Abstract
Triglyceride concentrations in dogs with hyperlipidemic disorders can exceed concentrations used by assay manufacturers for interference testing. High-speed centrifugation or the polar solvent LipoClear reduce triglyceride concentrations, but efficacy requires evaluation in veterinary species. We determined the effect of marked hypertriglyceridemia on canine biochemistry testing; assessed the ability of high-speed centrifugation or LipoClear to correct lipemic interferences; and determined if LipoClear introduces inaccuracy into biochemistry assays. Fifteen pooled canine serum samples were aliquoted and spiked with equal volumes of water or Intralipid [triglyceride concentration 33.9 mmol/L (3,000 mg/dL)]. Intralipid aliquots underwent lipid removal by high-speed centrifugation or LipoClear treatment, and a water-spiked aliquot underwent LipoClear treatment. Biochemistry panels were performed using a Vitros 4600 chemistry analyzer. Results were compared by paired t-test or Wilcoxon test. Total observed errors were considered clinically acceptable if below veterinary allowable total error (TEa) guidelines. Statistically significant (p ≤ 0.05) interferences were introduced by Intralipid for 15 of 15 analytes. Median observed error exceeded TEa for potassium and enzymatic carbon dioxide, neither of which were identified by the manufacturer as susceptible to lipemic interference. After centrifugation, median observed error exceeded TEa for potassium and chloride. LipoClear treatment resulted in median errors that exceeded TEa for total protein, chloride, and phosphorus. Given that severe lipemia can occur in dogs with primary or secondary hyperlipidemia, veterinary laboratories should perform their own interference testing at triglyceride concentrations relevant to their patient population and provide this information to clinicians to ensure optimal case management.
Introduction
Incorrect laboratory test results can lead to inappropriate diagnostic or treatment decisions.2,22 One cause of error is interference, which occurs when the reported concentration of the substance of interest is falsely increased or reduced by another substance present in the sample. 18 Lipemia, observed as the milky appearance of serum containing high concentrations of lipoproteins, can create interference by multiple mechanisms. 18 Such mechanisms include creating random light scatter, providing a substrate for nonspecific antibody binding, and physical effects, such as reduced oxygen diffusion. 18 The magnitude of these effects vary from small statistically significant, but likely clinically irrelevant, differences to larger changes capable of altering clinical interpretation.6,24,29 To ensure patient safety, it is therefore important that veterinarians are aware of the effect of lipemia on biochemistry testing.
Assessment of the effect of lipemia is required for full validation of laboratory assays. 11 However, for established commercial assays, veterinary laboratories often rely solely on data provided by the manufacturer regarding the effect of lipemia and other interferents. The repeatability of these claims has come into question; a human study failed to replicate manufacturers’ results for lipemic interference studies across a wide range of biochemistry analytes. 19 This may be an even greater issue in veterinary testing, given that manufacturers typically perform interference studies using human samples, which may not fully replicate the matrix effects of canine serum.3,25
It is also questionable if the degree of lipemia tested by manufacturers of human assays is adequate for assessment of likely interferences in veterinary samples. Lipemic interference testing typically involves spiking samples with known concentrations of triglycerides. 18 This does not fully recapitulate clinical lipemic samples in which increased turbidity is the result of increased concentrations of chylomicrons and very low density lipoproteins (VLDLs). 18 Although predominantly composed of triglycerides, VLDLs and chylomicrons also contain other substances including apolipoproteins and cholesterol.18,32 The correlation between lipemia and triglyceride concentration is therefore not absolute, but it is concerning that manufacturers often test the effects of only relatively moderate increases in triglycerides, typically in the region of 11.3 mmol/L (1,000 mg/dL).18,19 This threshold may be exceeded by dogs with primary hyperlipidemia27,33 or with elevated triglycerides secondary to a range of conditions including diabetes mellitus and hypothyroidism.9,12,14,17 Optimal management of cases with marked hyperlipidemia requires investigation of the effect of clinically relevant concentrations of triglycerides on laboratory test accuracy.
One argument against lipemic interference as a clinically relevant problem is that lipid-clearing techniques, such as high-speed centrifugation or addition of polar solvents, can be used to clear lipemia before analysis of lipemic samples. 18 However, human studies suggest that the centrifugation speeds commonly used in veterinary medicine are insufficient to fully correct lipemic interferences and may introduce additional sources of error, such as depletion of hydrophobic analytes.18,23 Although there is some evidence that non-polar solvents are more effective than centrifugation for removal of interference for specific analytes, 23 studies using human plasma reveal that a commercial polar solvent introduced bias for other analytes. 26 Similar studies have not been performed for veterinary samples.
