Abstract
We report herein Rhodococcus equi infection in an 11-y-old, male llama with a history of diarrhea and endoparasitism. Postmortem examination revealed granulomatous and ulcerative enteritis, pyogranulomatous mesenteric lymphadenitis, fibrinosuppurative peritonitis, and granulomatous hepatitis. Intralesional macrophages were laden with gram-positive cocci. Bacteriology identified R. equi, and cultures tested positive for R. equi choE and vapA genes by PCR. This case expands the reported spectrum of lesions associated with R. equi infections in llamas from pyogranulomatous bronchopneumonia and peripheral lymphadenitis to pyogranulomatous mesenteric lymphadenitis and enteritis. We also link a R. equi that is carrying the virulent-associated protein gene VapA to clinical disease in New World camelids.
Rhodococcus equi is, typically, a pathogen of foals leading to pyogranulomatous pneumonia with lymphadenitis, and less commonly enterocolitis and intraabdominal abscesses.3,13 Virulence-associated markers of R. equi have been well established. Virulence-associated protein antigens (VapA or VapB) have been implicated in R. equi virulence in a number of host species including horses. 12 In non-equid species, lymphadenitis is the most common presentation. However, R. equi has been linked to a broad spectrum of disease conditions in a wide range of domestic and wild animal species, worldwide. Examples include bronchopneumonia, osteomyelitis, subcutaneous abscesses, and disseminated infection involving lymph nodes and viscera in goats in North America, 15 and bronchopneumonia and lymphadenitis in wild boar in South America, Europe, and Asia. 7 A single case report in 1995 described R. equi infection in a llama as pyogranulomatous bronchopneumonia and pyogranulomatous lymphadenitis of tracheobronchial and mediastinal lymph nodes, and histologic evidence of necrosis associated with bacteria in hepatic and gastric lymph nodes and the spleen. 8 Bronchopneumonia was also reported as the primary presentation in a series of 4 dromedaries with rhodococcosis. 9 Herein we report a case of alimentary rhodococcosis in an adult llama and demonstrate that the causative R. equi carried the gene encoding virulence factor VapA.
An 11-y-old, intact male llama had a history of diarrhea and inability to rise. A heavy gastrointestinal parasite burden had been identified by the referring veterinarian in this and other llamas on the same premises. The llama had been dewormed regularly: twice yearly with ivermectin and with albendazole 3 d prior to presentation at the Lois Bates Acheson Veterinary Teaching Hospital (VTH; Oregon State University [OSU], Corvallis, OR). On presentation at the VTH, the llama had a body condition score (BCS) of 1 of 5, hyperglycemia of 14 mmol/L (reference interval [RI]: 4.9–7.3 mmol/L), a packed cell volume (PCV) of 0.25 L/L (RI: 0.27–0.45 L/L), total solids of 35 g/L (RI: 53–73 g/L) with an albumin of 16 g/L (RI: 53–73 g/L), and mildly decreased electrolytes with hypokalemia of 3.3 mmol/L (RI: 3.8–5.2 mmol/L), hyponatremia of 144 mmol/L (RI: 146–156 mmol/L), hypochloremia of 106 mmol/L (RI: 109–125 mmol/L), hypocalcemia of 1.8 mmol/L (RI: 2.1–2.7 mmol/L), and hypomagnesemia of 0.6 mmol/L (RI: 0.8–2.5 mmol/L). Ketonemia was present, with β-hydroxybutyric acid of 86 mmol/L (RI: 12–72 mmol/L), and mild elevations in aspartate aminotransferase at 14.9 µkat/L (RI: 1.1–3.9 µkat/L) and γ-glutamyl transferase at 1.5 µkat/L (RI: 0.27–0.77 µkat/L), which improved on repeat analysis and were attributed to being down and decreased feed intake, respectively.
Treatment was initiated for suspected wasting and hypoproteinemia as a result of a severe parasite burden. Therapy included fresh frozen plasma; lactated Ringer solution supplemented with hypertonic sodium chloride; flunixin meglumine; oral vitamin E; oral ponazuril; oral sucralfate; and assistance to rise every 4 h. Initial clinical response during the first 6 d of hospitalization included improved ability to stand, increased appetite, and mild improvement of blood chemistry values. On d 7, the llama deteriorated acutely, developing increased heart and respiratory rates, hypothermia, blood lactate of 8.9 mmol/L, acidemia, PCV of 0.35 L/L, and total solids of 28 g/L. Treatment was attempted with administration of 3 units of plasma; however, the llama died a few hours later and was submitted to the Oregon Veterinary Diagnostic Laboratory at OSU for postmortem examination.
