Abstract
We evaluated the oncotic pressure (plasma colloid osmotic pressure, πc) in a group of healthy, captive Asian elephants (Elephas maximus; n = 21) with a colloid osmometer with a membrane cutoff of >20,000 daltons. The median πc for these elephants was 26.3 mm Hg with an interquartile interval of 25.5–26.8 mm Hg. The mean πc value was 26.0 mm Hg ± SD 1.1. We found moderate correlation between albumin measured by electrophoresis and πc (r = 0.622; p = 0.003). After a 16-h water deprivation test in a subset of elephants (n = 16), a difference in πc was not detected, despite a significant increase in serum total proteins, urea, and osmolality. These results indicate that πc is not a sensitive indicator of hydration status in elephants after a short period of water deprivation. Use of oncotic pressure as a diagnostic tool in diseased Asian elephants warrants further investigation.
Elephants provide distinct challenges to the veterinary practitioner performing a physical examination. In our clinical experience, the subtle signs of mild dehydration, such as skin tent, tackiness of mucous membranes, and quantification of urinary output, are impractical in elephants given their thick, non-pliable skin, copious salivary secretions, extremely voluminous urine production, and limitations on physical examination because of patient behavioral compliance and human safety. In addition, studies of Asian and African elephant physiology continue to identify unique species characteristics, particularly in hemodynamics. Asian elephants are reported to have low serum osmolality. 5 Elephants have an erythrocyte volume ~3 times that of the horse. 14 African elephants have erythrocytes with increased flexibility and, in healthy states, they have plasma viscosity approaching that seen with advanced renal insufficiency in other species. 14 Asian elephants are reported to have albumin with polymorphism. 10 Close study of normal values and potential clinical applications of basic physiologic parameters in elephants are valuable.
Oncotic pressure (πc), also known as plasma colloid osmotic pressure, is the force exerted by plasma proteins that retains fluid in the intravascular rather than the interstitial compartment. 3 Albumin accounts for the majority of πc, but a combination of gamma globulins, fibrinogen, other plasma proteins, and their negative charges also contribute and create the Gibbs–Donnan effect.4,15 Oncotic pressure is altered by dehydration and conditions that affect vascular permeability and plasma protein production, loss, or transudation. 11 These include hemorrhage, hemolysis, sepsis, systemic inflammatory response syndrome, hypo- or hyperalbuminemia, protein-losing conditions, and hypergammaglobulinemia.3,9 Inflammatory mediators impact πc as a result of effects on transudation of albumin, which is under constant redistribution between the interstitial and intravascular spaces. 7 Oncotic pressure is also affected by fluid therapy. Monitoring πc can help determine patient hydration and reduce risks of fluid therapy–associated complications such as pulmonary edema. 3
A colloid osmometer/oncometer (COM) is an instrument used to measure πc. This machine has 2 chambers, separated by a membrane, each containing isotonic fluid. The selected membrane has a permeability (pore size) to restrict macromolecule movement based on molecular weight. Each type of membrane provides a characteristic reference number (cutoff value) indicated in daltons. Membranes with a cutoff of 10,000 or 20,000 daltons (Da) are commonly used in clinical applications. When a plasma sample is added to the test chamber, the increased πc of this chamber pulls water by osmosis across the membrane from the reference chamber. A pressure transducer measures the resulting pressure change, which is amplified and displayed as a positive, direct measure of πc of the sample. 9 Various methods for calculation of πc have been proposed; however, calculated results have proven unreliable in both human and veterinary patients.1,2,12 Therefore, direct measurement using a COM remains the standard for determining πc in a clinical setting.
Reference intervals (RIs) for πc in domestic species are typically 21–30 mm Hg (Table 1). 12 Oncotic pressure in African elephants (Loxodonta sp.) postmortem has been reported as much higher (mean 47.0 mm Hg, SD 12.9; n = 5); however, changes at death may alter πc. 6
Mean of Asian elephant oncotic pressure (πc; n = 21) in mm Hg compared to reported values in domestic species. 12

SEM = standard error of the mean.