We hypothesized that marked hyperlipidemia will interfere with biochemistry tests performed on canine serum, and that removal of lipid by high-speed centrifugation or the polar solvent LipoClear (StatSpin; Iris Sample Processing, Westwood, MA) will reduce lipemic interference but may introduce other errors. Study objectives were to 1) determine which biochemistry analytes, assayed using a Vitros biochemistry system (Ortho Clinical Diagnostics, Raritan, NJ), were altered by marked lipemia; 2) determine if LipoClear or high-speed centrifugation corrects lipemic interference; and 3) determine if addition of LipoClear alone creates error.
Materials and methods
Serum samples
Prospectively collected study samples from healthy dogs (n = 5) belonging to staff and students, as well as canine patients (n = 10) presented to the Texas A&M Veterinary Medical Teaching Hospital (TAM-VMTH; College Station, TX) for a variety of underlying health conditions, were obtained with owner consent and IACUC approval (healthy dogs: protocol 2016-0340 CA, expiration date 2020.01.09; clinical patients: protocol 2016-0341 CA, expiration date 2020.01.16) between 2017.06.21 and 2017.07.25. To ensure that a clinically relevant range of analyte concentrations was assessed, remnant serum remaining after clinical testing for canine patients at the TAM-VMTH was also used in the study. Owner consent to use this remnant material was not explicitly sought for this study, but owners were offered the option to deny permission for use of remnant material for research or teaching during the admission process.
For prospectively collected samples, 9 mL of whole blood was collected by routine venipuncture and serum separated by centrifugation at 1,500 × g for 15 min. Remnant samples were processed similarly as part of routine clinical testing at the TAM Veterinary Clinical Pathology laboratory. Both prospectively collected and remnant samples were included in the study only if free from lipemia, icterus, and hemolysis on visual inspection. Samples were stored at −80°C until analysis. Immediately before use, samples were thawed for 5 min in a 37°C water bath. To obtain a clinically relevant range of values for the various analytes, some samples were pooled. A total of 15 samples were assayed for each analyte.
Spiking studies
Each pool was split into 5 aliquots. Intralipid 20% emulsion (Baxter Healthcare, Deerfield, IL) was added to 3 aliquots for a final triglyceride concentration of 33.9 mmol/L (3,000 mg/dL), which was selected based on triglyceride concentrations reported in primary hyperlipidemia of Miniature Schnauzers. 33 An equal volume of water was added to the 2 remaining aliquots. One water-spiked aliquot and one Intralipid-spiked aliquot underwent no further processing before analysis. One Intralipid-spiked aliquot was centrifuged at 12,000 × g for 5 min, and the infranatant was separated and used for analysis. One water-spiked aliquot and one Intralipid-spiked aliquot were treated with LipoClear, according to the manufacturer’s instructions. Briefly, 500 µL of serum was added to a microcentrifuge tube (StatSpin LC40; Iris Sample Processing) containing 100 µL of LipoClear, vortexed, and allowed to stand for 5 min at room temperature. Samples were then centrifuged at 2,000 × g for 20 min and the lipid-cleared layer collected for analysis.
