Abstract
Pathogenic strains of infectious bursal disease virus (IBDV) are associated with increased morbidity, mortality, and immunosuppression in susceptible chickens. Backyard poultry is increasing in popularity in the United States, but very little is known about the prevalence and molecular epidemiology of IBDV within these flocks. We performed a retrospective study and phylogenetic analyses of IBDV detected in backyard chickens (BYCs) submitted to the California Animal Health and Food Safety (CAHFS) diagnostic laboratory system in 2009–2017. There were 17 CAHFS autopsy cases of very virulent IBDV (vvIBDV) segment A detected by RT-rtPCR in BYC flocks from 7 counties in California from 2009–2017. During this same time period, non-vvIBDV genotypes were detected by RT-rtPCR in 16 autopsy cases originating from BYC premises in 10 counties in California. Subsequent RT-PCR and phylogenetic analysis of a segment of the hvVP2 and VP1 gene identified vvIBDV, interserotypic reassortant IBDV (vvIBDV segment A and serotype 2 segment B), and non-vvIBDV (variant/subclinical IBDV and classic IBDV) strains in BYC flocks in California.
Introduction
Infectious bursal disease virus (IBDV; order Tymovirales, family Birnaviridae, genus Avibirnavirus) is a bi-segmented, double-stranded RNA virus. 1 Genome segment A of IBDV encodes for a polyprotein that is eventually cleaved into viral protein 2 (VP2), VP3, and VP4. 16 Genome segment B encodes for VP1, the RNA-dependent RNA polymerase (RdRp). 16 Traditional classification schemes have differentiated IBDV based on either antigenicity or pathogenicity. Based on viral neutralization assays, IBDVs are antigenically differentiated into serotype 1 or serotype 2 viruses, with all pathogenic strains in chickens belonging to serotype 1.1,16 Serotype 2 IBDVs were originally isolated from turkeys and are naturally avirulent viruses in both chicken and turkey hosts. Serotype 1 strains have traditionally been differentiated into classic and variant antigenic subtypes, with very virulent IBDV (vvIBDV) having an antigenic profile that aligns closely with classic IBDV.1,15,16 The occurrence of antigenic drift has been a major factor in the emergence of additional subtypes within the 2 main antigenic groups. 9
Pathogenicity assessments of IBDV have demonstrated that a wide range of virulence exists, and both segment A and segment B of the genome influence pathogenicity.5,6,11,16 Traditionally, serotype 1 strains are classified as either subclinical (sc), classic (cv), or very virulent (vv) pathotypes. 16 All of the genetic determinants of pathogenicity of IBDV have not been identified; however, several investigations have demonstrated that the hypervariable region of the outer capsid protein VP2 (hvVP2) as well as amino acid changes in the VP1 polymerase influence pathogenicity. The high genotypic and pathotypic variability induced by frequent genetic mutations and recombination events have led to the proposal of a new classification scheme based on genogrouping.7,12 IBDV genogrouping based on phylogenetic analysis of the hvVP2 gene of IBDV originating from 23 countries from North America, South America, Europe, and Asia placed isolates into 7 different genogroups (G). 12 Generally, G1 consisted of IBDV previously considered to be classic viruses, G2 of primarily variant IBDV, G3 of vvIBDV and vvIBDV reassortants, and G4–G7 lineages did not align with these 3 major genogroups. 12 Studies have also described the use of molecular analysis of hvVP2 segment A along with the VP1 of segment B of naturally occurring IBDV to identify interserotypic reassortants. 5
Compared to cvIBDV, disease induced by the vvIBDV pathotype is typically more acute with very pronounced lymphocyte depletion in the bursa of Fabricius in susceptible chickens. Relative to other IBDV pathotypes, vvIBDV-infected flocks typically exhibit higher morbidity, mortality, and a higher incidence of lymphocyte necrosis in non-bursal organs such as the spleen, cecal tonsils, isthmus, gut-associated lymphoid tissue, and thymus.14,15 The vvIBDV pathotype was first described in the 1980s in Belgium. 16 Subsequently, vvIBDV has rapidly spread throughout Europe, Asia, Latin America, and Africa. Although cvIBDV and scIBDV are endemic in most poultry-producing regions in the United States, vvIBDV was not detected and reported in the United States until 2008 in commercial chickens from Sonoma County, California.4,14 Detection was reported to induce a mortality of 26% and 34% in 11- and 14-wk-old IBDV-vaccinated pullets, respectively. Subsequently, vvIBDV has been detected in both commercial and backyard chicken (BYC) flocks in California and in commercial pullets in Washington State.2,13
We describe a retrospective analysis of vvIBDV, interserotypic reassortant IBDV (vvIBDV segment A and serotype 2 segment B), scIBDV, and cvIBDV strains detected by reverse-transcription, real-time PCR (RT-rtPCR), and sequence analysis of VP2 and VP1 of IBDV. These genotypes were detected in BYCs submitted to the California Animal Health and Food Safety (CAHFS) laboratory system in 2009–2017. We also describe the geographic and temporal distribution of IBDV in BYCs in California over the last decade (2009–2017).