We aimed to establish normal RIs for πc in a healthy population of Asian elephants (Elephas maximus) and to determine whether mild dehydration induced by a 16-h water deprivation could cause a statistically significant increase in πc in the population. The samples used were generated during a previously reported study. 5 All procedures were approved via Institutional Animal Care and Use Committee 201004899 through the University of Florida (UF). Elephants were owned and housed by the Ringling Brothers and Barnum & Bailey Center for Elephant Conservation in Florida.
In phase 1, 21 adult Asian elephants, all considered healthy based on serial physical examinations and normal behavior, were selected for blood sampling. Elephants were 11–67 y old and included 2 bulls, 1 castrated male, and 18 females. Using a sterile, 20-gauge needle, venous blood was collected from an ear vein of each elephant into 2 Vacutainer tubes (Becton, Dickinson, Franklin Lakes, NJ): a 10-mL red-top glass tube without additives or serum separator gel and a 3-mL green-top plastic tube containing 51 USP units of lithium heparin. Red-top tubes were filled first. Blood was centrifuged (10 min; 2,479 × g) after visible clotting occurred in red-top tubes, and within 1 h of collection in green-top tubes. Serum and plasma were separated and frozen in CryoVials (Simport Scientific, Beloeil, Quebec, Canada) for storage at −57°C (–70°F). Samples were submitted to laboratories in a frozen state and were thawed only on the day of testing. 13
The πc data were obtained on each plasma sample using a calibrated osmometer (Osmomat 050 colloid osmometer; Gonotec, Berlin, Germany). A membrane with a cutoff of >20,000 Da was used for all measurements. Serum chemistry panels were performed (Dimension Xpand clinical chemistry analyzer; Siemens Healthcare Diagnostics, Newark, DE) to obtain urea and creatinine concentrations. Serum protein electrophoresis panels were performed at the University of Miami Avian and Wildlife Laboratory (Miami, FL; Comparative pathology SPIFE 3000 system, Helena Laboratories, Beaumont, TX) to measure serum protein constituents that contribute to πc. The panel consists of albumin, alpha-1 globulins (α1), alpha-2 globulins (α2), beta globulins (β), and gamma globulins (γ). Total protein (TP) was determined by refractometry, and absolute individual protein fractions were determined by calculation using electrophoretogram-generated percentages. Osmolality by freezing point osmometer (FPO) was also obtained on all samples by methods described previously. 5
In phase 2, 7 mo after the initial blood collection, we performed a water deprivation test. Sixteen elephants were selected from the previously described group based on elephant availability and behavioral compliance for consecutive blood draws under time constraints. The group consisted of 1 castrated male and 15 female elephants (16–67 y old). This group typically received water ad libitum twice daily: once in the morning when shifting from barn to pasture, and once in the evening when returning to the barn for the night. As described previously, venous blood samples were collected at 2 times. 5 The initial blood samples (T0) were collected in the evening after the elephants were given free access to water and were moved into the barn. Water was withheld after the T0 sampling. The second blood samples (T1) were taken the following morning after the elephants were shifted to pasture. This was a mean of 16 h 34 min (min. 15 h 42 min; max. 17 h 33 min) post T0. After the T1 sampling, the elephants returned to receiving water as usual. The sampled elephants, therefore, had missed 1 routine water administration between T0 and T1. Packed cell volume (PCV) and TP (refractometer) were measured on each whole blood sample from the green-top tubes using standard techniques. 11 Serum and plasma were separated from the cellular component, frozen, and submitted to the laboratories for testing. Osmolality by FPO was also obtained on all samples by methods described previously. 5
Calculations were performed (MATLAB v.R2008b; MathWorks, Natick, MA). All statistical tests were performed at the 95% confidence interval with a p ≤ 0.05 considered significant. All comparison tests were 2-tailed. Given the small number of samples, results are presented as medians with interquartile intervals where appropriate.
In statistical analysis for phase 1, a Shapiro–Wilk test was used to evaluate data for normality, and p ≥ 0.05 was interpreted as indicating a normal distribution. The elephant πc means were compared to the reported reference means of comparison species (Table 1) using a Wilcoxon signed-rank test and t-test. Pearson product moment correlation coefficient (r) was used to assess correlations. In statistical analysis for phase 2, a Shapiro–Wilk test for normality was performed. Because not all data were normally distributed, Wilcoxon signed-rank tests were used to compare the T0 and T1 values for πc, PCV, TP, urea, creatinine, and osmolality.
