Abstract
In animals, salmonellosis is seen typically as enteritis and/or septicemia. Subclinical infection also occurs, and outwardly healthy animals can serve as reservoirs of infection. Reports of salmonellosis in elephants are rare, limited to a few serovars, and the gross and microscopic lesions of enteric salmonellosis in this species have not been described in detail. We present here, in 2 elephants in managed care settings, cases of salmonellosis that resulted from infection by Salmonella enterica serovar Muenchen and S. enterica serovar Montevideo, serovars that have not been described previously as the cause of salmonellosis in elephants, to our knowledge. We also review the literature on salmonellosis in elephants. Animal A, an adult Asian elephant that was euthanized because of gastrointestinal hemorrhage, had multifocal, necrotizing, suppurative enterocolitis, and necrotizing gastritis. Animal B, an adult African elephant with chronic, recurrent colic, followed by death, had necrotizing typhlocolitis. The origin of infection was not determined in either case. The animals came from different facilities and did not have a common feed source. Previously reported cases of salmonellosis in elephants were caused by Salmonella Dublin, Salmonella Typhimurium, or Salmonella Enteritidis. The definitive diagnosis of salmonellosis is made based on compatible gross and microscopic lesions, coupled with the detection of Salmonella spp. in the affected tissues. Effective biosecurity should be adopted to minimize the risk of salmonellosis in elephants in managed care.
Keywords
Salmonellosis is caused by Salmonella spp., gram-negative, facultative microaerophilic rods. Two species of Salmonella are known: Salmonella enterica and Salmonella bongori. 18 S. enterica is composed of > 2,000 serovars; S. bongori has 10 serovars. The taxon of the serotype is based on the somatic, capsular, and flagellar antigens (O, Vi, and H, respectively).5,18 The terms serovar and serotype are used interchangeably, but serovar is used in the Kauffman–White scheme and is the term used frequently by the Association of Public Health Laboratories and the World Health Organization.1,18 The name of the serovar comes from the location of first isolation or from a specific clinical syndrome in a given host.
Many animals carry salmonellae without showing clinical signs, although they can shed salmonellae and infect other individuals. The development of clinical signs depends on several factors, including stress, concurrent infection with other microorganisms, and the serotype infecting a particular animal species. For instance, Salmonella Typhi and Salmonella Paratyphi produce typhoid in people, whereas Salmonella Gallinarum produces a similar disease in poultry; Salmonella Abortusovis causes abortion in sheep, Salmonella Choleraesuis produces enteric disease and septicemia in pigs, and Salmonella Dublin causes a similar syndrome in cattle. The most common clinical presentations of salmonellosis in all animal species are enteritis and septicemia, the latter including various outcomes, such as pneumonia, meningitis, arthritis, and abortion.7,22,23
Salmonella spp. infections in elephants have been reported to have been caused by Salmonella Dublin, Salmonella Typhimurium, and Salmonella Enteritidis.8,10,12,15 Detailed descriptions of lesions associated with Salmonella spp. infection in this species are lacking. Here, we describe cases of salmonellosis caused by Salmonella Muenchen and Salmonella Montevideo, respectively, in 2 elephants in managed care. We also reviewed the literature of salmonellosis in elephants based on searches in PubMed, Web of Science, Scopus, and Google from pathogenesis to the described macroscopic and microscopic lesions of salmonellosis in elephants.
Two elephants were submitted for autopsy ~8 h after death to the Tulare and San Bernardino branches of the California Animal Health and Food Safety Laboratory (CAHFS), University of California–Davis (UCD), Davis, CA, USA. Animal A was a 48-y-old female Asian elephant that was euthanized because of gastrointestinal hemorrhage of unknown cause. Animal B was a 30–40-y-old female African elephant, with a clinical history of recurrent colic and acute lethargy, followed by recumbency and death.
On gross examination of the carcass of animal A, the mucous membranes were markedly pale. The gastric wall was severely thickened, the mucosa had many red ulcers throughout, and the serosa along the greater curvature was covered by a large amount of mucoid, yellow exudate. The small and large intestine were diffusely congested, and the mucosa was multifocally ulcerated and diffusely covered by a yellow and velvety adherent friable pseudomembrane (Fig. 1). The contents of the small and large intestine were liquid, yellow, and foul smelling.