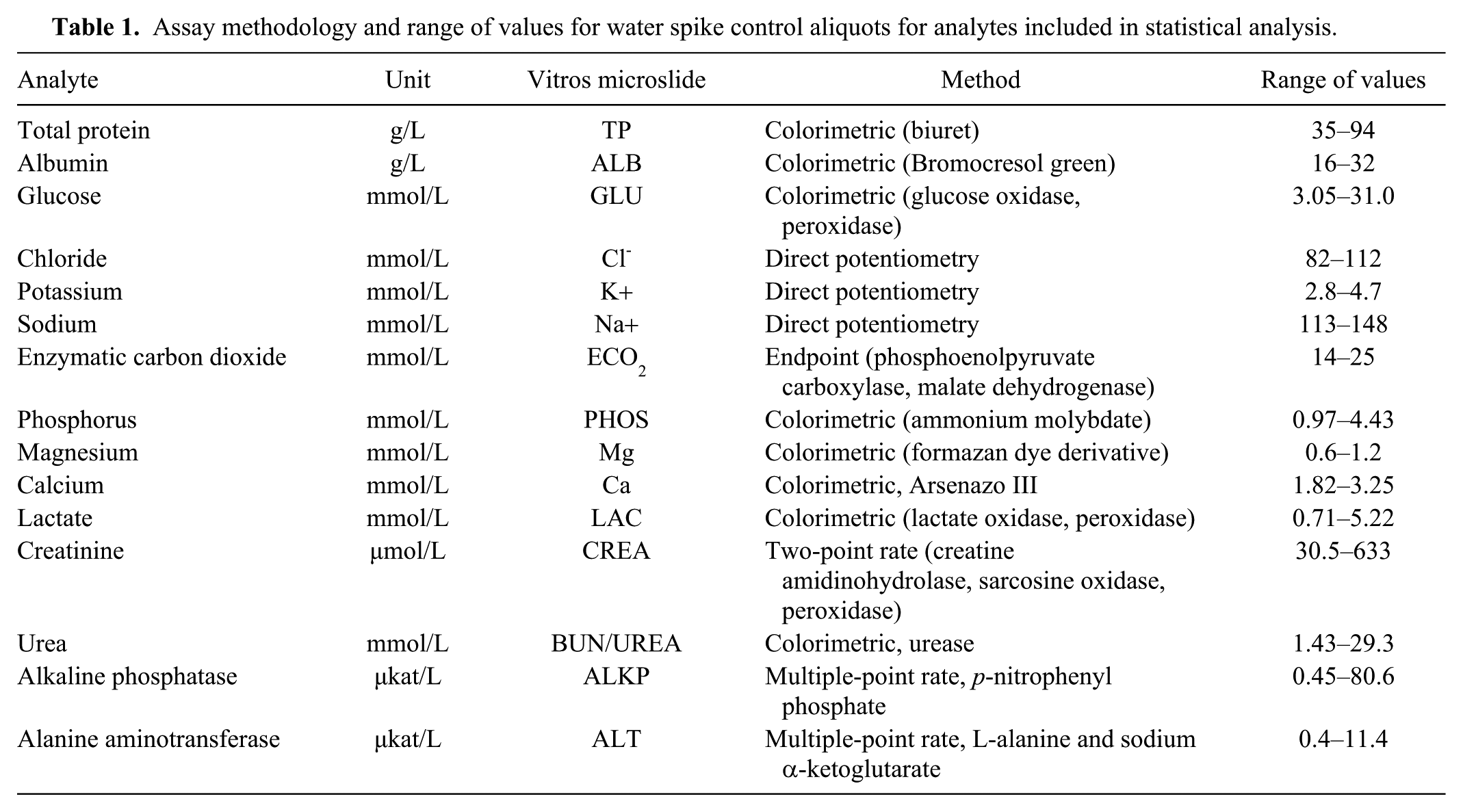
All assays were performed with dry-slide technology (Vitros 4600 biochemistry system; Ortho Clinical Diagnostics); details of the assays used are provided (Table 1).The instrument underwent daily quality control throughout the study and, to minimize effects of analytical variation, all aliquots from a single sample were analyzed consecutively in the same analytical run. For each aliquot, the following analytes were measured: total protein, albumin, glucose, chloride, potassium, sodium, enzymatic carbon dioxide, phosphorus, magnesium, calcium, lactate, creatinine, urea, total bilirubin, alkaline phosphatase (ALP), alanine aminotransferase (ALT), and gamma-glutamyl transferase (GGT). Results above the reportable range were obtained by reflex dilution, and results below the reportable range were excluded from analysis. For LipoClear-treated aliquots, results were corrected for dilution according to the manufacturer’s instructions, which require multiplication of most results by 1.2 (for sodium, chloride, and potassium, the user is required to compare the results of 10 or more non-lipemic samples with and without the addition of LipoClear). We used the results of the water-spiked controls and water-spiked aliquots treated with LipoClear for this calculation, resulting in a user-specific dilution factor of 1.1 for electrolytes.
Assay methodology and range of values for water spike control aliquots for analytes included in statistical analysis.
Data analysis
To determine if high triglyceride concentrations achieved by addition of Intralipid introduced bias, results for the Intralipid-spiked sample were compared to the non–LipoClear-treated, water-spiked control aliquot. To determine if high-speed centrifugation or LipoClear corrected biases that were introduced by Intralipid, the results for the Intralipid-spiked aliquot that underwent centrifugation or LipoClear treatment were compared to the non–LipoClear-treated, water-spiked control. To determine if LipoClear introduced bias, results for the LipoClear-treated, water-spiked aliquot were compared to the non–LipoClear-treated, water-spiked control. Given that the LipoClear-treated, water-spiked aliquot results were used to calculate the user-specific dilution factor for sodium, potassium, and chloride, electrolytes were not included in the assessment of bias introduced by LipoClear alone.
Analysis was performed (v.18.6; MedCalc Software, Ostend, Belgium). To statistically assess bias, data distribution was first assessed by the D’Agostino–Pearson test. If results for both the non–LipoClear-treated, water-spiked control and the treated aliquot were normally distributed, aliquots were compared by paired t-test, and if one or both datasets were non-normal, the aliquots were compared by the Wilcoxon rank test. Results are presented using both p ≤0.05 and p <0.0009 to define significance. An alpha of 0.0009 was selected based on a Bonferroni correction for the 57 statistical comparisons performed.
