Abstract
Bovine leukemia virus (BLV) infection has worldwide distribution in both dairy and beef herds. Our study was initiated in order to encourage control of BLV infection by using milk samples, in lieu of serum samples, to readily test lactating animals prior to dry-off and calving. Two Holstein dairy herds (A and B), with known status of BLV infection as determined by serology, were sampled by the collection of serum and fresh milk samples. Selected samples were tested using a USDA-licensed BLV antibody ELISA kit (Bovine leukemia virus antibody test kit; VMRD, Pullman, WA) for serum. Forty-one lactating cows from each herd were sampled. Herd A was confirmed to have endemic BLV infection; herd B was confirmed to be free of BLV infection. The milk ELISA results demonstrated 100% identification of positive and negative animals compared with the serum results. The correlation of the ELISA values between serum and milk samples was 97%, which supports the use of this BLV ELISA on milk samples.
Bovine leukemia virus (BLV; family Retroviridae, subfamily Orthoretrovirinae, genus Deltaretrovirus) has worldwide distribution in domesticated cattle populations.12,25 BLV was initially reported in 1969 as an infectious cause of lymphosarcoma in dairy cattle, with later reports of its occurrence in beef cattle as well.12,20 Following the propagation of BLV in cell culture, several laboratory assays were developed, with the agar gel immunodiffusion (AGID) serologic assay being the most widely used.8,21 Although the AGID assay has been instrumental in supporting BLV infection control efforts, other assays, such as serum ELISA and PCR have been used for increased sensitivity and earlier detection of infection.9,17,20,22,29 Since ~2000, there has been a concerted effort to encourage BLV testing in the United States in order to reduce the level of BLV infection in selected cattle populations.4,12,23–25 This interest has expanded to consider the potential of eradication of BLV infection from the broader populations of dairy cattle within the United States and Canada.1,2,7,10,23,25 Given that BLV infection can occur as a covert (subclinical) infection in a high percentage (≥90%) of the dairy cattle population, it is essential to have a very accurate assay for detection of subclinical infections for preemptive management decisions.1,13,16,22,29 Such decisions may include culling of BLV-seropositive cattle; early weaning of calves from BLV-seropositive dams; and possible use of freezing or pasteurization of colostrum to decrease the milk-associated spread of BLV to offspring.3,5,12,14,26
Milk ELISA for BLV antibody detection has been used for monitoring herds that have successfully eradicated BLV infection in various countries, 15 and for determining the progress of control programs in regions where BLV infection is still endemic. 11 Milk is a sample readily available for testing independent of the use of needles and blood collection, and milk can be obtained from any lactating cow prior to dry-off and calving.5,6,7,15 This is important because colostrum and milk are considered the major natural sources of BLV spread on the farm.3,12,14,16 Although the United States has yet to embark upon a total BLV eradication program, there is interest in regional control efforts leading to BLV infection–free populations.6,7,18,21,28 We therefore evaluated the application of the sole U.S. Department of Agriculture (USDA)-licensed BLV serum ELISA using milk samples.
We sampled 41 lactating Holstein cattle (2–10 y old) from each of 2 dairy herds in the northwestern United States. Our study was approved by the WSU Institutional Animal Care and Use Committee and conducted under ASAF 4889. The herds were identified as herd A (n = 110, milking and dry cows) and herd B (n = 175, milking and dry cows) for confidentiality. Herd A was known to be endemically infected by BLV based on prior herd serology tests (serum ELISA, data not shown). Herd B was known to be negative for BLV infection based on prior herd serology tests (serum ELISA, data not shown). We collected 2 red-top Vacutainer tubes (BD, Franklin Lakes, NJ) of blood and 2 sterile 50-mL, screw-top centrifuge tubes of milk from one-quarter of each animal. All samples were collected aseptically and refrigerated until processed. Serum was poured off blood clots after centrifuging the blood tube at 1,400 × g for 10 min. Whey was pipetted from under the visible lipid layer of milk after centrifuging at 2,000 × g for 15 min. Serum samples were diluted 1:25 in sample diluting buffer and tested according to the test kit (Bovine leukemia virus antibody test kit, cat. 284.2; VMRD, Pullman, WA). Whey samples were diluted 1:2 in sample diluting buffer and tested similarly to the serum. Samples were considered BLV antibody positive if the optical density (OD) was equal to or greater than the average of the positive control, per the kit insert. The manufacturer’s reported sensitivity to the USDA of the BLV ELISA on serum was 98%; specificity was 100% based upon a selected serum sample set.
Initially, 11 lactating cows from each herd were randomly chosen and sampled for proof of concept, and tested as described. All 11 cows from herd A were BLV antibody positive; all 11 cows from herd B were BLV antibody negative as determined by both serum and milk ELISA. The OD correlation was 99.2%. Statistical analysis was performed to determine the OD correlation (Excel 2013 data analysis toolpak; Microsoft, Redmond, WA). Thirty lactating cows were then selected at random from each of the herds and tested as described for BLV antibodies. The 30 cows in herd A were all BLV antibody positive, and the 30 cows in herd B were all BLV antibody negative, in the serum and milk ELISAs. The OD correlation for the 2 groups of 30 cows was 96.7%. There was good relationship between the serum and milk ELISA results, with correlation of ODs of 97.3% (Fig. 1).
