Abstract
The European Community’s leukosis key (EC key) is a well-known hematologic method for detecting bovine leukemia virus (BLV) infection in dairy cattle. The key identifies infected cattle with persistent lymphocytosis via a combination of lymphocyte count (LC) and age. Using the EC key to identify BLV-infected Japanese black (JB) cattle is problematic, however, given the inherently lower LCs of JB cattle compared to dairy cattle. We analyzed the LC in BLV-positive and -negative JB cattle and estimated LC cutoff values by age using receiver operating characteristic curve analysis. Among the 716 JB blood samples collected, 452 (63%) JB cattle were confirmed as BLV-positive by an antibody ELISA for ≥1-y-old cattle and by real-time PCR for <1-y-old cattle. The cutoff values for the LC in each age group were calculated as 6.3 × 109/L for <1 y, 5.9 × 109/L for 1 to <2 y, 5.5 × 109/L for 2 to <3 y, 4.5 × 109/L for 3 to <6 y, 4.3 × 109/L for 6 to ≤10 y, and 3.7 × 109/L for >10 y. The sensitivity and specificity of the estimated cutoff values were 0.49 (95% confidence interval: 0.44–0.53) and 0.81 (0.75–0.85), whereas those of the EC key were 0.20 (0.16–0.24) and 0.99 (0.97–1.00). Our LC cutoff values for screening JB cattle for BLV infection appear to be preferable to those of the EC key.
Enzootic bovine leukosis (EBL) is caused by bovine leukemia virus (BLV; family
Persistent lymphocytosis (PL), a chronically increased lymphocyte count (LC) with no associated overt clinical signs, is one of the major clinical characteristics of BLV-infected cattle. Approximately 30% of BLV-infected cattle that have anti-BLV antibodies develop PL,1,7 and cattle with PL have the potential to transmit BLV given their high BLV proviral loads.6,14 PL is also known to induce morbidity characterized by weakness and opportunistic infections as a result of a compromised immune system. 4 The European Community’s leukosis key (EC key) is a well-known hematologic test for BLV infection.8,9 In European countries with major dairy farming industries, the EC key has been used to detect PL in cattle in order to control and ultimately eradicate BLV infection.2,3
EBL is a problem in Japan; seroprevalences of ~40% and 30% have been reported in dairy and beef cattle, respectively. 10 In beef cattle farms, economic loss caused by BLV infection is a serious issue in Japan because cattle diagnosed with EBL lymphomas are not allowed to be slaughtered for consumption. For the prevention of among-farm and within-farm spread of BLV, there is a strong need for effective detection of BLV infection in beef farms. Because determination of LC is a simple and rapid method, LC has been routinely used to screen for PL in Japan. However, in the case of Japanese black (JB) cattle, which is a popular beef breed in Japan with ~1.6 million JB cattle raised and accounting for 42% of the Japanese cattle population (Ministry of Agriculture, Forestry, and Fisheries [MAFF]. Livestock statistics 2017 [Japanese]. Available from: http://www.maff.go.jp/j/tokei/kouhyou/tikusan/index.html), the numbers of white blood cells (WBCs) have been reported to be lower than in Holstein cattle.9,12 Thus, the EC key—which was devised based on the number of lymphocytes in dairy cattle—may be ill-suited for screening for high LCs in JB cattle. Therefore, we investigated the LC cutoff value for JB cattle to facilitate effective screening of PL cattle.
We conducted our study on JB cattle breeding farms in the Ibaraki Prefecture, located in eastern Japan, in which ~50,000 beef cattle (including both fattening cattle and breeding cattle) are raised on 606 farms (MAFF, Livestock statistics 2017 [Japanese]). As part of their BLV control activities, local veterinary officers collected blood samples from 716 JB cattle from 40 JB cattle breeding farms between February 2016 and June 2017. The farms were selected on a voluntary basis, and all breeding JB cattle and their newborn calves were inspected on each farm. Blood samples were collected from animals using potassium-EDTA blood collection tubes for blood cell counts and real-time PCR (rtPCR) tests, as well as serum tubes containing a clot activator for serologic tests. The samples were stored at 4°C; blood cell counts and serum separation were conducted on the day of sampling. WBC and lymphocytes were counted in whole blood samples using an automated veterinary multi-parameter hematology analyzer (Poch-100iV Diff; Sysmex, Kobe, Japan).15,17 Serum samples were separated (centrifuged at 1,710 ×
DNA was extracted from EDTA whole blood samples on the day of collection (automated nucleic acid extraction system, magLEAD12gC; magDEA Dx extraction kit; Precision System Science, Chiba, Japan). Real-time PCR for detection of the BLV
Cattle <1-y-old were defined as BLV-positive if they exhibited >0 BLV proviral copies/10 ng DNA, given the presence of maternal antibodies, which are present up to 6 mo of age. 13 Cattle ≥1-y-old were defined as BLV-positive if they were BLV antibody positive. The LC distributions in BLV-positive and -negative cattle in various age groups were compared using the Mann–Whitney U test. The correlation between the proviral load and the LC was examined via the Spearman rank order test.