Materials and methods
Retrospective analysis of IBDV cases
The CAHFS laboratory system consists of diagnostic laboratories in 4 locations in the State of California. The CAHFS backyard surveillance program provides subsidized autopsy and other laboratory services for backyard poultry flock owners with <1,000 birds on the premises. IBDV RT-rtPCR testing of the bursa of Fabricius is performed on chicken autopsy submissions with lesions suggestive of infectious bursal disease (IBD). Data from all BYC autopsy submissions positive for IBDV by RT-PCR in 2009–2017 were analyzed from the CAHFS electronic case reporting database. Data analyzed included mortality, age of chicken submitted, clinical signs, lesions, identification of coinfections, and the temporal and geographic distribution of IBDV-infected BYCs. The RT-rtPCR for IBDV segment A performed on diagnostic cases at CAHFS utilizes 2 TaqMan probes (Applied Biosystems, Foster City, CA). One probe is specific for vvIBDV (FAM-5′-CTCAGCTAATATCGATGC-3′) and the other probe targets non-vvIBDV resident strains that are considered endemic strains in California (VIC-5′-CAGCCAACATTGATGC-3′). 8
IBDV genotyping
Genome sequencing and phylogenetic analyses were successful on the hvVP2 segment A from 21 of 33 IBDV-positive samples and from the VP1 segment of 11 of 33 IBDV-positive samples detected from BYCs (Jackwood Laboratory, The Ohio State University). These viruses were detected and sequenced from bursa tissue homogenates that were transferred to Whatman FTA cards (MilliporeSigma, St. Louis, MO). Viral RNA was extracted from bursa samples on FTA cards using the method described previously. 