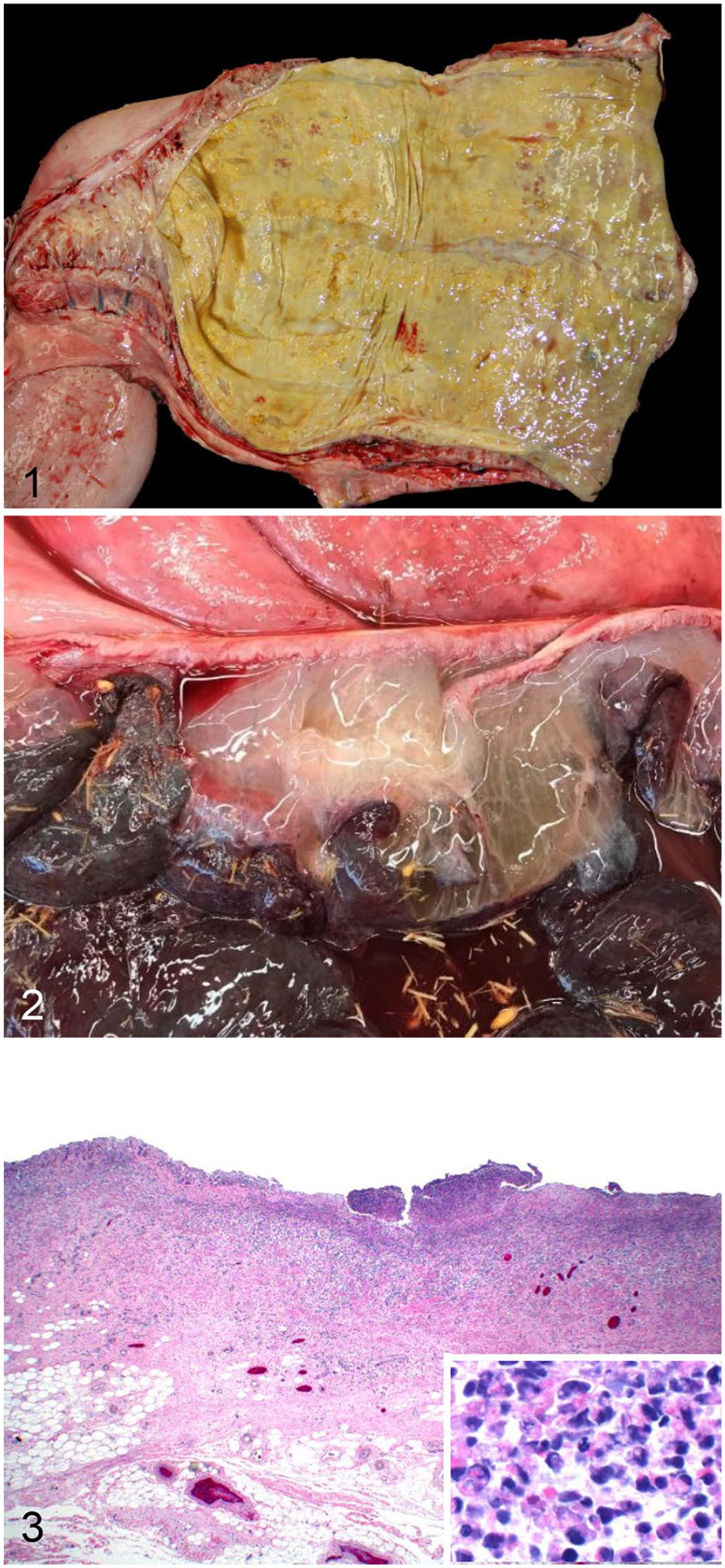
Intestine of 2 elephants with salmonellosis.
In animal B, the gastric mucosa was congested, and the small intestine had many ~2.5-cm, well-demarcated, dark-red ulcers. The cecal mucosa was diffusely congested, and the submucosa of the cecum and proximal colon were expanded by abundant colorless gelatinous edema (Fig. 2). The small and large intestinal contents were watery and red. The ileocolic lymph nodes were mildly enlarged. No other significant gross lesions were observed in either carcass.
From both animals, samples of lung, liver, kidney, spleen, lymph nodes, heart, adrenal gland, skeletal muscle, stomach, small and large intestine, and brain were collected and fixed in 10% neutral-buffered formalin (pH 7) for 48 h, dehydrated through graded alcohols to xylene, embedded in paraffin, sectioned at 4–6 µm, and stained with H&E. Selected sections of the small and large intestine were stained with Brown–Hopps Gram stain.
