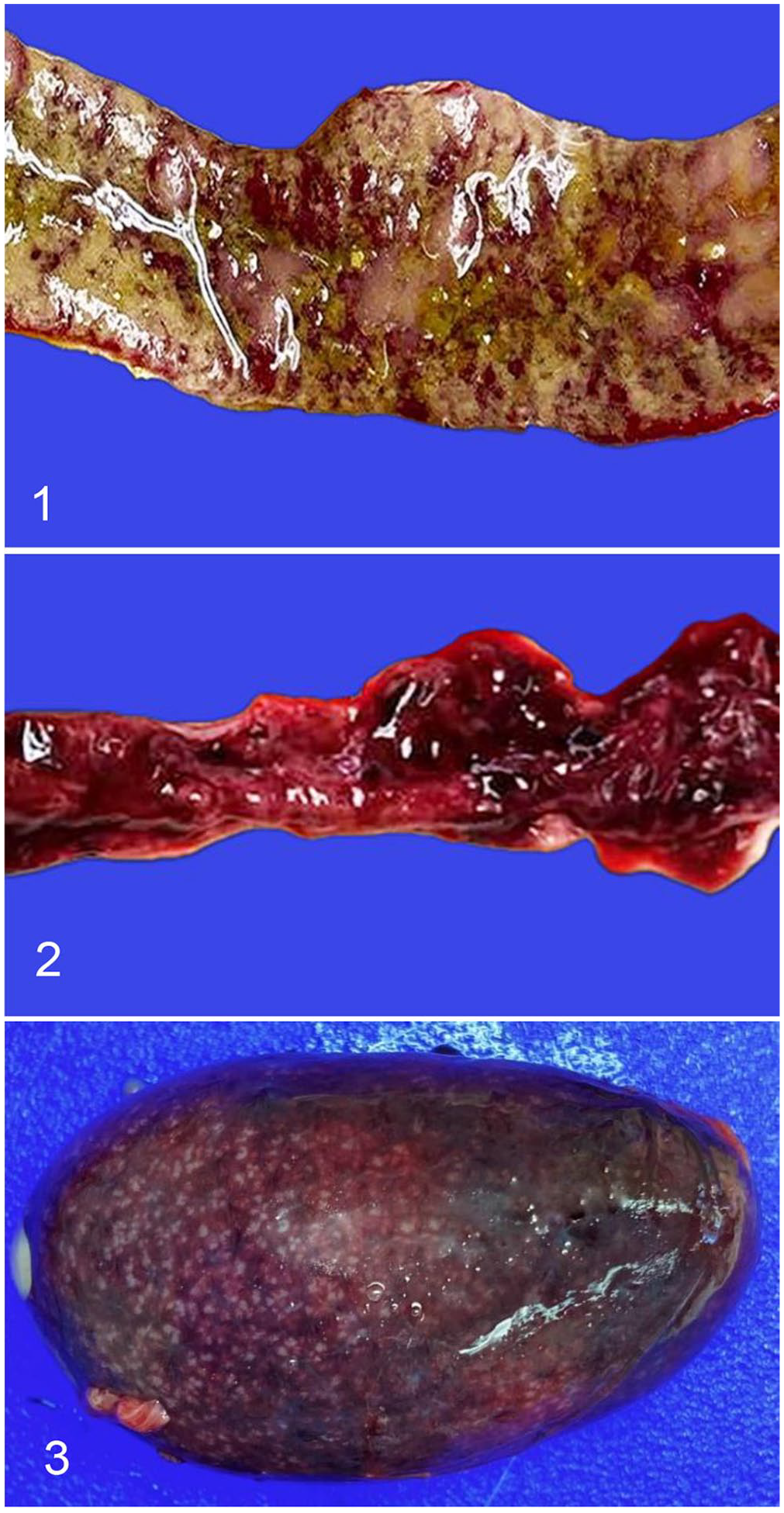
Gross lesions in turkeys with concurrent hemorrhagic enteritis virus infection and Clostridium perfringens type F necrotic enteritis.
Wet mounts of intestinal scrapings were negative for coccidial oocysts from the 4 birds. Samples of spleen, liver, lung, kidney, heart, air sac, proventriculus, intestine, pancreas, trachea, and infraorbital sinus margin were collected from the 4 birds, immersion fixed in 10% neutral-buffered formalin for 24–48 h, paraffin-embedded, and processed routinely, producing 4-µm thick H&E-stained slides.