10 RT-rtPCR was conducted (AgPAth-ID one step RT-PCR reagent kit; Applied Biosystems). RT-rtPCR on a 579-bp segment of the hvVP2 gene and a 1,051-bp fragment of the VP1 gene was performed as described previously. 12 The RT-rtPCR products were prepared for cycle sequencing (Wizard SV gel and PCR clean-up system; Promega, Madison, WI), which was conducted at CAHFS or the University of Wisconsin Biotechnology Center DNA Sequence Laboratory (Madison, WI). Nucleotide and predicted amino acid sequences were aligned, and phylogenetic analysis was conducted (Geneious v.8.1.8; Biomatters, Newark, NJ) as described previously. 12
Results
Epidemiology of vvIBDV
The first laboratory detection of vvIBDV in California BYCs originated from 2 BYC carcasses submitted to CAHFS from a backyard flock in Marin County in October 2008. From 2009 to 2017, 197 vvIBDV RT-rtPCR tests were performed on BYC autopsy submissions. The vvIBDV segment A was identified in 32 BYCs that originated from 17 of 197 (8.6%) BYC autopsy cases. Two cases were indeterminate, and 178 were negative for vvIBDV by RT-rtPCR. The 17 vvIBDV BYC cases originated from 7 counties in California: 13 originated from northern California (Sonoma, Yuba, Marin, and Yolo counties), 2 originated from the Central Valley (San Joaquin and Stanislaus counties), and 2 originated from Riverside County in southern California (Fig. 1A). The frequency of vvIBDV detected in BYCs was 0–5 cases per year, with the highest number (5 cases) of vvIBDV detected in 2010 (Fig. 2).
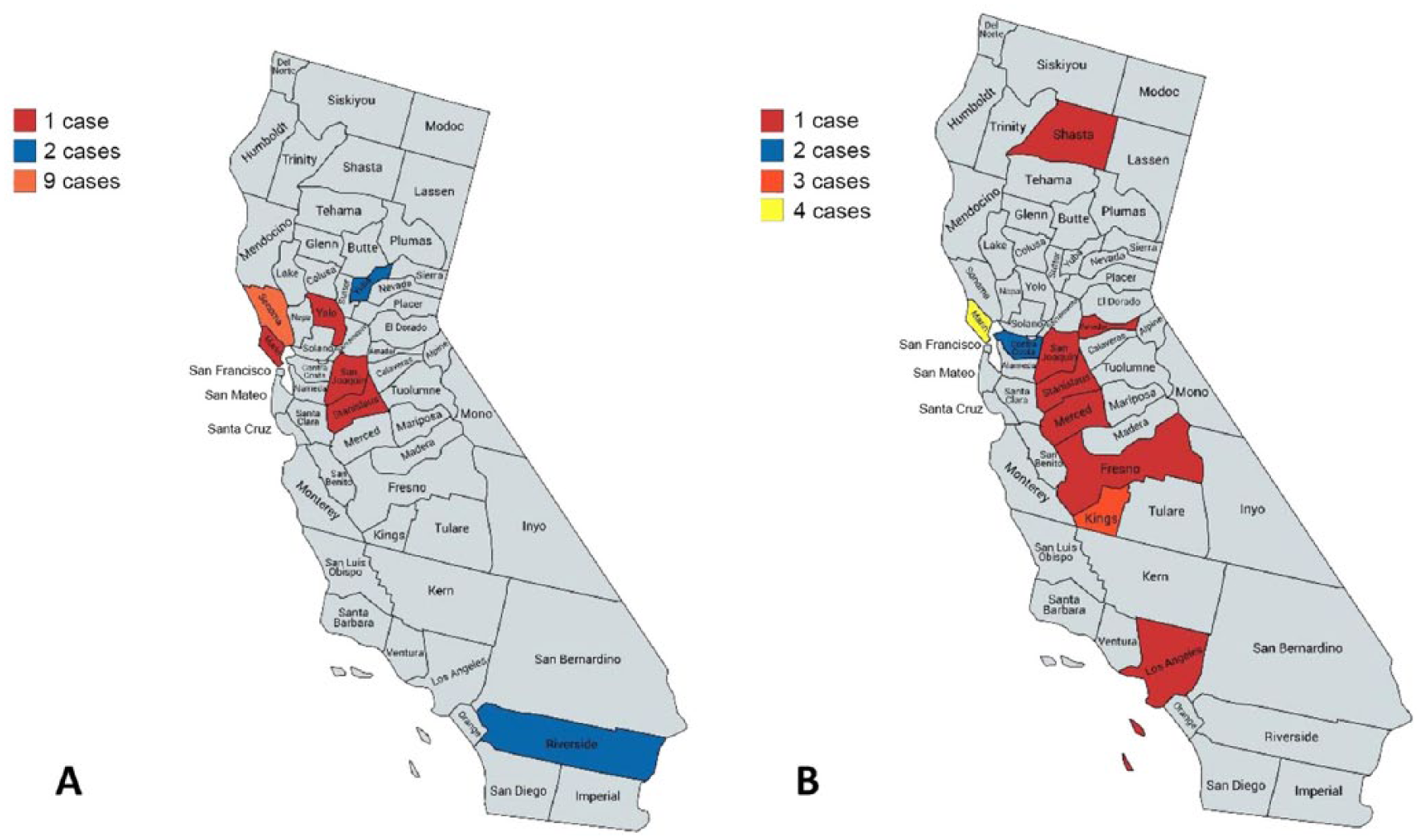
The geographic distribution by county of
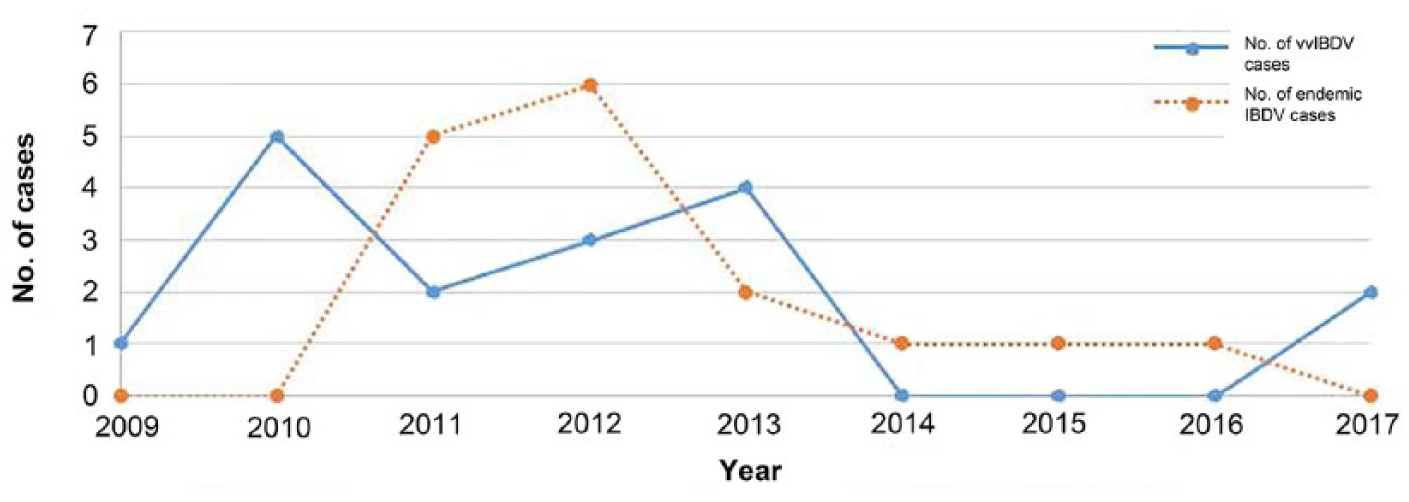
The number of cases of very virulent infectious bursal disease virus (vvIBDV) and endemic, non-vvIBDV detected in backyard chickens in California, 2009–2017.