On autopsy, the 111-kg, intact male llama was emaciated (BCS 1 of 5) with serous atrophy of pericardial, mesenteric, and femoral bone marrow fat, and generalized muscle wasting. Approximately 10 L of white, cloudy fluid mixed with feed and thick strands of fibrin filled the abdominal cavity. Hemorrhagic streaking was noted on the mesentery and abdominal serosal surfaces. Several accumulations of thick, white, fibrinopurulent material and loose adhesions connected loops of intestine to each other and loops of intestine with the mesentery (Fig. 1). A stronger adhesion between the jejunum and the mesentery was centered on an enlarged string of lymph nodes that contained purulent material when cut (Fig. 2). The adhered jejunum had a single transmural perforation (~5 mm diameter; Fig. 3) ~2 m orad to the ileocecal junction. The jejunal wall around the perforation was necrotic, green-gray, and thin-walled, and was bordered by ulceration with smooth edges. Smaller foci of ulceration were present in the adjacent, markedly expanded, and mildly hyperemic mucosa of the small intestine over a ~15 cm long segment (Fig. 3). The liver had myriad disseminated foci, interpreted as hepatitis. Most foci were gray and pinpoint to 1 mm diameter; fewer foci were dark red-brown, depressed, and 2–4 mm diameter; occasional foci were pale tan, protruding, and up to 3 mm diameter (Fig. 4). One larger nodule (5 mm diameter) was noted near the gall bladder. The spleen was subjectively small. Stomach compartments C1 and C2 had severe submucosal edema. The pericardial sac contained ~250 mL of straw-colored, clear fluid.
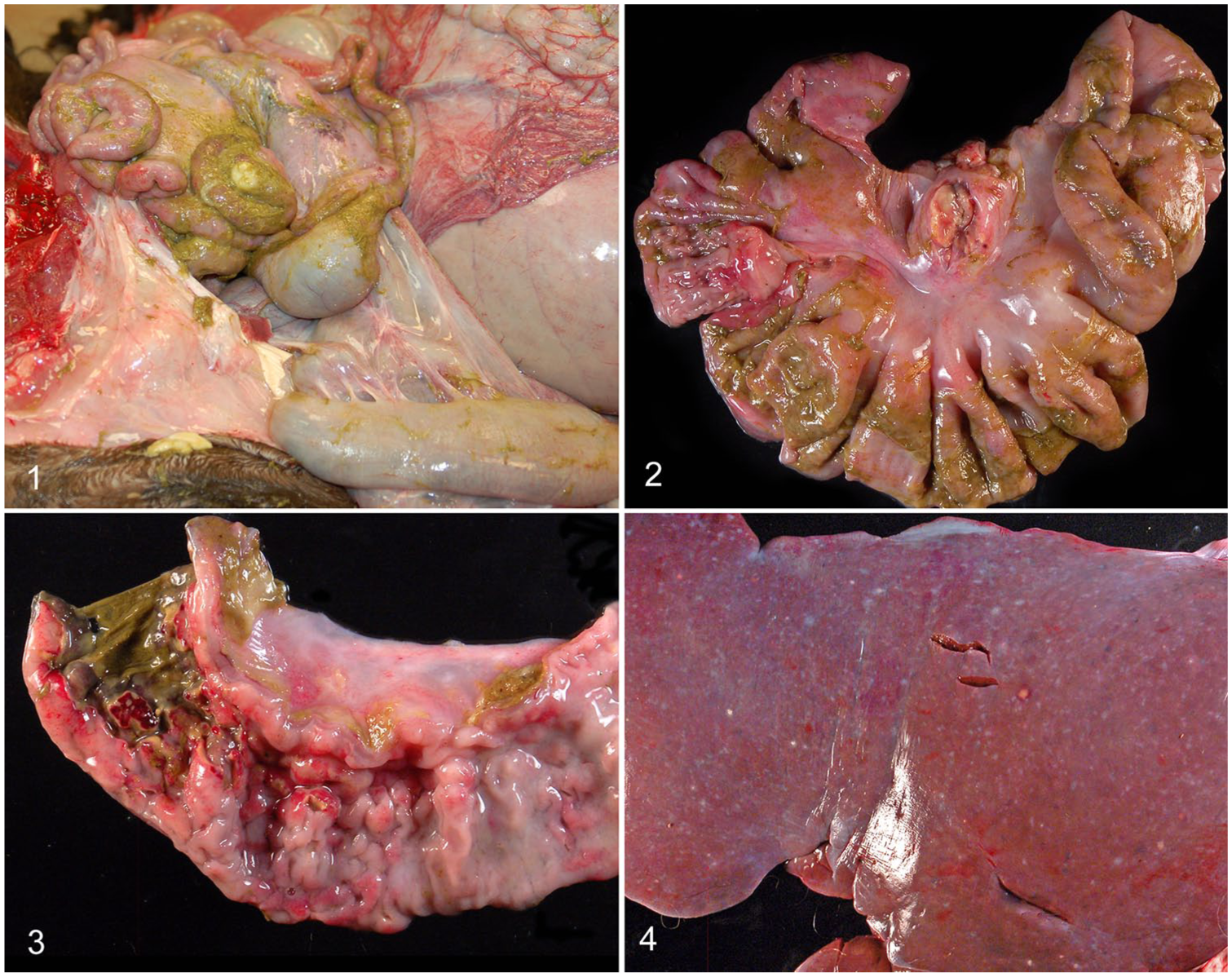
Llama with Rhodococcus equi infection.