Microscopically, there was necrosis and proliferation of the splenic white pulp, and numerous lymphoid cells had large, basophilic-to-amphophilic, glassy INIBs and marginated chromatin (Fig. 4). The small intestine had villus blunting, necrosis, and hemorrhage of the lamina propria (Fig. 5). A pseudomembrane of necrotic debris, mixed inflammatory cells, and bacterial colonies was present over the intestinal mucosa. Basophilic INIBs were observed within lymphoid cells in the lamina propria of the villi. Large numbers of gram-positive rods were observed in the intestinal lumen and coating the necrotic villi.
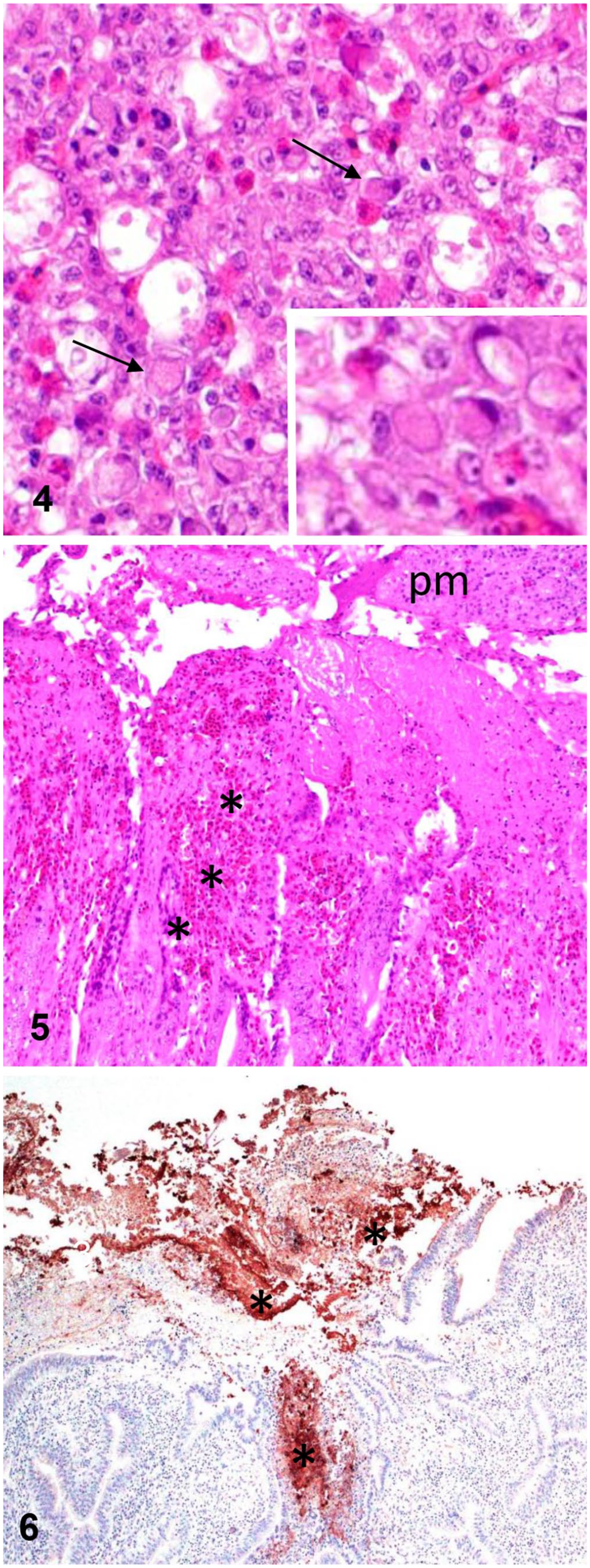
Microscopic changes in turkeys with concurrent hemorrhagic enteritis virus infection and Clostridium perfringens type F necrotic enteritis.
