Abstract
Six, 5–6-wk-old pigs, from 3 farms of the same company, with significant loss of body condition were submitted for postmortem evaluation. Macroscopically, the main lesion observed in all of the pigs was thymic atrophy. Microscopically, all of the pigs had thymic atrophy, superficial lymphocytic fundic gastritis, atrophic enteritis, superficial colitis, and neutrophilic and lymphocytic rhinitis, leading to a diagnosis of porcine periweaning failure-to-thrive syndrome. In the pigs from 2 of the farms, many of the thymic corpuscles had infiltrates of neutrophils and degenerate cells, in some cases infiltrating the surrounding parenchyma.
Porcine periweaning failure-to-thrive syndrome (PFTS) is a clinical condition affecting nursery pigs that occurs within 2–3 wk of weaning. Affected animals exhibit anorexia, lethargy, and progressive debilitation, and repetitive oral behavior such as chomping and licking despite the absence of infectious, nutritional, management, or environmental factors. 5
Upon gross examination, affected pigs appear emaciated, the thymus is severely atrophic, and the gastrointestinal tract is empty. Characteristic histologic lesions include thymic atrophy, superficial lymphoplasmacytic fundic gastritis, villus atrophy in the small intestine, superficial colitis, lymphocytic and neutrophilic rhinitis, and mild nonsuppurative meningoencephalitis.5,6 A diagnosis of PFTS should be considered if the debilitated piglet has all 3 histologic lesions: superficial gastritis, small intestinal villus atrophy, and thymic atrophy. 8
The etiology of the disease remains unclear, and attempts at experimental reproduction using tissue homogenates have failed, suggesting that the syndrome may not have an infectious etiology. 7 Individual genetic susceptibility to this syndrome has been reported. 12 Genomic regions containing genes that have been linked to phenotypes associated with the syndrome, such as depression and intestinal villus atrophy, have been identified in PFTS-affected piglets.1,15
Six piglets, from 3 different farms (A–C) of the same company, with significant loss of body condition were brought to the Faculty of Veterinary Medicine of the University of Murcia (Spain) for postmortem evaluation. The piglets from farm A were 6 wk old (2 wk post-weaning), and 5 wk old (2 wk post-weaning) from farms B and C. All piglets had been vaccinated against porcine circovirus 2 (PCV-2) at 3 wk of age.
Piglets were euthanized, and autopsies were performed. Upon gross examination, all 6 animals had thymic atrophy, and the small intestine was flaccid, with walls thin, transparent, and distended. One piglet from farm A had cranioventral consolidation of the lung with fibrin deposits on the pleura; in one piglet from farm C, the lung failed to collapse and had multifocal tan-mottled consolidated areas.
Samples of thymus, lung, small and large intestine, stomach, liver, kidney, spleen, superficial inguinal lymph nodes, tonsil, nasal turbinates, cerebrum, and cerebellum of all animals were fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with hematoxylin and eosin. Immunohistochemistry was performed for PCV-2 (monoclonal antibody [mAb] PCV36A9; Ingenasa, Madrid, Spain) and porcine reproductive and respiratory syndrome virus (PRRSV, species Betaarterivirus suid 1; mAb SDOW17; Rural Technologies, Brookings, SD).
Microscopic examination of all 6 pigs showed thymic atrophy (Fig. 1A), superficial lymphocytic fundic gastritis (Fig. 1B), atrophic enteritis (Fig. 1C), superficial colitis, and neutrophilic and lymphocytic rhinitis. Interstitial pneumonia was seen in 1 pig from farm A, 2 from farm B, and 1 from farm C. Fibrinous bronchopneumonia and hepatic steatosis were observed in one pig from farm A and one from farm B. Lymphocytic meningitis was seen in one pig from farm C (Table 1).