For effective detection of PL in BLV-infected JB cattle, we determined LC cutoff values at which false-negative results are minimized, using receiver operating characteristic (ROC) curve analysis. Sensitivity was represented by the proportion of BLV-positive cattle that were correctly identified, and specificity was represented by the proportion of BLV-negative cattle that were correctly identified. Cutoff values were calculated using the sensitivity and specificity values that maximized the Youden index (sensitivity + specificity − 1).5,19 Because JB breeding cattle have a long productive life, we modified the age group from that of the EC key by adding older age groups (>6 y): the age groups were <1 y, 1 to <2 y, 2 to <3 y, 3 to <4 y, 4 to <5 y, 5 to <6 y, 6 to ≤10 y, and >10 y. LC cutoff values were calculated for each age group.
Because BLV-positive cattle >1-y-old were identified based on the antibody test, it might be rarely possible that false-negative results were obtained during the early phase of infection. Thus, as a complementary study, we conducted a simple simulation to validate the sensitivity and specificity of LC cutoff values. We assumed that a false-negative result for the antibody test in the early phase of infection occurred with a probability of 6%, based on the results of a previous study. 11 We randomly selected BLV-negative cattle ≥1-y-old (199 cattle) with a probability of 6% and re-defined them as BLV-positive cattle. Using the re-defined infection status of cattle, we estimated the LC cutoff value via ROC analysis, then calculated sensitivity and specificity. A total of 1,000 iterations were performed. All statistical analyses were performed using R software (v.2.15.3; https://www.r-project.org/).
Of the 716 JB cattle tested, 452 (63%) were confirmed as BLV-positive (Supplementary Table 1). The number of lymphocytes in BLV-positive cattle (median: 4.6 × 109/L; 5th–95th percentile: 2.3–9.7 × 109 lymphocytes/L) was significantly higher than the number in BLV-negative cattle (median: 2.3 × 109/L; 5th–95th percentile: 2.2–7.3 × 109/L;
The estimated cutoff values (Table 1, Fig. 1) were 47–57% lower than the positive thresholds of the EC key. The sensitivity and specificity based on the estimated cutoff thresholds were 0.49 (95% confidence interval [CI]: 0.44–0.55) and 0.81 (95% CI: 0.75–0.85). The sensitivity of the EC key was lower (0.20; 0.16–0.24) than that of our estimated cutoff, and its specificity was 0.99 (0.97–1.00). The results of the simulation provided almost the same values of sensitivity and specificity as the estimated values (Supplementary Table 3, Supplementary Fig. 2). Thus, the estimated sensitivity and specificity of our cutoff values were deemed to be robust. Based on the classification by the estimated cutoff value, the distribution of the proviral load (median: 259; 5th–95th percentile: 0.09–637 copies/10 ng DNA) was significantly higher in lymphocytotic cattle than in non-lymphocytotic cattle (median: 14; 5th–95th percentile: 0–330 copies/10 ng DNA;
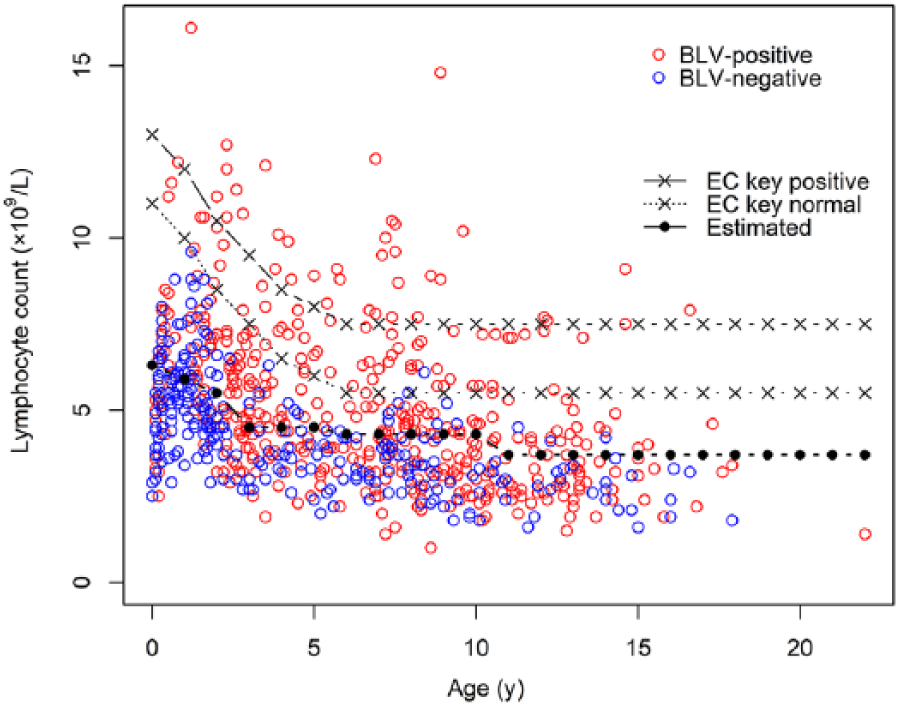
