Abstract
Elephant endotheliotropic herpesvirus (EEHV) is one of the most important viral infectious diseases affecting the elephant population worldwide, especially juveniles and young adults. We developed a chromogenic in situ hybridization (ISH) test for detection of EEHV in Asian elephants (Elephas maximus). Digoxigenin (DIG) DNA probes from the polymerase and terminase genes of EEHV were synthesized using a PCR DIG-labeling method, and detection of hybridized probe to target EEHV DNA was carried out by anti-DIG immunolabeling. Distribution of EEHV-1A and EEHV-4 genomes was found to be prominent in mononuclear phagocytic cells of spleen and endothelial cells of visceral organs. ISH enables the detection of EEHV infection and has applications in understanding pathogenesis of EEHV in Asian elephants.
Elephant endotheliotropic herpesvirus (EEHV; order Herpesvirales, family Herpesviridae, subfamily Betaherpesvirinae, genus Proboscivirus, species Elephantid betaherpesvirus 1) is one of the most important viral infectious agents in elephants worldwide.3,8,10,15,21 EEHV has been classified into 8 genotypes, including EEHV-1A, EEHV-1B, and EEHV-2–7.5,6,19,23 Some genotypes of EEHV have been reported to infect Asian elephants (Elephas maximus), but not African elephants (Loxodonta africana).7,8,13,14,18,19,21 Elephants affected clinically by EEHV infection may develop lethargy and cardio-vascular problems, such as edema and hemorrhages in the visceral organs, for which the exact pathogenesis remains unclear.1,7,10,11,14
Propagation of EEHV in a cell culture system has not been successful.4,7,10 Detection of EEHV is generally based on clinicopathologic findings, PCR and gene sequencing, or ELISA.4,9,10,17,19,22 However, absence of EEHV antibodies in EEHV cases by ELISA does not guarantee the absence of EEHV infection, because only 80% of PCR-positive cases showed seroconversion by ELISA. 17 Tests to directly detect EEHV genomes are usually used to confirm the presence of EEHV in elephants or identify shedders of EEHV in the elephant’s herd.3,4,9,10,22 Although PCR results are not sufficient to describe the pathogenesis underlying the disease, this technique is vital to understanding disease transmission.3,12,15 To explain the complete pathogenesis of the disease caused by EEHV, further methods are needed.
Previously, a study of EEHV genome distribution by real-time PCR demonstrated that viral DNA was detected in all of the visceral organs tested, with the highest viral load in the heart, tongue, and liver. 12 However, it remains to be determined whether EEHV exhibits tropism for inflammatory cells, endothelial cells, or parenchymal tissue, given that DNA extraction for PCR analysis was done on whole tissue lysates. Furthermore, variation in the lesions sampled and the amount of EEHV-infected cells obtained for PCR testing might influence the quantity of viral genome. The viral load detected in each organ by PCR might not be suitable to explain the pathogenesis of the disease. An alternative method to overcome this limitation is of great interest. We developed and validated an in situ hybridization (ISH) technique for detection of EEHV infection in Asian elephants to aid in a better understanding of viral pathogenesis.
Formalin-fixed, paraffin-embedded (FFPE) tissues from captive (private elephant camps) EEHV-infected Asian elephants aged 4 y (case 1) and 5 y (case 2) were included in the study. The cause of death was diagnosed as infection with EEHV-1A and EEHV-4, respectively, using PCR and gene sequencing, as described below. Tissue samples from a 1-d-old (stillborn) elephant (case 3), which tested negative for EEHV infection by PCR, were used in our study as a negative control (Table 1). Tissue samples were fixed in 10% buffered formalin and embedded in paraffin. Histologic staining was performed on 4-μm thick sections with hematoxylin and eosin; an additional 4 or 5 sections per block were cut and mounted on (3-aminopropyl) triethoxysilane-coated slides for ISH testing.
Summary of elephant history included in our study of in situ hybridization for detection of elephant endotheliotropic herpesvirus (EEHV) in Asian elephants.