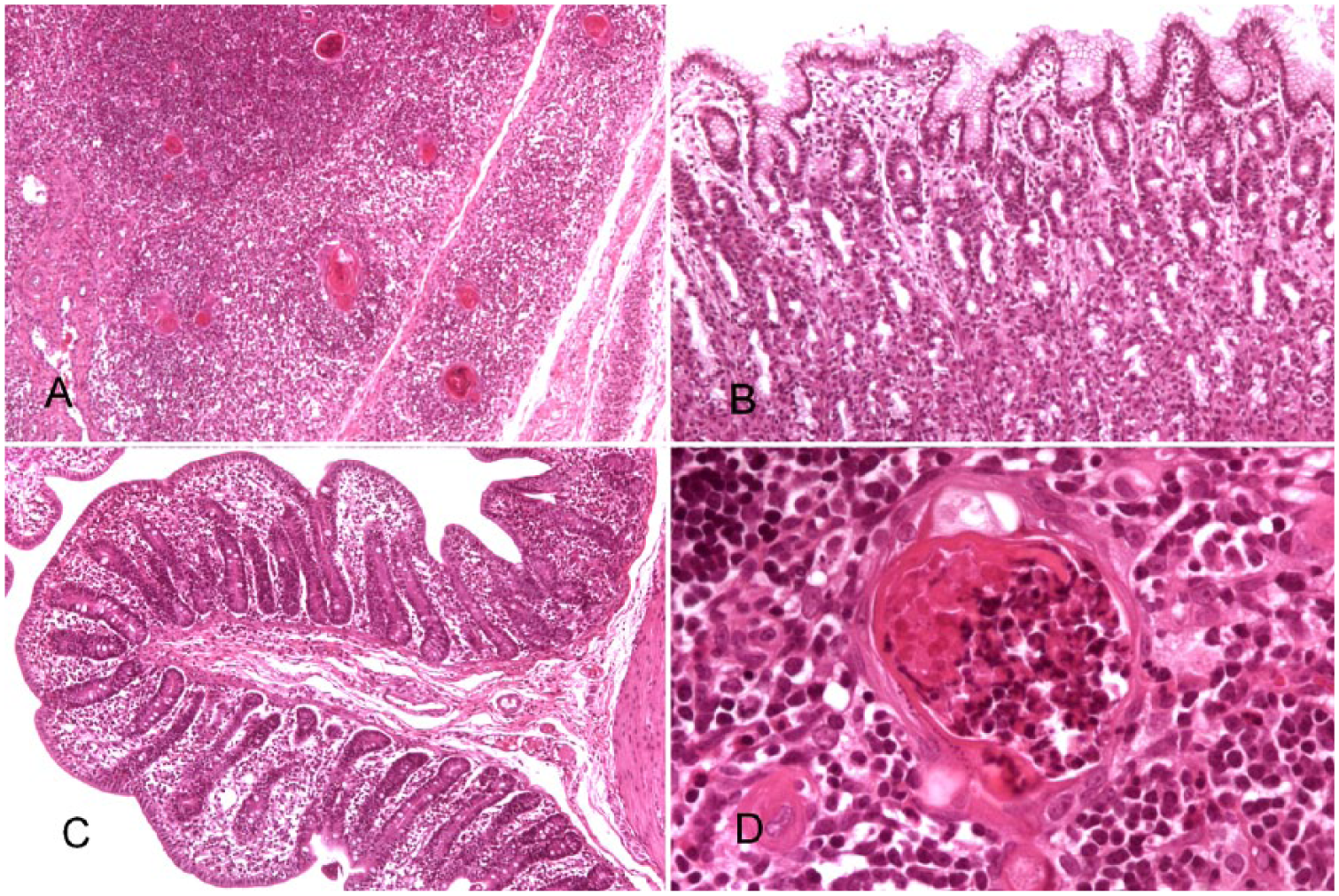
Histologic lesions in piglets with periweaning failure-to-thrive syndrome. H&E.
Microscopic lesions observed in piglets from farms A–C.

= lesion observed; – = lesion not observed.
The thymus of all 6 pigs had severe reduction of cortical thickness. In the pigs from farms A and C, many of the thymic (Hassall’s) corpuscles were variably infiltrated with neutrophils, ranging from a mild infiltrate between the reticular epithelial cells to the corpuscle being full of cellular debris and/or inflammatory cells (Fig. 1D). Occasionally, the corpuscle was ruptured and neutrophils infiltrated the surrounding parenchyma.
Immunohistochemically, dendritic cells were positive for PCV-2 and PRRSV in the tonsils and superficial inguinal lymph nodes of all 6 pigs, and in the lung of 2 pigs from farm B and 1 pig from farm C. No positive immunostaining was seen in the thymus of any animal.
Thymic atrophy is one of the main microscopic lesions needed in a diagnosis of PFTS. 8 This lesion has been described in association with infection by highly pathogenic PRRSV (HP-PRRSV) strains4,10 or classical swine fever virus (CSFV; species Pestivirus C).2,11 In our case, PRRSV-positive cells were detected in the tonsil and lymph node of all pigs and in the lung of 3 of 6 pigs, although positive immunostaining was not detected in the thymus. Infection with HP-PRRSV strains has not been described in Spain to date. Spain is also free of CSFV, and the characteristic lesions of this disease are quite different from those described herein. 2 Other possible causes of thymic atrophy include inadequate nutrition, intoxicants, or iatrogenic causes, 14 although in our case, the set of microscopic lesions observed and the age of affected pigs pointed to a diagnosis of PFTS,5,6 ruling out this list of causes as possible etiologies of the process.
Thymusitis is rare. 3 In pigs, granulomatous thymitis has been reported in animals infected with PCV-2, 9 but in our cases, there were no granulomatous lesions, PCV-2 antigen was not detected in the thymus, and we believe that this thymic lesion is unique. As has been described previously, thymic involution and atrophy have similar histologic changes, 14 and the presence of neutrophils in thymic corpuscles has been associated with involution, 3 However, in our case, the presence of neutrophils and degenerate epithelial cells and neutrophils, which sometimes filled the corpuscles and extended into the adjacent parenchyma when the corpuscles ruptured, and the age of these pigs (5–6 wk) compared to the expected time of thymic involution reported in pigs (~180 d old), 13 suggests a pathologic process consistent with atrophy rather than thymic involution.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