Data on the age of chickens submitted was available for 15 of 17 of the vvIBDV BYC cases. The reported age of vvIBDV-infected chickens was 4–18 wk, with a mean age of 11.5 wk, median of 11 wk, and mode of 16 wk of age. Data on flock size were available in 8 vvIBDV cases and ranged from 1 to 100 chickens on affected premises, with an average flock size of 37 chickens. Mortality was recorded in 10 of the 17 vvIBDV cases and was 0–67%, with a mean of 28%. The breeds of BYCs were reported in several vvIBDV cases as American Game (1), Maran (6), Cochin (1), Barnevelder (1), Brahma (1), Cornish cross (4), and Rhode Island Red cross (1).
The most commonly reported clinical history in the vvIBDV submissions was either lethargy (6 of 17 cases) or sudden death with no prior clinical disease observed in the flock (6 of 17 cases). Respiratory distress was reported in 3 of 17 cases, inappetence in 2 of 17, and ataxia in 2 of 17 cases. The most frequently reported histologic lesion in BYCs with vvIBDV was severe lymphocyte depletion in the bursa of Fabricius (15 of 17 cases). Lymphocyte depletion in the spleen, cecal tonsils, and thymus was described in 10 of 17 cases. Other histologic findings described in vvIBDV case submissions were respiratory lesions (3 of 17), nephrosis (1 of 17), Marek’s lymphoid tumors (1 of 17); no other lesions were described in 1 case.
One concomitant disease was detected in 10 of 17 vvIBDV cases, 2 concomitant diseases were detected in 1 of 17 cases, and no concomitant diseases were detected in 6 of 17 cases. Coinfections detected in vvIBDV BYC autopsy cases were colibacillosis (6 of 17), Marek’s disease (3 of 17), coccidiosis (2 of 17), aspergillosis (1 of 17), and intestinal tapeworms (1 of 17).
Epidemiology of non-vvIBDV
Non-vvIBDVs were detected by RT-rtPCR from 51 BYCs that originated from 16 of 197 (8.1%) autopsy cases submitted to the CAHFS laboratory from 2009 to 2017. Three cases were indeterminate for non-vvIBDV, and 184 cases were negative for non-vvIBDV by RT-rtPCR. The 16 positive BYC cases of non-vvIBDV originated from 10 counties distributed in north, central, and southern California (Fig. 1B). The frequency of non-vvIBDV cases detected in autopsy submissions ranged from 0 to 6, with the peak of 6 cases detected in 2012 (Fig. 2). Case data on the age of chickens submitted were available in 14 of 16 cases. The ages of BYCs positive for non-vvIBDV ranged from 3.5 to 23 wk, with a mean age of 8 wk and a median of 6.3 wk. Data on flock size was available for all 16 cases and ranged from 5 to 800 chickens, with an average of 200 chickens on affected premises. Mortality was reported in 15 of 16 cases and ranged between 1.5–80%, with a mean mortality of 14.6%. The breeds reported in some non-vvIBDV case submissions were American Game (9), Cochin (1), Maran (1), Rhode Island Red (1), Speckled Sussex (1), Tolbunt Polish (1), White Langshan (1), Black Langshan (1), and Ameraucana (1).
Clinical signs reported for non-vvIBDV submissions were lethargy (5 of 16), increased mortality (5 of 16), neurologic signs (4 of 16), respiratory signs (3 of 16), and bloody stool (1 of 16). No clinical signs were reported in 1 of 16 submissions. Microscopic findings in non-vvIBDV autopsy cases were as follows: lymphoid cell depletion in the bursa of Fabricius (13 of 16), enteritis (4 of 16), typhlitis (4 of 16), fungal granulomas in the respiratory system (3 of 16), lymphoid depletion in spleen (2 of 16), encephalomalacia (2 of 16), lymphoid tumors (3 of 16), pneumonia (3 of 16), polyserositis (1 of 16), hepatitis (1 of 16), and pericarditis (1 of 16).
Concomitant diseases were detected in all 16 of the non-vvIBDV cases. One concomitant disease was detected in 7 of 16 cases, 2 concomitant diseases were detected in 5 of 16 cases, and 3 concomitant diseases were described in 5 of 16 cases. Diseases detected in addition to IBD were coccidiosis (5 of 16), vitamin E deficiency (4 of 16), aspergillosis (3 of 16), Marek’s disease (3 of 16), E. coli polyserositis (2 of 16), group D intestinal salmonellosis (1 of 16), trichomoniasis (2 of 16), runting/stunting syndrome (1 of 16), trauma (1 of 16), histomoniasis (1 of 16), transmissible viral proventriculitis (1 of 16), E. coli hepatitis (1 of 16), enteritis (1 of 16), and necrotic enteritis (1 of 16).
Phylogenetic analysis of IBDV in backyard chicken cases
Using RT-rtPCR, a 579-bp fragment of the IBDV hvVP2 gene was amplified from 21 of 33 IBDV cases detected in bursa tissue samples from backyard poultry flocks submitted to CAHFS in 2009–2017 (Table 1; Fig. 3). Using the predicted amino acid sequence of the hvVP2 gene, the viruses were phylogenetically placed into 3 genogroups (G1–G3). The G3 group (vvIBDV) contained 14 viruses; 6 samples contained G2 viruses (similar to variant strains); and 1 sample (K1201563) contained a G1 virus (Lukert strain; Table 1; Fig. 3). The markers for virulence (253Q and 284A) were identified in all viruses except the G1 Lukert strain virus, in which 253H and 284T were identified, indicating that it may be a vaccine strain. No segment A serotype 2 IBDV strains were observed.