In phase 1, the measured oncotic pressure data was a median of 26.3 mm Hg with an interquartile interval of 25.5–26.8 mm Hg. The Shapiro–Wilk test indicated that phase 1 data did not have a normal distribution (p = 0.04). The mean πc value was 26.0 mm Hg ± SD 1.1. When evaluating πc results in light of protein fractions measured by electrophoresis, there was a moderate correlation between albumin and πc (r = 0.622; p = 0.003). No other protein fractions exhibited a significant correlation with πc value. Sign-rank and t-tests were used to evaluate whether the observed elephant πc mean agreed with the published πc means in domestic species. A significant difference was observed for all comparisons (p < 0.001).
In phase 2, the Shapiro–Wilk test indicated that the T0 distribution was not normal (p = 0.01), but the T1 data may have a normal distribution (p = 0.28). The rank-sum test did not detect a significant difference in the median values of πc samples taken at T0 and T1 (p = 0.242). Both a sign-rank test (p = 0.408) and a paired t-test (p = 0.488) indicated no significant change in the paired samples of T0 and T1. From the T0-to-T1 time of water deprivation, there was a significant increase in TP, urea, and osmolality in this sample set as published previously. 5
The πc values in our healthy, captive Asian elephants were similar to data in other domestic species (Table 1). The narrow interquartile interval suggests that πc is tightly regulated in these elephants. In phase 2 data (Table 2), there was no detected change in πc from mild dehydration, which contrasts with the significant changes in TP, urea, and osmolality. 5 Our results indicate that πc was not a sensitive indicator of hydration status in these elephants. It is important to note that the level of dehydration in our study was mild and the sample population was small. It is expected that substantial dehydration in an elephant would impact πc, similar to other species.
Oncotic pressure (πc) values in mm Hg after a 16-h water deprivation test in 16 Asian elephants.
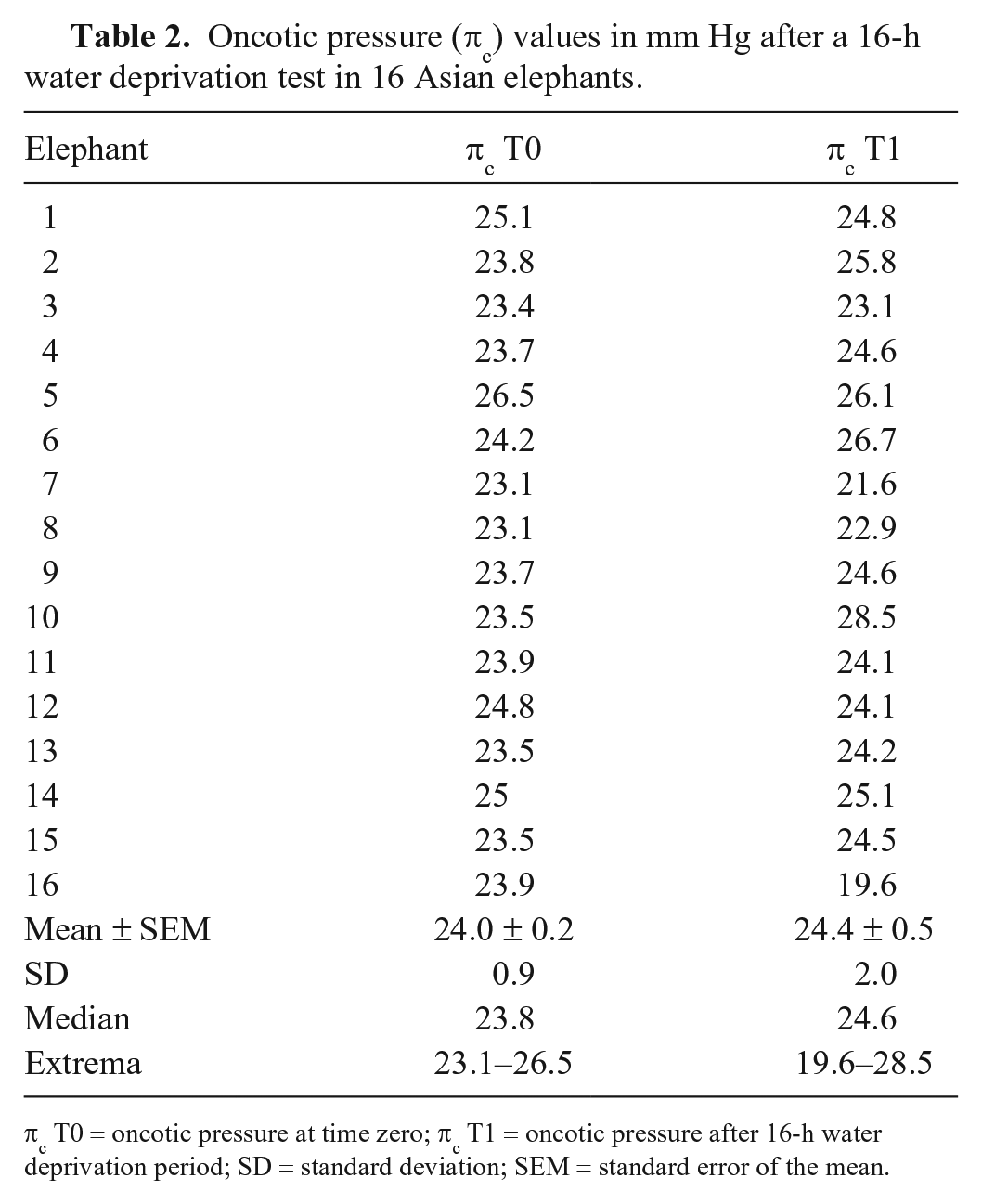
πc T0 = oncotic pressure at time zero; πc T1 = oncotic pressure after 16-h water deprivation period; SD = standard deviation; SEM = standard error of the mean.