To verify EEHV infection in the studied animals, viral DNA was extracted from frozen heart tissue (DNA extraction kit, Macherey-Nagel, Dauren, Germany) in accordance with the manufacturer’s recommendations. The oligonucleotide primers of polymerase and terminase genes used in our study were as described previously. 10 Conventional PCR was performed under the following conditions: 95°C for 2 min, 35 cycles of 95°C for 30 s, 55°C for 30 s, and 72°C for 1 min, and a final extension at 72°C for 2 min. PCR products were analyzed by electrophoresis, and specific bands were observed under an ultraviolet illuminator. PCR products were then further analyzed by DNA sequencing, as described previously. 14
DNA probes for EEHV were prepared from the tissue samples of case 1 (PCR DIG probe synthesis kit, Roche, Basel, Switzerland), according to the manufacturer’s instructions. Briefly, the PCR test was carried out under the conditions described earlier, with primers derived from polymerase and terminase genes, except instead of the addition of nucleotides, a digoxigenin (DIG)-labeled nucleotide was used. Validation of the hybridization probe was performed with 1.5% agarose gel electrophoresis followed by southern blot immunolabeling. Chromogenic DNA ISH was performed according to a previous published protocol, 2 with minor modification. Briefly, 4-µm thick FFPE sections were deparaffinized with xylene and subsequently rehydrated in absolute ethanol, 90%, 70%, and 50% ethanol, and water, respectively, then rinsed in phosphate-buffered saline (PBS). Thereafter, the sections were treated with proteinase K (200 µg/mL; VWR, Radnor, PA) in prewarmed 20 mM Tris-HCl for 30 min at 37°C. Slides were then washed 3 times in PBS with Tween 20 for 5 min each and in 2× saline–sodium citrate (SSC) solution for 15 min. Slides were prehybridized for 5 min at 95°C in hybridization buffer (Hybridization cocktails II, Bio Basic, Markham, Canada) prior to incubating at 95°C for 10 min with DIG-labeled probes (3 ng/µL) covered by coverslips. Slides were then transferred to a humidified chamber and further incubated for 15 h at 42°C. Then, unhybridized probes were removed by stringency wash (3 times with 2× SSC and 50% formamide for 5 min each, then 3 times with 2× SSC for 5 min each) to minimize nonspecific hybridization. The sections were then incubated for 1 h at 37°C in a 1:200 solution of anti-digoxigenin antibody (Roche) in Tris-buffered saline (TBS). After incubation, slides were washed with TBS 3 times for 5 min each and then incubated with 1:200 horseradish peroxidase–conjugated goat anti-mouse secondary antibody (Vector Laboratories, Burlingame, CA) in TBS for 45 min at room temperature (RT). Next, 3,3’-diaminobenzidine tetrahydrochloride (DAB; KPL, Gaithersburg, MD) was applied for 5 min at RT. Slides were then counterstained with hematoxylin, dehydrated, and coverslipped. Finally, they were examined under a light microscope, and photos were taken. Negative controls were sections of case 1, 2, and 3 that were hybridized with PBS, instead of probes.