Summary of sequence analysis of infectious bursal disease virus (IBDV) from backyard chicken autopsy cases submitted to CAHFS, 2009–2017.
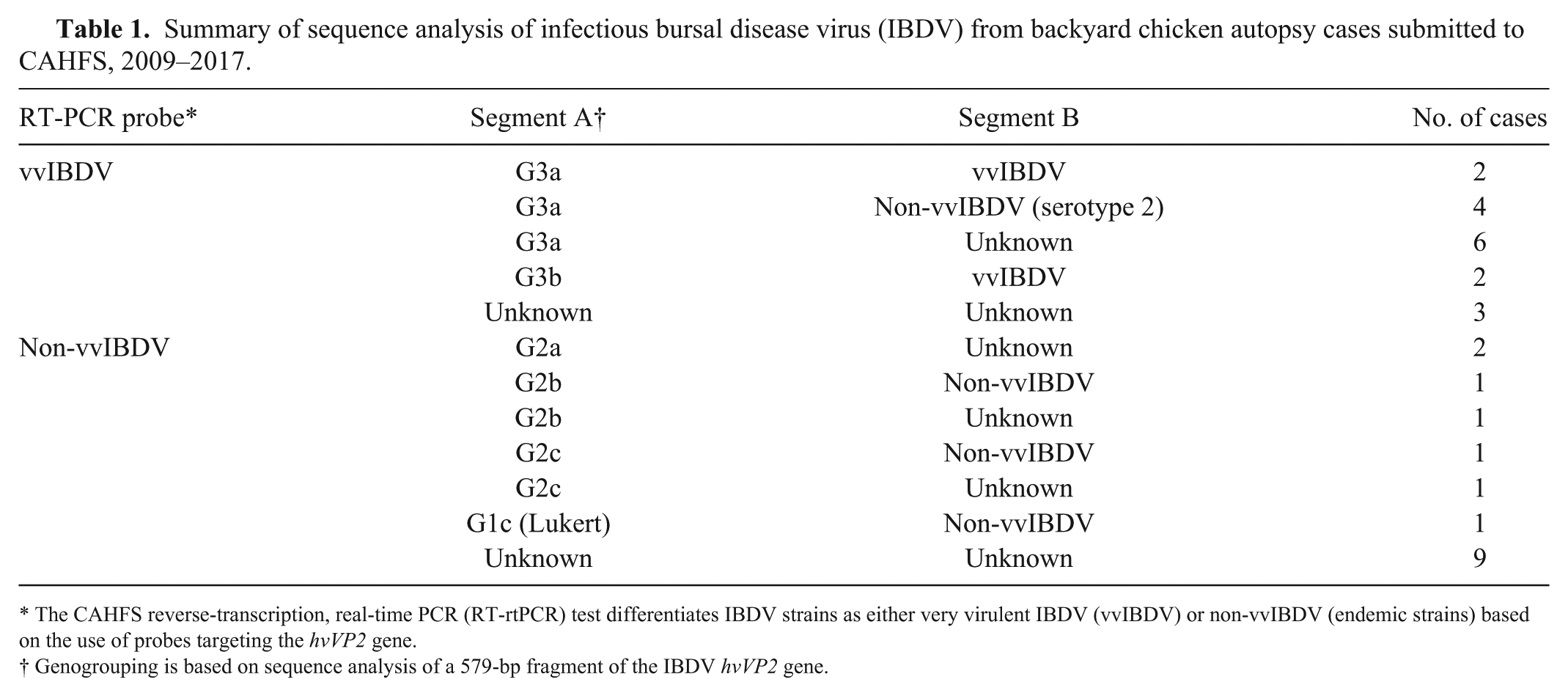
The CAHFS reverse-transcription, real-time PCR (RT-rtPCR) test differentiates IBDV strains as either very virulent IBDV (vvIBDV) or non-vvIBDV (endemic strains) based on the use of probes targeting the hvVP2 gene.
Genogrouping is based on sequence analysis of a 579-bp fragment of the IBDV hvVP2 gene.