To provide an indication of likely clinical significance, percentage bias was calculated for each aliquot using the equation: bias(%) = [(treated aliquot – water-spiked control)/water-spiked control)] × 100%. A positive result therefore reflects overestimation of the analyte in the treated sample, and a negative result, underestimation. Median percentage bias was compared to the American Society for Veterinary Clinical Pathology’s (ASVCP) allowable total error (TEa) for the analyte, which is a quality goal based on the magnitude of measurement error considered clinically relevant by an expert panel of clinicians and clinical pathologists. 13 For analytes with different TEa for above, below, or within reference interval (RI) results, the median bias was compared to the most conservative TEa (i.e., if TEa was 5% for within and above RI samples and 10% for below RI samples, median bias was compared to the 5% threshold).
Results
Sample characteristics
Concentrations above, within, and below RI were achieved for most analytes (Table 1). No samples had above RI for albumin, chloride, or enzymatic carbon dioxide, or below RI for phosphorus, creatinine, ALP, or ALT. Total bilirubin and GGT were excluded from analysis because concentration for one or more aliquots was below the reportable range for 10 and 11 samples, respectively.
Effects of high concentrations of lipid
There were statistically significant differences (p ≤0.05) between the water-spiked controls and the Intralipid-spiked samples for all analytes (Table 2). After applying a Bonferroni correction (p <0.0009), the effect of lipid remained significant for chloride (median bias: 4.7%, p <0.0001), potassium (6.5%, p <0.0001), sodium (2.9%, p <0.0001), enzymatic carbon dioxide −21.8%, p = 0.0001), phosphorus (9.3%, p = 0.0001), calcium −2.3%, p = 0.0001), ALP (4.4%, p = 0.0002), ALT (5.5%, p = 0.0001), and magnesium −6.3%, p = 0.0005). Median bias exceeded TEa for potassium and enzymatic carbon dioxide.
Lipemic interferences identified by comparison of Intralipid-spiked aliquots to water-spiked controls, and effect of lipid clearance by high-speed centrifugation or LipoClear treatment.
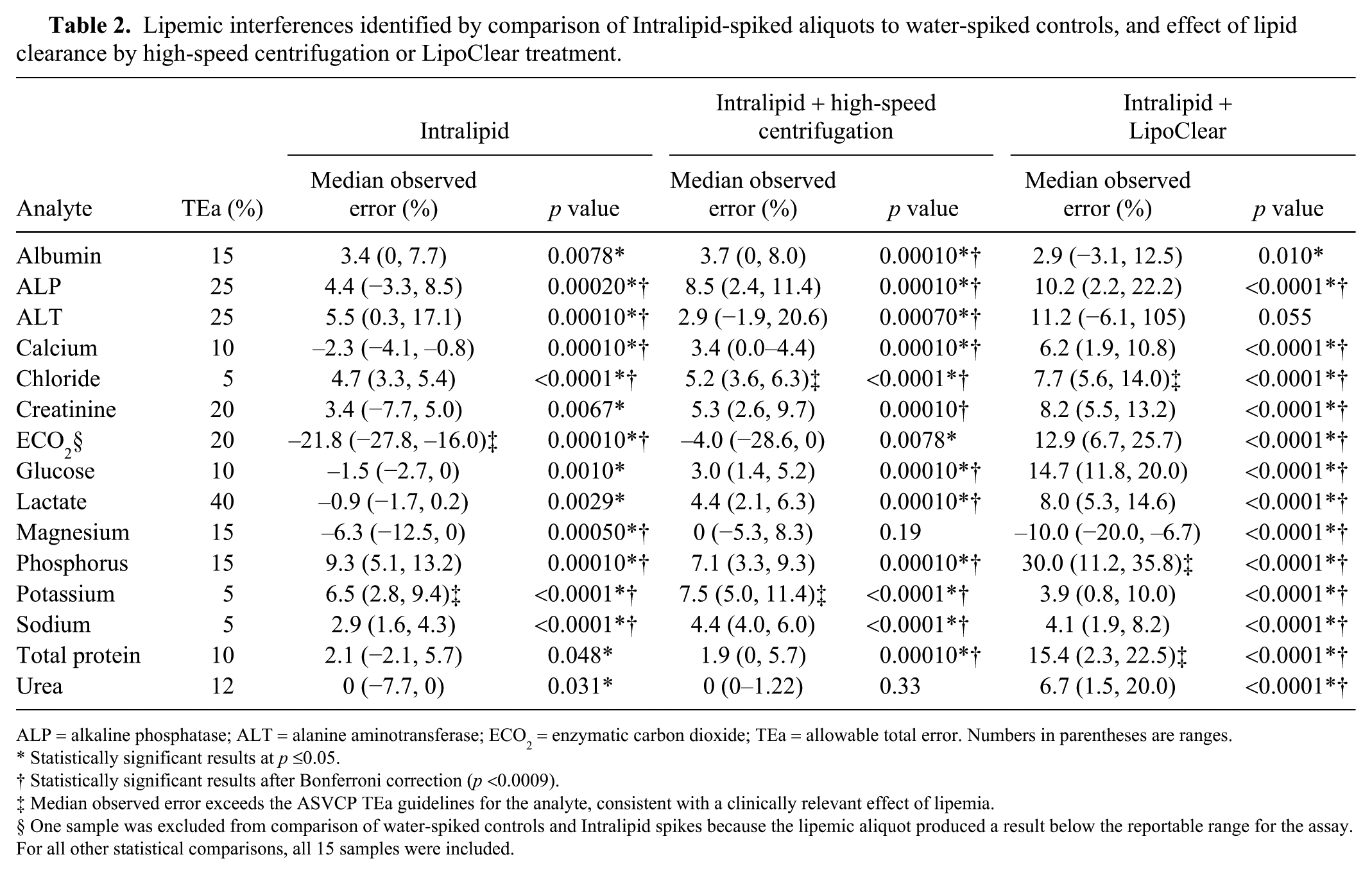
ALP = alkaline phosphatase; ALT = alanine aminotransferase; ECO2 = enzymatic carbon dioxide; TEa = allowable total error. Numbers in parentheses are ranges.
Statistically significant results at p ≤0.05.
Statistically significant results after Bonferroni correction (p <0.0009).
Median observed error exceeds the ASVCP TEa guidelines for the analyte, consistent with a clinically relevant effect of lipemia.