Scatter plot of lymphocyte counts (cells × 109/L) by age (y). Red and blue circles represent BLV-positive and -negative cattle, respectively. EC key positive = positive values derived from the European Community’s leukosis key; EC key normal = negative values derived from the EC key; Estimated = estimated cutoff values.
We aimed to optimize the LC cutoff value for BLV-infected JB cattle to facilitate effective screening for PL. A higher concentration of lymphocytes was detected in BLV-positive JB cattle than in BLV-negative JB cattle. The sensitivity of the cutoff values determined via ROC analysis was considerably higher than that of the EC key. This suggests that applying the EC key in JB cattle is highly likely to result in some infected cattle going undetected. This concern has also been pointed out, and a hematologic key for BLV infection in JB cattle has been suggested 9 ; when applied to our LC data, sensitivity was 0.39 (95% CI: 0.34–0.45) and 0.55 (0.51–0.60) when suspected cases were excluded or included as lymphocytotic, respectively. The sensitivity of 0.49 (0.44–0.53) of our cutoff values, in detecting lymphocytotic JB cattle, is similar to the previously developed cutoff value, 9 and either of these methods is preferable to the use of the EC key in screening for lymphocytosis in JB cattle. However, the specificity was slightly reduced in comparison with that of the EC key when using our proposed cutoff values. Considering the trade-off between sensitivity and specificity, reduced specificity would be unavoidable when sensitivity is improved. However, the specificity of our cutoff values was >80%, which justifies their use as a BLV screening tool, considering that the diagnosis of BLV is confirmed by more definitive laboratory tests such as ELISA and rtPCR. It should be noted that our approach of using the proposed cutoff value does not consider the relative cost of false positives versus false negatives. 16 In the case of a farm with a low prevalence, the financial cost to the farmer of any false positives must be considered.
Applying our cutoff values for BLV in JB cattle farms enables the effective detection of JB cattle with lymphocytosis, although ELISA and rtPCR are required for confirmation of current infections. On farms with BLV-infected cattle, our cutoff values will assist in screening for JB cattle with PL, and such evidence-based planning will encourage farmers to take active control measures against BLV.
Supplemental Material
DS1_JVD_10.1177_1040638719848449 – Supplemental material for A hematologic key for bovine leukemia virus screening in Japanese black cattle
Supplemental material, DS1_JVD_10.1177_1040638719848449 for A hematologic key for bovine leukemia virus screening in Japanese black cattle by Masataka Akagami, Shoko Oya, Yuki Kashima, Satoko Seki, Yoshinao Ouchi and Yoko Hayama in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank all of the livestock owners for their cooperation in this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grant projects related to the promotion of regional research and development by the Ministry of Education, Culture, Sports, Science and Technology in Japan.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