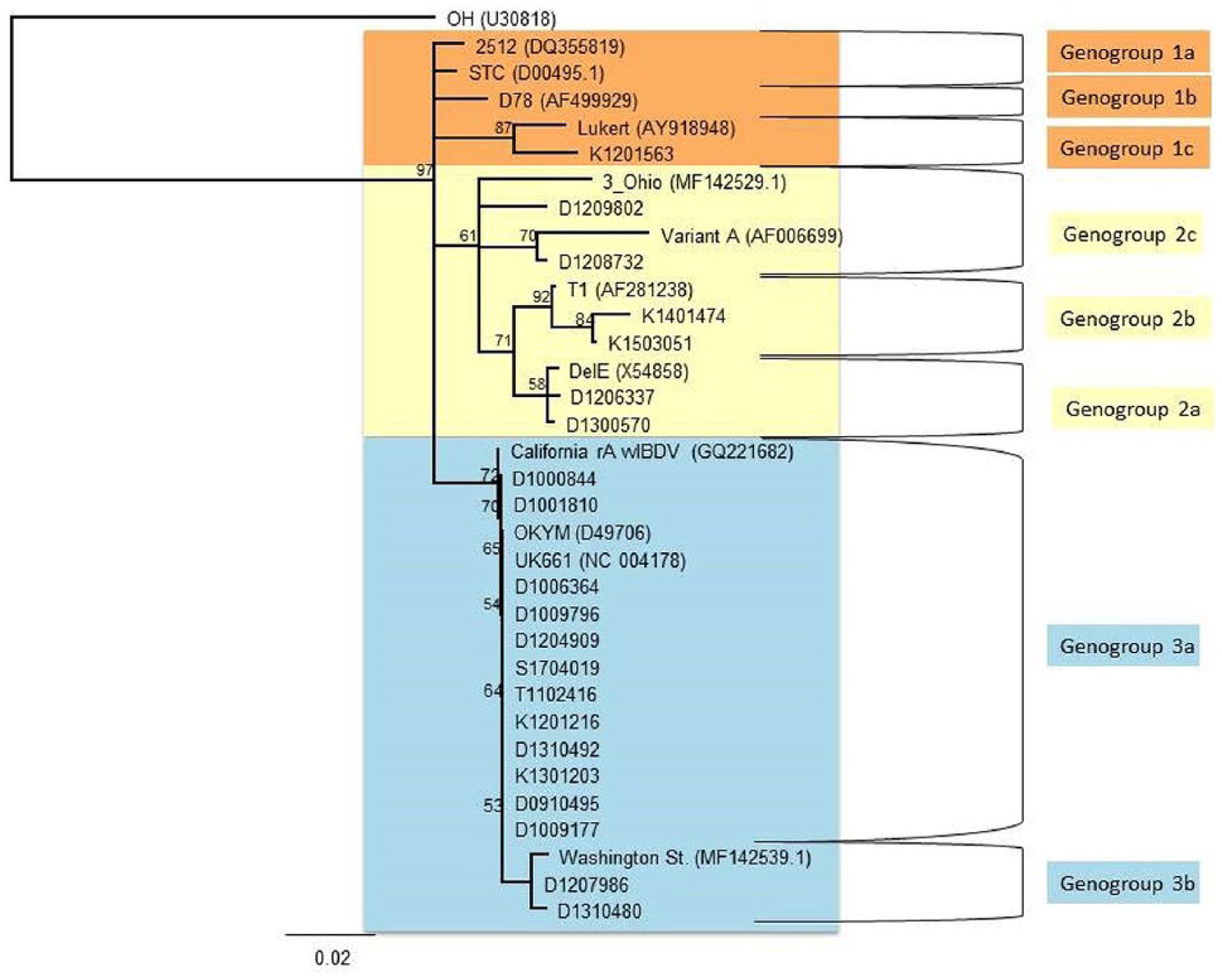
Phylogenetic analysis of the hvVP2 nucleotide sequences from infectious bursal disease virus (IBDV) using the neighbor-joining method. Color coding was used to differentiate G2 groups, with genogroup 1 highlighted in orange, genogroup 2 highlighted in yellow, and genogroup 3 highlighted in blue. The IBDV reference strains included in the phylogenetic analysis have GenBank accessions in parentheses.
The 6 G2 viruses are typical of variant strains. At least 1 backyard IBDV was detected in each of the 3 G2 lineages (G2a–c). The 14 G3 viruses contained VP2 amino acids typically seen in vvIBDV strains (222A, 242I, 256I, 294I, and 299S). Backyard flock vvIBDV was observed in the G3a and G3b lineages. Viruses from these backyard flocks were not found in the G3c lineage.
To differentiate between true and reassorted vvIBDV, a 1,051-bp fragment of genome segment B (VP1 gene) was amplified using RT-rtPCR (Table 1; Fig. 4). We were unable to amplify the VP1 gene from 9 backyard G3 vvIBDV and 13 non-vvIBDV cases. Sequence analysis of genome segment B from the vvIBDV that was successfully amplified indicated that 4 G3 viruses contained the VP1 gene associated with high virulence in vvIBDV strains (Fig. 4). Four G3 viruses (D0910495, D1310492, S1704019, and T1102416) had a genome segment B typical of serotype 2 IBDV and thus are interserotypic reassorted strains.