One sample was excluded from comparison of water-spiked controls and Intralipid spikes because the lipemic aliquot produced a result below the reportable range for the assay. For all other statistical comparisons, all 15 samples were included.
Effects of high-speed centrifugation on lipemic interferences
After high-speed centrifugation, statistically significant differences (p ≤0.05) between water- and Intralipid-spiked samples were no longer present for urea or magnesium (Table 2). After application of a Bonferroni correction (p <0.0009), there was also no longer a statistically significant difference for enzymatic carbon dioxide. The direction of bias remained the same for most analytes, but glucose, lactate, and calcium had negative biases for the unprocessed lipid-spiked sample, as well as positive biases after centrifugation. Median bias remained higher than TEa for potassium (7.5%, p <0.0001), and, additionally, slightly exceeded TEa for chloride (5.2%, p <0.0001).
Effects of LipoClear on lipemic interferences
After LipoClear treatment, statistically significant differences (p ≤0.05) were present between water-spiked controls and Intralipid-spiked, LipoClear-treated samples for all analytes except ALT (Table 2). After application of a Bonferroni correction (p <0.0009), there was no longer a statistically significant difference for albumin. The direction of bias was consistent with the unprocessed lipid-spiked samples, except for glucose, enzymatic carbon dioxide, lactate, urea, and calcium, which had 0% or negative median biases for the lipid-spiked samples and positive biases after LipoClear treatment. Median bias no longer exceeded TEa for potassium (3.9%, p <0.0001) or enzymatic carbon dioxide (12.9%, p <0.0001), but did exceed TEa for total protein (15.4%, p <0.0001), chloride (7.7%, p <0.0001), and phosphorus (30%, p <0.0001).
Effects of LipoClear alone
Addition of LipoClear to non-lipemic samples resulted in statistically significant differences (p ≤0.05) from the untreated controls for all analytes except total protein and albumin (Table 3). Differences remained statistically significant after application of a Bonferroni correction (p <0.0009) for glucose (11.5%, p = 0.0001), enzymatic carbon dioxide (12%, p <0.0001), phosphorus (22.9%, p <0.0001), ALP (4.4%, p = 0.0004), and magnesium −10%, p <0.0001). The direction of biases was the same as for addition of LipoClear to lipemic samples. Median bias exceeded TEa for total protein and phosphorus.
Interferences introduced by addition of the lipid-clearing agent LipoClear in the absence of lntralipid.