An impression smear of the lymph node was Wright-Giemsa–stained and examined by light microscopy. Neutrophils and macrophages with intracellular bacteria were identified. On histology, the jejunum was focally ulcerated, and thick mats of fibrin and cellular debris covered the serosa (Fig. 5). At the site of the perforation and elsewhere, the submucosa was markedly expanded by multifocal-to-coalescing aggregates of macrophages and mature and degenerate neutrophils admixed with fibrin and cellular debris. Necrotic debris frequently contained plant material and was heavily colonized by mixed bacteria, predominantly gram-negative slender bacterial rods. Cytoplasmic, basophilic stippling in many macrophages (Fig. 6) was identified as gram-positive cocci on special-stained recuts (Fig.7). Other segments of small intestine had lymphoplasmacytic-to-necrotizing enteritis characterized by moderate lymphoplasmacytic infiltrates with fewer neutrophils and eosinophils along the base of villi, as well as extending up into the villus lamina propria, and rare crypt necrosis. The architecture of mesenteric lymph nodes was largely effaced by dense accumulations of degenerate neutrophils and macrophages laden with short, gram-positive cocci. The liver had extensive subcapsular hemorrhage. Hepatic lobules and plates were disrupted by random foci of inflammation or areas of hemorrhage and pigment-laden macrophages. The latter rarely contained metazoans with a morphology suggestive of trematodes (Fig. 8). Foci of inflammation comprised neutrophils and macrophages with intracellular bacteria (Fig. 9) that were confirmed as gram-positive cocci. Some portal areas had mild fibrosis or moderate infiltrates of lymphocytes, plasma cells, and rare neutrophils and macrophages.
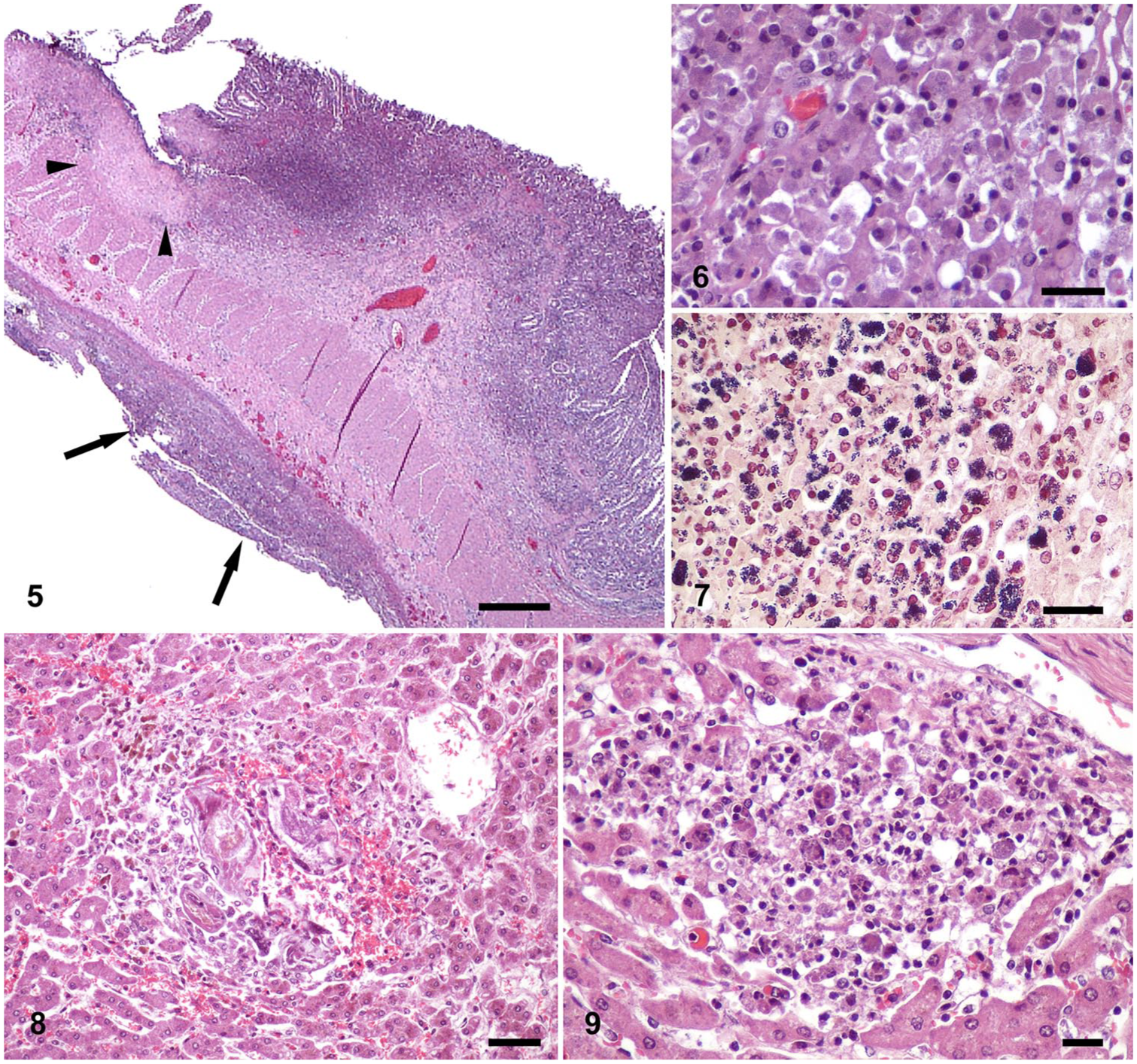
Granulomatous-to-necrotizing jejunitis and hepatitis in a llama.
Other histologic findings included areas of epithelial loss within glandular pits of C1/C2 compartments of the stomach, often associated with macrophages and lymphocytes, and marked submucosal edema. The spleen had severe reduction in both white and red pulp in relation to prominent smooth muscle trabeculae. Fibrin accumulations were present in variable amounts over the serosae of most abdominal organs. No significant abnormalities were evident in the kidney, lung, heart, esophagus, cerebellum, brainstem, and cerebrum.