The mean value of πc of 26.0 mm Hg ± SD 1.1 in this Asian elephant group is strikingly different from the previously reported value in African elephants of 47.0 mm Hg ± SD 12.9. 6 This suggests that results from the previous study may not be reflective of normal values, possibly as a result of dehydration or postmortem collection. Our results indicate that a πc value in an elephant that is outside the RI of 23.8–28.2 mm Hg (= mean ± 2 SD) may alert the clinical practitioner to an altered physiologic state caused by illness or environmental stressors. Diseases reported in elephants in which πc could be relevant include hemorrhagic disease caused by endotheliotropic herpesvirus, acute enterocolitis such as caused by Salmonella sp., protein-losing nephropathy, and, less commonly, hepatic insufficiency caused by Fasciola jacksoni. 8 Evaluation of πc in the ill elephant warrants investigation to better understand the clinical implications of πc changes in this species.
Albumin was found to have moderate correlation with πc in this Asian elephant group. In domestic species, 75–80% of πc force is generated by albumin. 11 The negative charge on albumin and other proteins creates noncovalent bonds with cations such as sodium and potassium. These cations exert an osmotic force, known as the Gibbs–Donnan effect, that is additive to πc and is independent of the plasma cation concentration.3,4 Healthy elephants are known to have concentrations of cations, such as sodium, that are significantly lower than in other species. 5 Further research is indicated to explore the effects of albumin changes and electrolyte interactions on πc in elephants.
Artifacts were not observed in our study, but may be noted under conditions that affect πc, such as hemolysis, significant dehydration, recumbency versus standing, tourniquet application, high glucose concentrations affecting equilibration across the machine membrane, or false elevations with citrated or EDTA plasma. 13 Given the large body mass of elephants, it is feasible that drawing blood from a peripheral vein during prolonged recumbency, such as an anesthetic event, could result in artifactual changes in πc.
The roles of elephant plasma proteins are not yet fully understood. Continuing to explore the normal physiology of both Asian and African elephants in states of health will help veterinary practitioners develop appropriate laboratory tests and therapies for these species.
Footnotes
Acknowledgements
We thank the staff of the Ringling Bros. and Barnum & Bailey Center for Elephant Conservation; Dr. Carolyn Cray; and Heather Hoffman for assistance in sample processing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded in part by the NIH/NCRR Clinical and Translational Science Awards to the University of Florida KL1 RR029888 and UL1 RR029890. Additional support was provided by the Batchelor Foundation.