Microscopically, samples of the heart, lungs, spleen, lymph nodes, liver, and tongue from the 2 EEHV-positive elephants (cases 1 and 2) were markedly hemorrhagic and infiltrated by lymphocytes and macrophages (Figs. 1, 2). Eosinophilic intranuclear inclusion bodies were prominent in the endothelial cells of visceral organs, including blood vessels in the heart, lung, liver (Supplementary Figs. 1–3, arrows), and submucosal layer of the colon and ileum (Fig. 2; Supplementary Fig. 4, arrows).
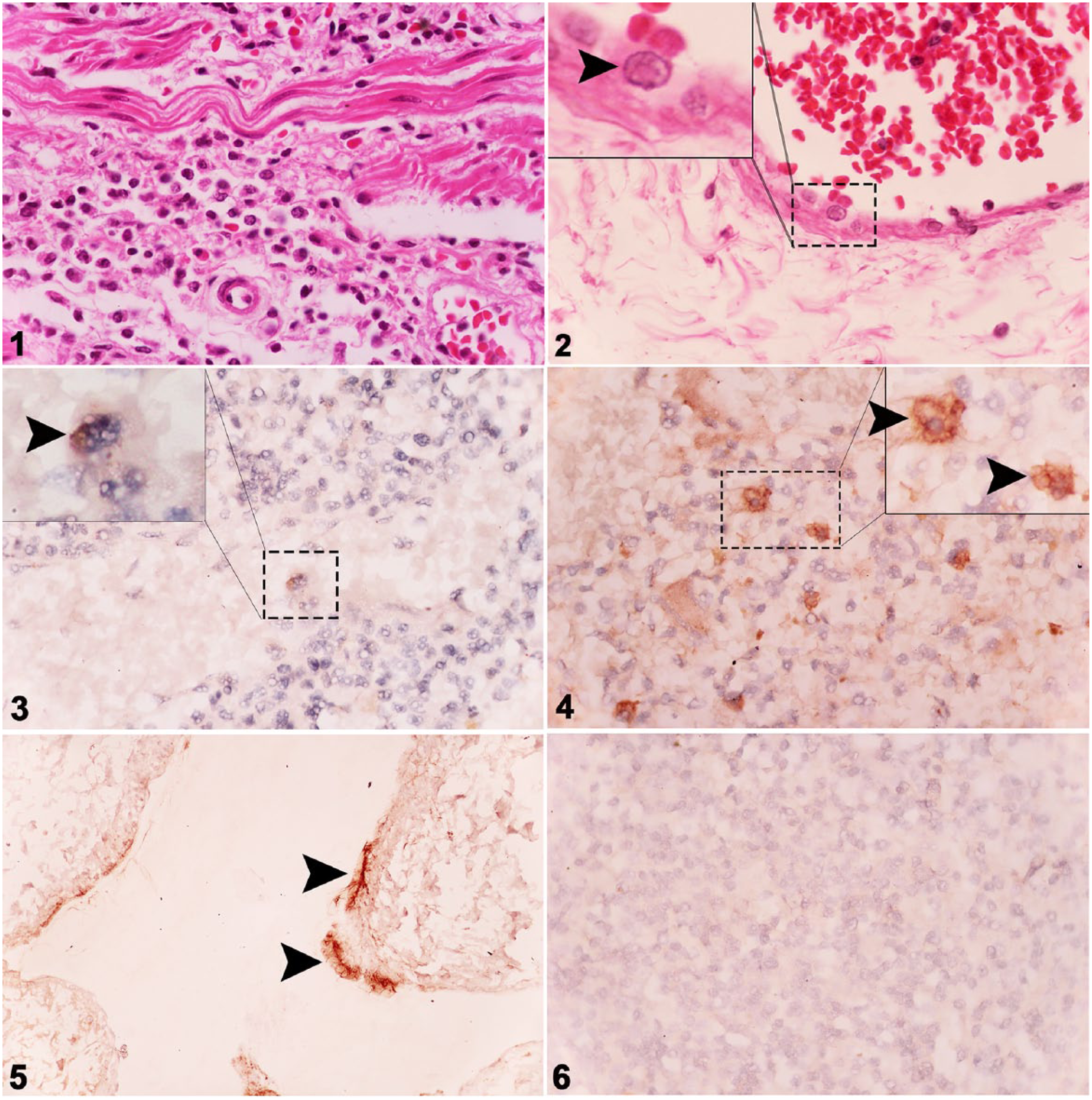
Histologic section from the colon of an Asian elephant (case 2) infected with elephant endotheliotropic herpesvirus (EEHV)-4, with hemorrhage and lymphohistiocytic inflammation of the lamina propria. H&E.
The negative control animal (case 3) was negative for EEHV infection by both histologic examination and PCR (data not shown). Chromogenic ISH of EEHV genomes from cases 1 and 2 revealed genome distribution throughout the visceral organs; tissue samples from case 3 were negative by ISH (Table 2). The occurrence and greater intensity of signals were observed mainly in mononuclear phagocytic cells and endothelial cells of the spleen, heart, and liver, and to a lesser extent in the lung, kidney, and intestine (Figs. 3–5). Furthermore, the signals generated by the probes hybridizing to the polymerase and terminase genes were shown to be similar in organ distribution and signal intensity (Table 2). Case 3 tested negative for EEHV genome by ISH (Fig. 6).
Detection of elephant endotheliotropic herpesvirus genomes by in situ hybridization in selected organ tissues.

NA = not available; Pol. = polymerase gene; Ter. = terminase gene. Occurrence and intensity of signal: – = no signal; + = mild; + + = moderate; + + + = high.
Our chromogenic ISH test for detection of EEHV genome revealed that the viral genome was distributed predominantly in the peripheral blood mononuclear cells (PBMCs) and endothelial cells of the spleen, liver, and heart. Given that alphaherpesviruses in humans and animals usually infect epithelial cells, while being disseminated systemically through PBMCs,16,20 our findings might point to the important role of PBMCs underlying the pathogenesis of EEHV infection in Asian elephants. To clarify this point, however, further studies are needed.
Supplemental Material
DS1-4_JVDI_10.1177_1040638718773810 – Supplemental material for Development of in situ hybridization for detection of elephant endotheliotropic herpesvirus in Asian elephants
Supplemental material, DS1-4_JVDI_10.1177_1040638718773810 for Development of in situ hybridization for detection of elephant endotheliotropic herpesvirus in Asian elephants by Varankpicha Kochakul, Kittikorn Boonsri, Saruda Tiwananthagorn, Chalermchart Somgird, Chatchote Thitaram, Kidsadagon Pringproa in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank P Tankaew, from the Veterinary Diagnostic Laboratory, Faculty of Veterinary Medicine, Chiang Mai University, for excellent technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Center of Excellent in Elephant Research and Education, Chiang Mai University, Chiang Mai, Thailand.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