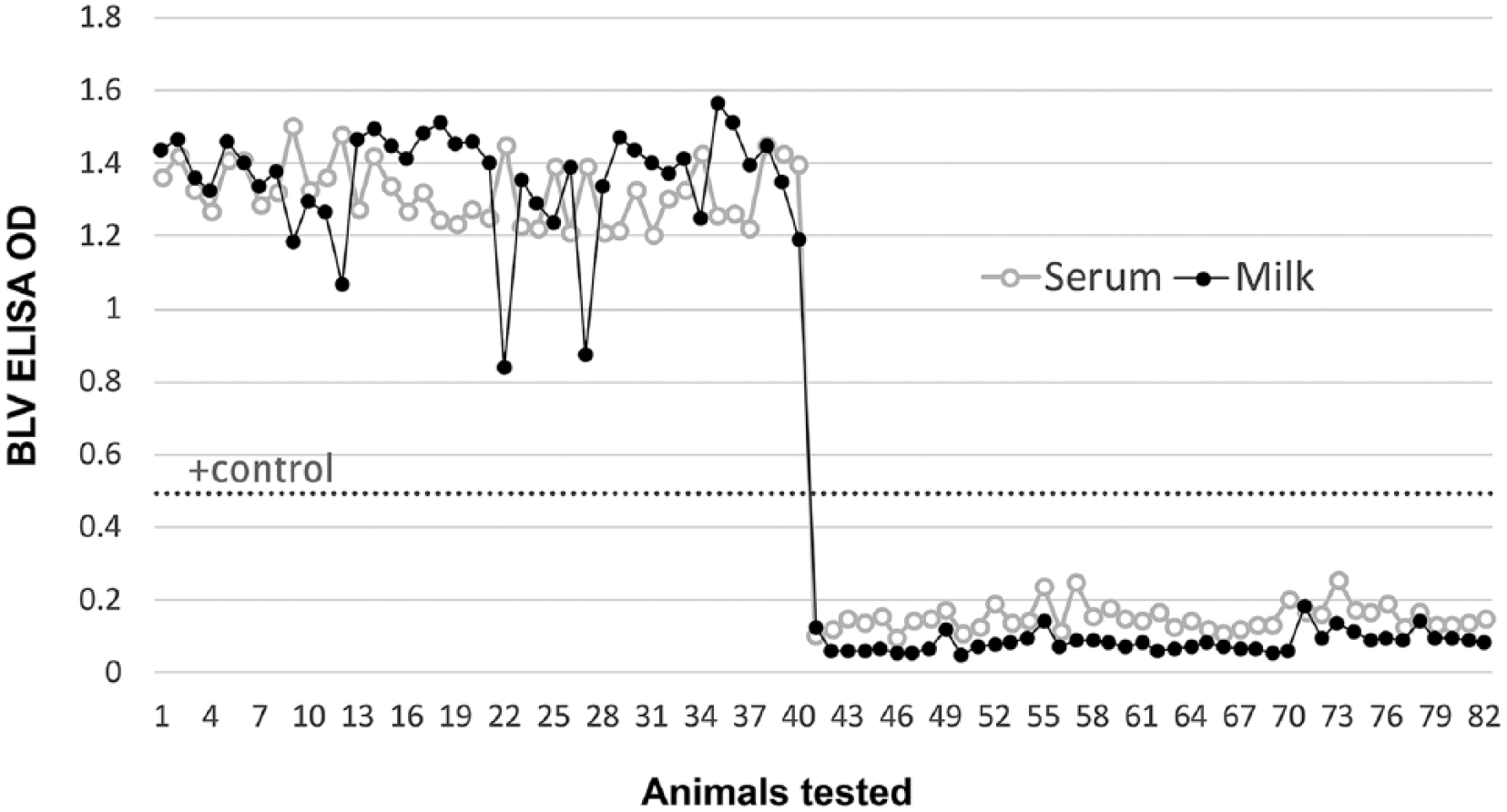
Graphic representation of the total animals tested (n = 82) for bovine leukemia virus (BLV) antibodies from 2 herds. Herd A = 1–41; herd B = 42–82. Correlation between the BLV serum and milk antibody ELISA optical density (OD) results was 97.3%.
Our results are consistent with prior studies comparing milk with serum as a means of testing for BLV antibodies, and support the use of the BLV serum ELISA, on properly collected and processed milk samples.6,11,15,18 Studies should be expanded to include whether the VMRD assay can be used successfully on bulk tank milk or pooled milk samples.22,29 Given that it has been reported that up to 10% more of recently infected dairy cattle can test positive by BLV-specific PCR in comparison with the serum ELISA, 22 it would be useful to determine if the milk ELISA would offer increased sensitivity during the first few weeks following BLV infection in lactating animals. The use of milk for detection of BLV-specific antibodies allows producers to collect samples, and together with their herd veterinarian, develop strategies to reduce the spread of BLV, such as by heat-treating the milk or withdrawing milk from BLV-infected cows from calf-feeding programs.1,12,19,27
Footnotes
Acknowledgements
We thank the herd managers from herds A and B for access to the animals used in our study, Drs. A Bredenberg and G Barrington who contributed to the completion of this project, and L Love for assistance with manuscript preparation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The project was funded in part from VMRD through laboratory supplies for assays, and a travel stipend for one author (DM DeAvila) to a national meeting.