Routine bacteriology was performed. Large numbers of mixed gram-positive and -negative bacteria were cultured from the necrotic small intestine and lymph nodes, consistent with intestinal flora in aerobic conditions and large numbers of Bacteroides sp. in anaerobic conditions. Large numbers of Escherichia coli grew from the lymph node. Rhodococcosis was suspected based on histopathology, and further examination of cultures confirmed gram-positive diplococci to vertical rods most closely resembling Rhodococcus equi, in moderate growth from both tissues. Real-time PCR tests for the R. equi choE gene and the R. equi virulence gene vapA was performed at the University of Kentucky Veterinary Diagnostic Laboratory (Lexington, KY) and were positive, with Ct values of 29.6 and 25.6, respectively.2,10
The dominant histomorphologic lesions in our case were pyogranulomatous jejunitis, lymphadenitis, and hepatitis with intracellular bacteria. Many bacterial infections cause predominantly neutrophilic inflammation. Some notable exceptions are infections with bacteria such as Mycobacterium sp., R. equi, Corynebacterium pseudotuberculosis, and Yersinia pseudotuberculosis, which have evolved mechanisms to resist or escape clearance. 1 The initial bacteriologic analysis of mesenteric lymph node reported growth of E. coli under aerobic, and Bacteroides sp. under anaerobic, conditions, as well as the mixed flora expected in the intestine. Only closer examination of cultures prompted by histopathology results revealed several colonies of gram-positive diplococci and vertical rods consistent with R. equi. Identification may be further complicated given that concurrent infections with other gram-positive bacterial pathogens have been reported. 9 Cytology of affected lymph nodes can provide a rapid and inexpensive means to establish a preliminary diagnosis of rhodococcosis based on bacterial morphology, and was beneficial in our case.