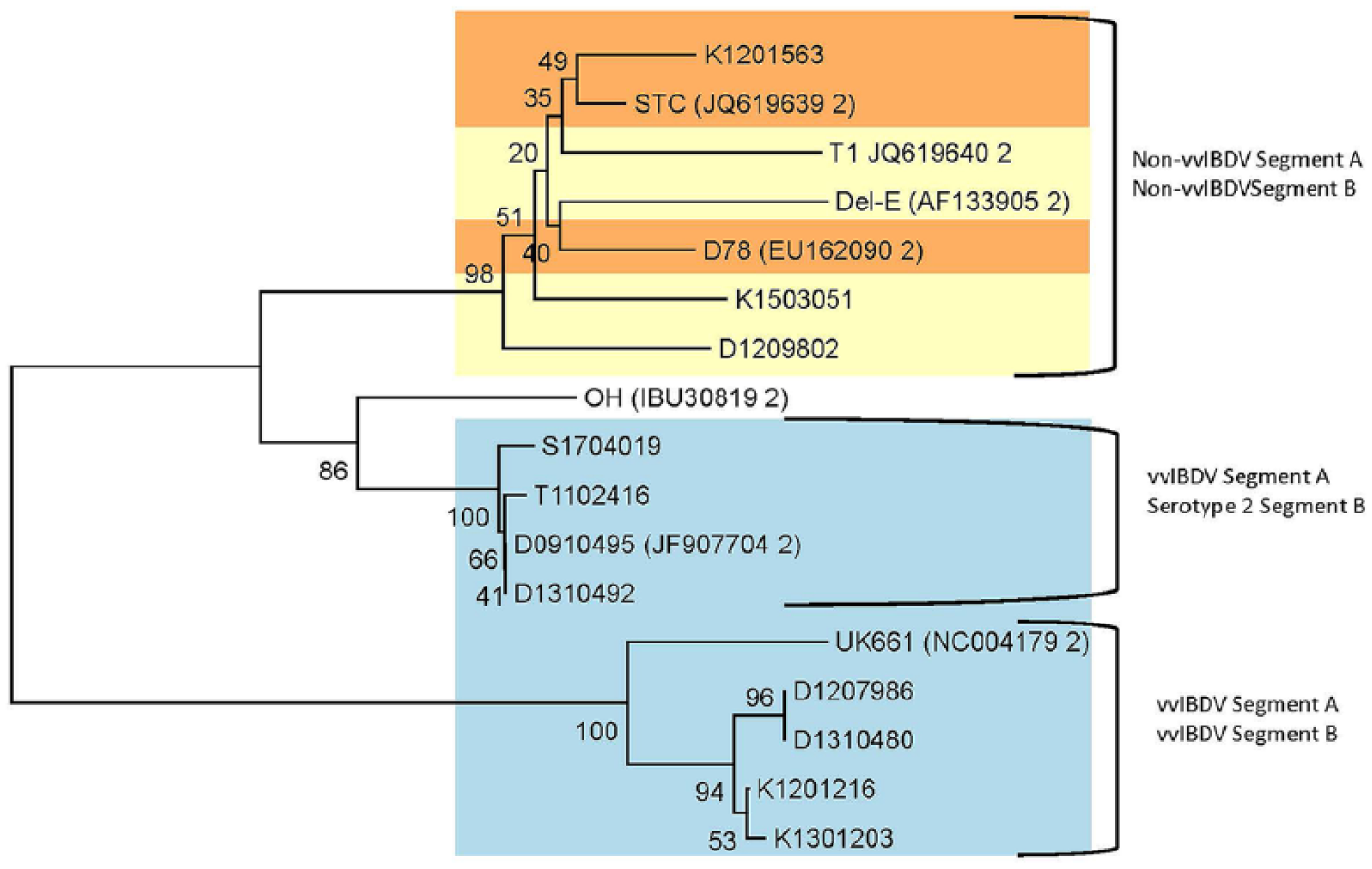
Phylogenetic analysis of VP1 nucleotide sequences of infectious bursal disease virus (IBDV) using the neighbor-joining method. The colored boxes are coded to match the correlated genogroups in the segment A tree in Figure 3. The IBDV reference strains included in the phylogenetic analysis have GenBank accessions in parentheses.
Discussion
From 2009 to 2017, similar numbers of vvIBDV and non-vvIBDV cases were diagnosed at CAHFS. Based on sequence analysis of segments A and B of IBDV, 4 of 17 cases reported as vvIBDV were true vvIBDV; 4 of 17 cases were interserotypic reassortant viruses. We were unable to perform sequencing of hvVP2 and VP1 from 12 of 33 and 22 of 33 IBDV samples retrieved from FTA cards, respectively. One reason may be that the quantity of virus was too low to obtain sufficient amounts of amplified product for sequencing. We were able to sequence almost twice as many hvVP2 compared to VP1 fragments. Also, it may be that RNA degraded on FTA cards; the 579 hvVP2 fragment could be identified more often than the larger 1,051 fragment of VP1. Based on sequence analysis, the non-vvIBDV segment A detected at CAHFS aligned with classic (G1) and variant (G2) IBDV strains.