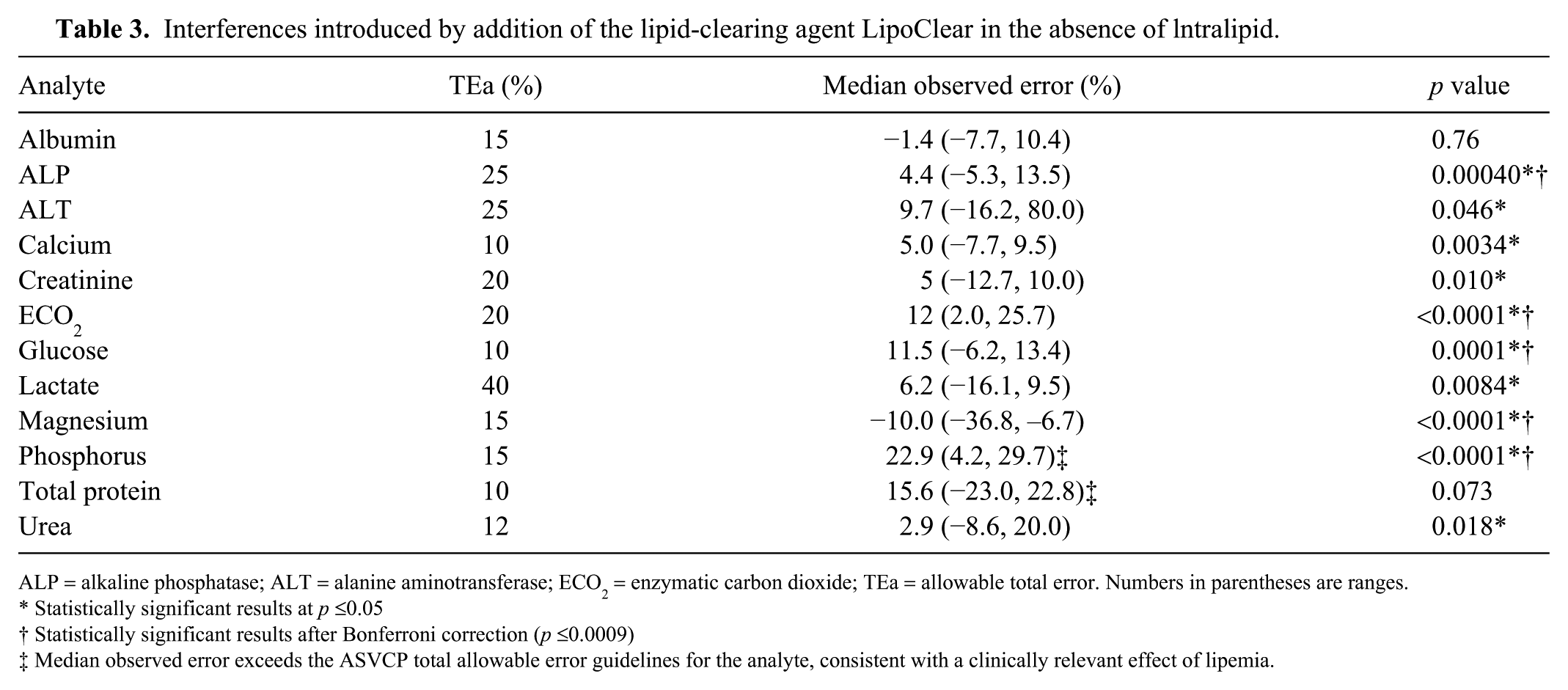
ALP = alkaline phosphatase; ALT = alanine aminotransferase; ECO2 = enzymatic carbon dioxide; TEa = allowable total error. Numbers in parentheses are ranges.
Statistically significant results at p ≤0.05
Statistically significant results after Bonferroni correction (p ≤0.0009)
Median observed error exceeds the ASVCP total allowable error guidelines for the analyte, consistent with a clinically relevant effect of lipemia.
Discussion
A triglyceride concentration of 33.9 mmol/L (3,000 mg/dL) introduced statistically significant interferences for most biochemistry analytes measured by a widely used dry-slide chemistry system. In most instances, the magnitudes of effect were considerably lower than the TEa values for the analytes, as defined by the ASVCP, 13 and are therefore unlikely to affect patient care. However, median bias exceeded TEa for enzymatic carbon dioxide, and above-or-within RI potassium. These findings should be interpreted with some caution given that Intralipid does not perfectly mimic lipemia, 18 but suggest that clinically important overestimation of potassium, and underestimation of enzymatic carbon dioxide, may occur in markedly lipemic canine samples. High-speed centrifugation failed to bring median bias below TEa for potassium, demonstrating that high-speed centrifugation is insufficient to remove all lipemic interferences. Addition of LipoClear not only failed to correct several lipemic interferences, but also introduced other potentially clinically significant errors.