Microscopically, the intestinal epithelium in animal A was absent-to-necrotic, and covered by a thick pseudomembrane of fibrin, large numbers of neutrophils, fewer lymphocytes and histiocytes (Fig. 3), and colonies of gram-negative coccobacilli. Fibrinous exudate dissected the mucosa, submucosa, muscularis, and serosa. The underlying lamina propria was moderately expanded by neutrophils and fewer lymphocytes, plasma cells, and histiocytes. Occasionally, the crypts were dilated and contained neutrophils and necrotic cellular debris; infiltrates of neutrophils and lymphocytes extended through the muscularis and submucosa. Animal A also had necrotizing and pleocellular gastritis, with severe fibrosis, transmural edema, and diffuse mucosal hemorrhage. In the small intestine, cecum, and colon of animal B, the superficial mucosa was covered by a pseudomembrane of fibrin, RBCs, cell debris, and numerous short gram-positive and -negative bacilli. The submucosa was diffusely expanded by abundant flocculent pale eosinophilic material, and lymphatic vessels were dilated. Throughout the mucosa, lumens of several vessels were occluded by fibrin thrombi. In the small intestine, there was blunting of villi and loss of crypts. No other significant microscopic lesions were observed in any of the tissues of either elephant.
Salmonella spp. cultures using 18–20-h incubation in tetrathionate enrichment broth (Vet Med Biological Media Services, UCD) were set up with samples of liver, lymph node, small intestinal swab, and colon swab from animal A, and intestinal contents, cecal swab, mesenteric lymph node, and kidney from animal B. Salmonella sp. group C2 was isolated from liver, mesenteric lymph node, small intestine, and colon from animal A. Salmonella sp. group C1 was isolated from the mesenteric lymph node and small intestine of animal B. Serotyping was performed on the Salmonella sp. isolates as described previously.1,18 The isolates from animals A and B were typed as Salmonella Muenchen and Salmonella Montevideo, respectively. In addition, a real-time PCR assay 3 detected a fragment of the Salmonella-specific invA gene in liver, lymph node, small intestine, and colon of animal A.
Samples of liver, lung, mesenteric lymph nodes, small intestine, and colon, and small intestinal and colonic contents were aseptically collected from both animals. In addition to the Salmonella spp. enrichment culture described above, samples of stomach were collected from animal A, and cecum and kidney tissue samples were collected from animal B. These samples were inoculated onto Columbia 5% sheep blood agar (Hardy) and MacConkey agar plates (Hardy) and incubated in 7% CO2 at 37°C for 48 h. Small intestine and colon from both animals, stomach from animal A, and kidney and cecum from animal B were cultured anaerobically on Brucella and phenylethyl alcohol blood agars (Anaerobe Systems) and incubated for 72 h in an anaerobic environment at 35–37°C. Clostridioides difficile culture using cycloserine–cefoxitin–fructose agar medium (Thermo Scientific) was also performed on small intestine and colon contents from both animals. Yersinia spp. culture using cefsulodin–irgasan–novobiocin agar (Thermo Scientific), with 3 weekly subcultures from inoculated PBS broth stored in the refrigerator, were performed on small and large intestinal contents from both animals and from lymph node from animal A. Citrobacter freundii and Escherichia coli were isolated from stomach, small intestine, and colon contents of animal A. C. perfringens was isolated from the small intestinal contents of animals A and B, and from colon contents and kidney of animal B. The C. perfringens isolates were characterized as type A by PCR assay as described previously. 2 No other microorganisms were isolated from any of the samples cultured.
In both animals, samples of small intestine, colon, and cecum contents were assayed for C. perfringens toxins alpha, beta, and epsilon, as well as enterotoxin, using 2 commercial capture ELISA kits (Bio X and Techlab; respectively). Aliquots of the same intestinal content samples from animal A were also processed for C. difficile toxins A and B using a commercial ELISA kit (Techlab). The 3 ELISAS were performed according to the manufacturers’ instructions and were negative for all of the toxins tested.
Fecal flotation did not reveal any evidence of intestinal parasitism in either animal. Heavy metal screening on liver of both animals using inductively coupled plasma–atomic emission spectroscopy was unremarkable.
A diagnosis of salmonellosis was established in both cases based on the gross and microscopic lesions coupled with isolation of Salmonella spp. from affected tissues and bodily fluids of these animals. Isolation of Salmonella spp. from intestinal contents alone is not sufficient for a diagnosis of salmonellosis given that individuals can be asymptomatic carriers of the pathogen. 8 The role of other intestinal pathogens in the enteric disease of these elephants was considered less likely based on culture, clostridial toxin ELISA, and fecal flotation results.