Trachea, liver, spleen, and intestine were swabbed and cultured on MacConkey and 5% sheep blood agar at 37°C and 7% CO2 for 48 h. Intestinal content was plated onto Brucella agar with 5% sheep blood and incubated at 37°C anaerobically. Moderate numbers of bacterial colonies with a double halo of hemolysis were isolated on anaerobic culture, compatible with C. perfringens. Gram staining and conventional biochemical testing and matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry confirmed that the colonies were C. perfringens. 20 The C. perfringens isolate was type F (carrying the cpa and cpe genes) by PCR. 19 Immunohistochemistry for C. perfringens was performed on formalin-fixed, paraffin-embedded sections of intestine, and most of the rods previously described in H&E- and Gram-stained sections showed positive immunoreactivity (Fig. 6). 11
HEV was detected by real-time PCR of pooled spleen tissue samples with a cycle threshold (Ct) value of 12.2. 16 Rapid serologic testing for the detection of HEV antibodies (ProFLOK HEV-T Ab ELISA; Zoetis) was performed on 6 µL of serum from each live bird. No HEV antibodies were detected in any of the birds.
The results of the gross and microscopic examinations and ancillary tests confirmed infection by HEV, and also indicated concurrent C. perfringens NE. C. perfringens type G was not detected. However, cases of NE have been reported in turkeys and other avian species from which C. perfringens type G was not isolated. We retrieved no cases of NE in association with C. perfringens type F in turkeys using the literature search terms “necrotic enteritis; NE; C. perfringens; Clostridium perfringens, type F” in a search of Google, PubMed, CAB Direct, Web of Science, and Scopus, suggesting that no descriptions of this condition have been reported in turkeys; the role of C. perfringens enterotoxin (CPE) in the pathogenesis of NE is unknown.
Concurrent infections of HEV and NE in turkeys may be linked to numerous factors related to the host, the environment, and the pathogens. Moreover, production management practices and stressors influence disease severity and the resulting economic losses. HEV is considered endemic in commercial turkey flocks in the Central Valley of California, and its immunosuppressive effect has been associated with increased opportunistic infections (e.g., by E. coli and Clostridium spp.).7,15 Disease control is based on strict biosecurity, farm cleaning and sanitation, good husbandry practices, and appropriate vaccination. The commercial turkey flock from which our cases were submitted had been vaccinated when ~5-wk-old with a live HEV vaccine (Hygieia Biological Laboratories). This vaccine is derived from a cell-cultured propagated avirulent strain type 2 avian adenovirus in a lyophilized preparation, administered via drinking water and protecting against virulent HEV field strains. 10 Serologic testing of the 3 live turkeys at 10-wk-old was negative for HEV antibody titers. Seroconversion after appropriate application of a live avirulent HEV vaccine is reported to occur within 21 d post-vaccination. 21 Moreover, once the vaccine is administered according to established vaccination standards and the bird’s immune system is stimulated, HEV antibody titer should persist for > 470 d for most turkeys. 15
Possible reasons for vaccine failure and lack of seroconversion in the flock include improper storage and handling of the vaccine, incorrect vaccine administration, high levels of maternal antibodies in the flock, or infection prior to development of adequate vaccinal immunity. Based on lesions observed, our cases were most likely acute HEV infections. Clinical signs, mortality, and lesions are typically observed 5–6 d after infection of a turkey flock with HEV. 15 Our cases are consistent with acute HEV infection; the 12.2 Ct value of the HEV PCR indicates a high viral load.
In our cases, immunosuppression by HEV likely predisposed turkeys to overgrowth of C. perfringens and NE development. A 1995 study of NE in turkeys in California found that 15.3% of turkey autopsy case submissions had clinical NE, and it was hypothesized that mucosal damage caused by HEV predisposed turkeys to NE. 7 In the 1995 study, the disease was observed in commercial turkey flocks 6–11-wk-old, and a clinical NE diagnosis was based on gross and microscopic lesions of hemorrhage and necrosis, observed solely in the intestines. 7
The treatment regimen for the flock described ensured that the welfare of the birds was maintained, as well as implemented steps to reduce financial losses. Flock management included the prompt removal of dead birds, therapeutic use of antibiotics followed by supportive therapy (probiotics, vitamins, electrolytes), as well as litter management, water sanitation, and strict biosecurity.
Footnotes
Acknowledgements
We thank CAHFS technicians for the processing of samples by ELISA, PCR, histotechnology, and immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