Virulence-associated markers of R. equi have been well established. High or intermediate virulence-associated protein antigens (VapA or VapB, respectively) have been tightly linked to R. equi virulence in horses and experimentally infected animals. 12 Coding genes are carried on bacterial plasmids. Ten different VapA plasmids have been identified and can be characterized by plasmid profiling. Alternative methods for detection of virulent R. equi include detection of vapA genes by PCR, as performed in our case, or VapA protein (e.g., by Western blot).
The single case report of rhodococcosis in a llama described pyogranulomatous bronchopneumonia and caseous lymphadenitis of tracheobronchial and mediastinal lymph nodes. There was also histologic evidence of necrosis associated with bacteria in hepatic and gastric lymph nodes and the spleen. 8 Neither the lungs nor thoracic lymph nodes were affected in our case, most consistent with a primary infection of the intestinal tract via ingestion. Bronchopneumonia was also reported as the primary presentation in a series of 4 dromedaries, one of which had a single lesion in C3 of the stomach. 9 Lesions in the liver of our llama were a combination of R. equi-containing pyogranulomas and rare foci of necrosis and hepatitis associated with intralesional trematodes, rather than degenerative as was the previously reported hepatic amyloidosis in dromedaries. 9 In foals, extrapulmonary disease caused by R. equi is associated with a worse prognosis than infections confined to the lungs. 13
Of public health interest is the increasing evidence that R. equi can be detected, in the absence of lesions, in the intestines and lymph nodes in animal species regularly consumed by humans including cattle, swine, horses, 17 wild boar (Sus scrofa), and deer. 14 Concern exists about the zoonotic potential of R. equi, 11 and is heightened by the common resistance of the agent to macrolides and rifampicin and the lack of established protocols for treatment of infections by resistant isolates. 4 This is particularly problematic, given that infections in humans occur most commonly in immunocompromised individuals, for example, secondary to other infections, such as HIV/AIDS, or after iatrogenic immunosuppression, such as in transplant recipients. 16 Emaciation and splenic atrophy were noted in our llama, suggesting that reduced immune competence may have contributed to the development of clinical rhodococcosis in this animal. Splenic atrophy was attributed to increased cortisol levels in a stressed animal, and the poor body condition of this camelid.
Serologic testing using ELISA with reference R. equi strain as antigen (ATCC 33701) identified R. equi antibodies in 7% of examined serum samples from alpacas in Italy. 6 Similar to C. pseudotuberculosis, R. equi can be readily identified in the soil on farms with a high incidence of rhodococcosis. 5 Thus, camelids in close association with horses or on pastures where horses have been kept may be at increased risk for developing rhodococcosis. High stocking density and neutral pH may contribute to accumulation of R. equi in the environment, and high environmental temperatures may favor disease development. 3 Studies aimed at identifying specific management factors that would result in control or prevention of R. equi pneumonia in foals have failed to do so. 5 A series of 4 dromedaries with fatal pulmonary rhodococcosis has been reported on a single farm bordering a wildlife sanctuary with zebras among other ungulates. 9 However, widespread outbreaks in New World camelids either by horizontal transmission or via fomites, including contaminated environment, have not been reported to date.
Our case is the second report to detail the lesions of rhodococcosis in a New World camelid, and expands the spectrum of lesions reported with R. equi infections in llamas from pyogranulomatous bronchopneumonia and peripheral lymphadenitis 8 to pyogranulomatous enteritis and mesenteric lymphadenitis. R. equi carrying virulent-associated factor has not been linked previously to clinical disease in New World camelids, to our knowledge. Infection with R. equi should be considered as a potential differential diagnosis in camelids with pyogranulomatous lymphadenitis, pneumonia, gastroenteritis, and abdominal abscesses.
Footnotes
Acknowledgements
We thank the personnel of the bacteriology, pathology, and histology sections of the Oregon Veterinary Diagnostic Laboratory for excellent technical assistance. We thank the laboratory personnel at the University of Kentucky Veterinary Diagnostic Laboratory for competent and expedient molecular testing of bacterial cultures. Dr. Daw was a veterinary student at Oregon State University and worked on this patient during a fourth-year rotation. Dr. Daw is now at the College of Veterinary Medicine, Mississippi State University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