The colonization of the intestinal mucosa by Salmonella sp. begins along crypt M cells that lack a surface mucus layer.5,22 In other areas of the intestinal mucosa covered by mucus, Salmonella spp. use flagella for penetration.13,22 Following cell penetration, ligands and receptors interact.13,14 The initial adherence of Salmonella spp. has been hypothesized to be mediated by fimbriae, and it has therefore been proposed that this virulence factor defines the susceptibility of the various strains of Salmonella spp.13,14 After the pathogen has adhered to a cell membrane, a type III secretion system is used to inject molecules that stimulate phagocytosis,14,22 forming the so-called Salmonella-containing vacuoles. The bacterium inhibits phagosome–lysosome fusion, preventing bacterial killing by macrophages. The bacteria can reside and replicate in a phagolysosome or phagosome. After they are infected by the bacteria, macrophages transport the microorganism to mesenteric lymph nodes via lymphatic circulation or to the liver through the portal vein.5,6,22,23 A type I secretion system is used by Salmonella spp.9,14 to kill the macrophages via pyroptosis, specifically by the activation of caspase-1 in macrophages.6,9,19
Cases of salmonellosis reported previously in elephants were caused by Salmonella Dublin, Salmonella Typhimurium, and Salmonella Enteritidis.4,8,10,15,16,20,21,24,25 To our knowledge, Salmonella Muenchen and Salmonella Montevideo have not been associated with salmonellosis in elephants before. The cases of salmonellosis reported previously in elephants in managed care were associated with immunosuppression linked to environmental and biological stressors such as transportation, calving, and overcrowding.4,15 In our cases, there was no known history of stressors, and the predisposing factors remain, therefore, undetermined. The gross lesions described in cases of enteric salmonellosis in elephants have ranged from redness of the distal small intestinal or colonic mucosa to catarrhal or fibrinohemorrhagic enteritis and colitis.4,8,10,12,15,20,21,24,25 In our 2 cases, the intestinal lesions were fibrinohemorrhagic, necrotizing, and severe, which could be a consequence of the immune status of the animals or inherent to the virulence of the Salmonella serovars involved.
The cases of salmonellosis described previously in elephants included lesions both in the intestinal tract and in extra-intestinal organs, indicative of sepsis. Among the latter, the liver and spleen were the 2 main organs affected, showing necrosis and inflammation. 16 In our cases, the lesions were restricted to the intestinal tract, although Salmonella sp. was isolated from several extra-gastrointestinal organs, suggesting septicemia. Gastric lesions are not typical of salmonellosis in any animal species and, to our knowledge, they have not been described in elephants before. 16 The ulcerative gastritis observed in one of our elephants could have been associated with reflux from the heavily contaminated small intestine. Because Salmonella sp. was not isolated from the stomach, we cannot confirm that gastritis was another manifestation of salmonellosis in this elephant. The lack of lesions in infected extra-intestinal organs from which Salmonella spp. were isolated suggests that dissemination of the infection outside the intestinal tract was a terminal event with no time for lesions to develop in those organs, including the stomach.
Various Salmonella serotypes have been isolated from animals of several species in managed care that had been taken recently from the wild. 16 Such animals were suspected to have already been infected when collected, but confirmation of the origin of infection was not available. To avoid contamination of holding facilities when recently collected animals are introduced, a full diagnostic workup, including Salmonella spp. testing, and quarantine before introduction, is recommended. In our cases, the animals had been in managed care for their entire lives, and there was no history of introduction of new arrivals from the wild or elsewhere in the recent past.
Most of the cases of salmonellosis that occur in wildlife are associated with environmental contamination from untreated or poorly treated sewage, and from contaminated feces of humans and domestic animals in close contact with wildlife.16,17 Most outbreaks of salmonellosis in animals in managed care are believed to be associated with water or feed contamination.24,25 In our 2 cases of salmonellosis, the animals did not share a common food source and were from different, geographically distant facilities at which each animal had been a long-term resident. No information was available indicating if these animals had been housed with other elephants, nor if they had been tested previously for shedding of Salmonella spp. The origin of the infection remains therefore undetermined.
Salmonellosis is an important public health concern. In the managed care setting, risk mitigation should include husbandry practices to reduce contamination and spread of pathogens such as Salmonella spp. Control of pests, including free-ranging rodents and birds that many facilitate dissemination, is important. 11 Regular cleaning and removal of animal waste from enclosures are also essential management strategies to reduce the risk of disease in resident animals as well as to in-contact human caretakers and the public.
Footnotes
Acknowledgements
We thank the UC Davis Veterinary Medicine Teaching and Research Center (VMTRC) team of clinicians, Fernando Ochoa, Paula Yant, Dr. Janet Moore, and Dr. Patricia Blanchard, for their help.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