Our analysis aimed to determine the likely clinical relevance of lipemic interferences rather than relying solely on statistical significance. Clinical relevance was assessed by comparing median bias to TEa guidelines from the ASVCP. 13 TEa defines acceptable performance for an analytical method 13 ; we used TEa to determine the maximum acceptable difference between results for aliquots of the same sample. 20 The ASVCP recommendations were based on consensus of a panel of veterinary clinical pathologists and specialist veterinarians regarding the maximum difference that can be tolerated before diagnosis or treatment decisions are likely to be negatively affected. 13 Comparing interferences to TEa therefore helps to determine their likely clinical importance. This is an advantage over relying on statistical comparison alone, given that if enough samples are analyzed, statistical significance can be detected even when the magnitude of effect is too small to impact patient care.15,28 Furthermore, interference studies inevitably involve large numbers of statistical comparisons, risking type 1 errors as a result of multiplicity. 15 Given that analytical error is composed of both bias and imprecision, 13 it could be argued that our approach is too conservative. If we had combined the magnitude of biases induced by lipemia and the instrument’s coefficient of variation for each analyte, we may have identified additional clinically relevant errors. Nevertheless, our simple approach does highlight the interferences that are of most obvious clinical relevance.
Detection of errors exceeding TEa in our study demonstrates that veterinary users should perform interference studies for their own instruments using interferent concentrations relevant to their patient population, rather than relying solely on manufacturer’s interference data. Datasheets for the slides used in our study did not identify lipemic interferences for potassium or enzymatic carbon dioxide [Ortho Clinical Diagnostics: Vitros chemistry products K slides, coatings 800 and above instructions for use (Publication J32746:6), Vitros chemistry products ECO2 slides (for coatings 3500 and above) instructions for use (Publication J40112:4)]. Three possible explanations for the differences between our results and datasheet claims are suggested. First, it may be that the interferences for potassium and enzymatic carbon dioxide do not occur until triglycerides are more markedly increased than the concentrations tested by the manufacturer. The datasheets for the assays used herein report testing up to triglyceride concentrations of 9.04–10.2 mmol/L (800–900 mg/dL). We used a considerably higher concentration of 33.9 mmol/L (3,000 mg/dL). This was selected because triglyceride concentrations >33.9 mmol/L (3,000 mg/dL) have been reported in Miniature Schnauzers with primary hyperlipidemia.31,33 Second, our results may reflect differences in serum matrix between the canine samples that we used and the human samples used by the manufacturer. This is considered less likely, given that both our data and the manufacturer’s data are derived from addition of Intralipid rather than species-specific material. Third, it is possible that our results reflect differences between manufacturer and in-clinic data, similar to investigations using human plasma or serum that failed to replicate at least some manufacturer data regarding lipemic interferences, even when using identical triglyceride concentrations. 19 This could reflect lab-to-lab variability in instrument performance or lot-to-lot variability in reagents.
A weakness of both our data and the data reported by most instrument manufacturers is the use of Intralipid, which is a parenteral nutrition solution containing high concentrations of triglycerides with a particle size of 200–600 nm. 18 This does not fully replicate lipoproteins in circulation, given that VLDL fall below this size range, and larger chylomicrons exceed it. 18 Concentrated human triglyceride-rich lipoproteins are commercially available as an alternative, potentially more physiologically relevant, spiking material. 1 However, differences in lipoprotein profiles between dogs and humans limit their relevance in veterinary studies. 32 An alternative used by some investigators is to compare results for naturally occurring grossly lipemic samples before and after ultracentrifugation. This is helpful in identifying lipemic interferences that are not replicated by Intralipid. For example, this approach was used to identify lipemic interferences for human ceruloplasmin, prealbumin, and transferrin that were not seen with addition of Intralipid. 5 However, if interferences are detected with Intralipid spikes but not native plasma, this could reflect a failure of centrifugation to adequately clear plasma rather than an absence of interference.