Phylogenetic analysis of the backyard IBDV strains indicated that the strains were in 3 genogroups. The K1201563 virus strain in G1 had an hvVP2 sequence typical of a Lukert strain vaccine virus. The IBDV strains in the G2 phylogenetic group are endemic in the United States, are often seen in commercially reared chickens,10,12 and are typical of variant IBDV strains. The G3 viruses that we detected in backyard flocks are typical of vvIBDV. The G3a and G3b lineages in this genogroup have been found previously in commercial poultry flocks. 12 A third lineage of vvIBDV (G3c) has been observed in commercial poultry, but this lineage was not found in the backyard flock samples we tested. 10 Lineage G3a contains the type strains of vvIBDV (UK661 and OKYM) plus the original vvIBDV isolated in California. 14 The vvIBDV from Washington State is in a different lineage (G3b). Although the D1310480 virus is also in G3b, it has 2 mutations (D212 and I 252) that are not observed in the Washington State virus. This suggests that the vvIBDVs are evolving as they replicate in these backyard populations.
Four G3 viruses (D0910495, D1310492, S1704019, and T1102416) had a genome segment B typical of serotype 2 IBDV and thus are vvIBDV reassorted strains with potentially lower virulence for commercial chickens. 5 The other 4 G3 viruses that we were able to amplify and sequence contained a VP1 typical of true vvIBDV strains, which can cause high mortality in commercial chickens. Although confounding factors such as coinfections can influence mortality, the clinical presentation and 0–67% mortality history in these backyard flocks suggest that the true vvIBDVs are able to induce high mortality in some backyard breeds. This is concerning because the IBDV strains in backyard poultry are a risk to commercially produced chickens. Biosecurity in backyard flocks is frequently minimal or nonexistent and thus biosecurity in commercial poultry farms located near these flocks is crucial. The G3 vvIBDVs are of greatest concern given their potential to cause high mortality and devastating economic losses. The original vvIBDV identified in California is in the G3a lineage. The G3b Washington State virus and outlier (D1310480) appears to indicate viral evolution as opposed to a new introduction. Previous studies have shown that only 1 or 2 amino acid changes are necessary in the hvVP2 region to affect pathogenicity 1 and antigenicity. 9 The possibility that these viruses are becoming better adapted to replication in backyard poultry breeds cannot be excluded, and additional studies are needed to assess the impact these mutations may have on the pathogenicity of these vvIBDVs for commercial chickens.
Geographically, the vvIBDV and vvIBDV reassortant cases were restricted mainly to counties in northern California, whereas the non-vvIBDV cases were widely distributed in counties throughout the state. Interestingly, the high density of vvIBDV-positive backyard flocks in northern California correlates with the spatial distribution of vvIBDV reported in commercial chicken flocks within the state. 13 A high concentration of positive vvIBDV commercial flocks based on spatial cluster analysis of vvIBDV was detected in California between 2008 and 2011. 13
In 2009–2017, the total number of cases and frequency of cases detected per year were similar for vvIBDV/vvIBDV reassortant and non-vvIBDV cases. The average mortality reported for vvIBDV/vvIBDV reassortant case submissions was approximately double the mortality reported for non-vvIBDV cases. The increased virulence of vvIBDV and vvIBDV reassortants compared to classic and variant strains have been reported previously in both natural and experimental IBDV infections.5,6,11 Although lymphocyte depletion in the bursa of Fabricius was the most commonly reported lesion for both vvIBDV/vvIBDV reassortant and non-vvIBDV backyard submissions, the detection of concomitant diseases was higher with non-vvIBDV infections. In the 16 CAHFS cases of non-vvIBDV, a total of 13 different concomitant diseases were described compared with 5 other concomitant diseases detected with vvIBDV and vvIBDV reassortant cases. This result is not surprising given that classic and variant IBDV are associated with an increased incidence of opportunistic infections and comorbidity. 3 We found that the median age of BYCs submitted with vvIBDV and vvIBDV reassortant infection was higher than the median age of BYCs submitted with non-vvIBDV classic and variant virus infection. Although data may support that an increased virulence of vvIBDV and vvIBDV reassortant viruses may be correlated with increased susceptibility in older chickens, further investigation is needed. 16 With respect to breeds of BYCs affected, there were 7 different breeds reported in vvIBDV cases and 9 breeds for non-vvIBDV, with American Game chickens being the most commonly reported breed in which IBDV was detected. In spite of this, the exact correlation between IBDV susceptibility and breed of chickens needs to be examined further.
One disadvantage of our study is that autopsy case submissions may not represent the true prevalence and distribution of IBD in BYCs because owners throughout the state may not utilize the diagnostic laboratory to evaluate sick or dead birds. In addition, IBDV strains correlated with uncomplicated subclinical infections or mild virulence may be under-detected given that owners are less likely to submit birds displaying minimal clinical disease. Obtaining representative clinical samples for IBDV surveillance from the BYC population in California is a challenge, and we utilized molecular epidemiology data obtained from CAHFS autopsy submissions to investigate the genetic and pathotypic diversity of IBDV in BYCs. The identification of vvIBDV, cvIBDV, scIBDV, and interserotypic IBDV highlights the genetic diversity of IBDV in BYCs in California and highlights the importance of biosecurity in both backyard and commercial poultry flocks.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