With the caveat that differences may exist between the effects of physiologic lipemia and Intralipid, our results suggest that when measured using a Vitros chemistry system, potassium and enzymatic carbon dioxide should be interpreted with some caution in dogs with marked hypertriglyceridemia. This is of particular relevance for management of conditions in which hypertriglyceridemia is likely to occur in combination with electrolyte and acid-base derangement, such as diabetic ketoacidosis. The median bias for potassium was 6.5%, slightly exceeding the 5% TEa for within and above RI results and below the 10% TEa acceptable for below RI results. 13 To provide clinical context, it has been recommended that insulin therapy be delayed in diabetic ketoacidosis until potassium concentration is at least 3.5 mmol/L. 21 A 6.5% positive bias would mean that a dog with a true potassium concentration of 3.4 mmol/L would have a predicted measured potassium of 3.6 mmol/L. This demonstrates that the effect of marked hypertriglyceridemia is unlikely to lead to gross errors, such as failure to recognize severe hypo- or hyperkalemia, but could affect monitoring for trend and therapeutic decisions made in response to small changes in potassium concentration. 21
When considering the clinical implications of our data, it is necessary to emphasize that a single biochemistry system was used and results are likely not directly transferable to other analyzers. The Vitros biochemistry system relies on dry-slides that contain masking layers designed to reduce interference [Ortho Clinical Diagnostics, Vitros microslide assay summary (Publication J23323)]. Our findings therefore may not be relevant to performance of wet chemistry systems or other dry-slide technologies. Products from different manufacturers may show marked differences in the effect of interferences for the same analyte. For example, icterus may cause overestimation or underestimation of phosphorus, or have no effect depending on the analyzer used. 10 Additionally, we focused on median bias across 15 serum pools with various analyte concentrations. This provides an assessment of average performance across a range of clinically relevant analyte concentrations, but could fail to identify any interferences that vary markedly with analyte concentration, as has been reported for the effect of hemolysis on bilirubin. 16
Our results suggest that high-speed centrifugation is helpful before analysis of severely lipemic samples, but does not completely reverse the effects of severe lipemia. High-speed centrifugation reduced median bias to below TEa for enzymatic carbon dioxide, but not potassium. Median bias for chloride was also shifted from slightly below to slightly above TEa. This small change may reflect the effects of random measurement error (i.e., imprecision) on our estimates of median bias, rather than a genuine change introduced by centrifugation. In contrast, the large reduction (17.8%) in median bias for enzymatic carbon dioxide is suggestive of a true reduction in interference after centrifugation. The ability of high-speed centrifugation to correct or reduce some but not all lipemic interferences is similar to findings for human samples.7,8,23 Our use of Intralipid may have exaggerated the effectiveness of high-speed centrifugation. The size of Intralipid particles overlaps with chylomicrons, rather than VLDL. Large and medium-sized VLDL (~60–200 nm diameter) can contribute to turbidity of lipemic samples and are reported to be less effectively cleared by high-speed centrifugation than chylomicrons. 18 This may be clinically relevant, given that increased concentrations of VLDL occur in Miniature Schnauzers with primary hyperlipidemia 30 and a variety of secondary canine hyperlipidemic conditions, including nephrotic syndrome, 4 chronic renal disease, 4 and endocrinopathies. 32 Ultracentrifugation at speeds of ≥100,000 × g is considered the gold standard for correction of lipemic interferences, but ultracentrifuges are not widely available in veterinary settings. 18
One somewhat surprising finding was that high-speed centrifugation altered the direction of biases for glucose, lactate, and calcium after centrifugation. Although both lactate and calcium biases consistently remained significant after Bonferroni correction, the magnitude of these errors is small when compared to TEa for these analytes. This suggests that these effects may represent random analytical error and the inevitable false-positive results associated with multiple statistical tests. An alternative explanation could be that this is an artifact introduced by the study design. To correct for dilution, lipid-spiked aliquots were compared to control aliquots spiked with an equal volume of water. Centrifugation led to separation of the added lipid fraction from the infranatant, potentially leading to slight differences in the dilutional effect of adding Intralipid versus water.
Our data do not support the use of LipoClear for canine serum samples given that, consistent with human studies, we identified errors introduced by addition of LipoClear.7,26 Although LipoClear treatment of lipemic samples reduced median biases below TEa for enzymatic carbon dioxide and potassium, median biases exceeding TEa were introduced for chloride, total protein, and phosphorus. The error for phosphorus remained both statistically significant and greater than TEa even when LipoClear was added to non-lipemic samples, suggesting that this was a direct effect of LipoClear treatment rather than failure to correct lipemic interferences. The effect on total protein was no longer statistically significant when LipoClear was added to non-lipemic samples, but there was a large increase in the median and range for observed errors compared to results for Intralipid alone or after centrifugation. This suggests that LipoClear considerably increased imprecision, which may reflect lack of consistent separation from plasma during processing. The effect of LipoClear alone could not be assessed for chloride because the manufacturer requires the user to determine a user-specific dilution factor for electrolytes using non-lipemic samples. The product insert reports that LipoClear may introduce error for total protein and phosphorus, but the effect on chloride is not included in the limitations section; the manufacturer predicts an underestimate of total protein, rather than the overestimate reported herein [Iris Sample Processing, LipoClear package insert (Publication 54-004752-001 Rev. B)]. We suggest that if a veterinary laboratory does opt to use LipoClear for lipid clearance, then efforts should be made to assess in-house performance.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was generously funded by the American Kennel Club Canine Health Foundation (U Jeffery, 02343-A). The contents of this publication are solely the responsibility of the authors and do not necessarily represent the views of the Foundation. The results of our study were presented as an oral abstract at the 2018 American College of Veterinary Pathology annual meeting, Nov 3–7, Washington, DC.
