Abstract
Apelin is a peptide known for its insulin-sensitizing effect. This study aimed to examine the relationship between apelin and fatty liver disease. Non-diabetic patients were evaluated by ultrasound in this prospective, single-center, case-control study between 1 April 2020 and 1 April 2021. Serum apelin levels were measured, and the relationship with ultrasonography results and degree of hepatosteatosis was examined. Statistical analysis was carried out using NCSS 2007. This study was conducted with 80 non-diabetic participants, 53 females and 27 males. The mean age of the cases was 40.55 ± 13.82 (interquartile range = 27–51) years, and their mean body mass index (BMI) was 29.34 ± 7.44 (interquartile range = 23.4–35.2) kg/m2. There was no hepatosteatosis according to ultrasound in 42% (n = 34) of the cases. When patients with or without hepatosteatosis were compared, there was no difference in terms of gender, alanine aminotransferase (ALT), and hemoglobin levels, whereas fasting blood glucose (FBG), HOMA-IR, triglycerides, LDL-cholesterol, BMI, creatinine, and age were higher in the group with hepatosteatosis. Apelin was found to be lower in patients with non-diabetic hepatosteatosis than in those without hepatosteatosis (p = .001). As the degree of fatty liver increased, the apelin level decreased further. There was also a negative correlation between apelin and age. However, the relationship between apelin and hepatosteatosis was independent of age. In non-diabetic patients with fatty liver disease, compared to those without fatty liver disease, serum apelin level was lower and was negatively correlated with the degree of steatosis, independent of age.
Keywords
Introduction
Non-alcoholic fatty liver disease is a global health problem. Its prevalence is around 25% and is increasing day by day. Non-alcoholic fatty liver disease ranges in a spectrum from simple fatty infiltration to steatohepatitis with inflammation and fibrosis. Steatohepatitis can progress to liver cirrhosis and hepatocellular cancer. These patients also have increased cardiovascular mortality.
Adipose tissue can synthesize and release various molecules that act as hormones, such as cytokines, extracellular matrix proteins, and adipokines.1,2 Apelin, one of these adipokines, is produced and secreted by the heart, liver, brain, kidney, lung, hypothalamus, stomach, and adipose tissue. Its secretion is regulated by insulin. Apelin increases insulin sensitivity and affects glucose and lipid metabolism.3,4 It also plays a role in inflammation, maintaining vascular tonus, and stimulating angiogenesis.
The increase in apelin levels in the presence of NAFLD in diabetic patients has been shown in a few previous studies.5,6 However, the relationship between the degree of steatosis and serum apelin levels in NAFLD has not yet been clearly shown. This study aimed to evaluate the relationship between serum apelin levels and the presence of steatosis and its ultrasonographic degree in NAFLD patients without diabetes.
Materials and methods
After obtaining approval of the ethics committee of Prof. Dr. Cemil Tascioglu City Hospital, dated 31 March 2020 and numbered 48670771/73, among the patients who applied to the internal diseases outpatient clinic for any reason, those who were over 18 years of age, who had an ultrasound (USG) performed, and had no chronic comorbidities were included in this case-control study. According to anamnesis of a patient, laboratory results, medical record, and ICD-10 codes, patients with chronic liver disease, viral hepatitis, autoimmune hepatitis, Wilson’s disease, hemochromatosis, chronic kidney failure, diabetes mellitus, malignancy, chronic inflammatory disease, acute or chronic infectious disease, ethylism or drug abuse, pregnant or breastfeeding women, and patients under immunosuppressive treatment were not included in the study (Figure 1). Assessment of hepatosteatosis was carried out using a Siemens Acuson Juniper diagnostic ultrasound system (Mountain View, CA, USA) with a convex probe. Images were examined on the same monitor under the exact same lighting conditions. Hepatosteatosis was graded according to the severity of steatosis: normal (grade 0), liver attenuation slightly less than the spleen (grade 1), more pronounced difference between liver and spleen and intrahepatic vessels not seen or at a slightly higher attenuation than the liver (grade 2), and markedly reduced liver attenuation with a sharp contrast between the liver and intrahepatic vessels (grade 3). Patients who did not have fatty liver in USG constituted our control group and those with fatty infiltration were divided into three groups according to the stage of fatty liver disease. Gender, age, body mass index (BMI), fasting blood glucose (FBG), creatinine, alanine aminotransferase (ALT), LDL-cholesterol (LDL), triglyceride, hemoglobin, and homeostatic model assessment insulin resistance (HOMA-IR) values of the study participants were recorded. Volunteer participants were included in the study from 1 April 2020 to 1 April 2021. Study population flow chart.
Blood samples taken from the participants for the measurement of apelin were centrifuged at 4000 r/min for 10 min and then stored at −80°C. BMI was obtained by dividing the weight in kg by the square of the height in meters. HOMA-IR was calculated with the following formula: Fasting serum insulin (uIU/Ml) * Fasting serum glucose (mg/dL)/405.
Statistical analysis
Power analysis was performed with the G*power (version 3.1.9.7) program. The minimum calculated total sample size of the study was 64 (effect size = .8, alpha error = .05, power = .95). The NCSS (Number Cruncher Statistical System) 2007 (Kaysville, Utah, USA) program was used for the statistical analysis. The suitability of quantitative data for normal distribution was tested by Kolmogorov–Smirnov test and graphical examinations. When evaluating the study data, descriptive statistical methods (mean, standard deviation, median, frequency, ratio, minimum, and maximum) were used, as well as the Student-t Test for the comparison of normally distributed variables in two groups in comparison of quantitative data, and Mann–Whitney U test for two-group comparisons of variables that did not show normal distribution. The Kruskal–Wallis test was used for comparisons of three and more groups that did not show normal distribution, and the Dunn–Bonferroni test was used to determine the group that caused the difference. The Pearson chi-square test and the Fisher–Freeman–Halton test were used to compare qualitative data. Spearman correlation analysis was used to evaluate relationships between quantitative variables. Logistic regression analysis was used for multivariate evaluations. Significance was evaluated at the p < .05 level.
Results
Descriptive data of the patients.

aStudent-t Test.
bPearson chi-square test.
**p < .01.
Biochemical characteristics of the patients.
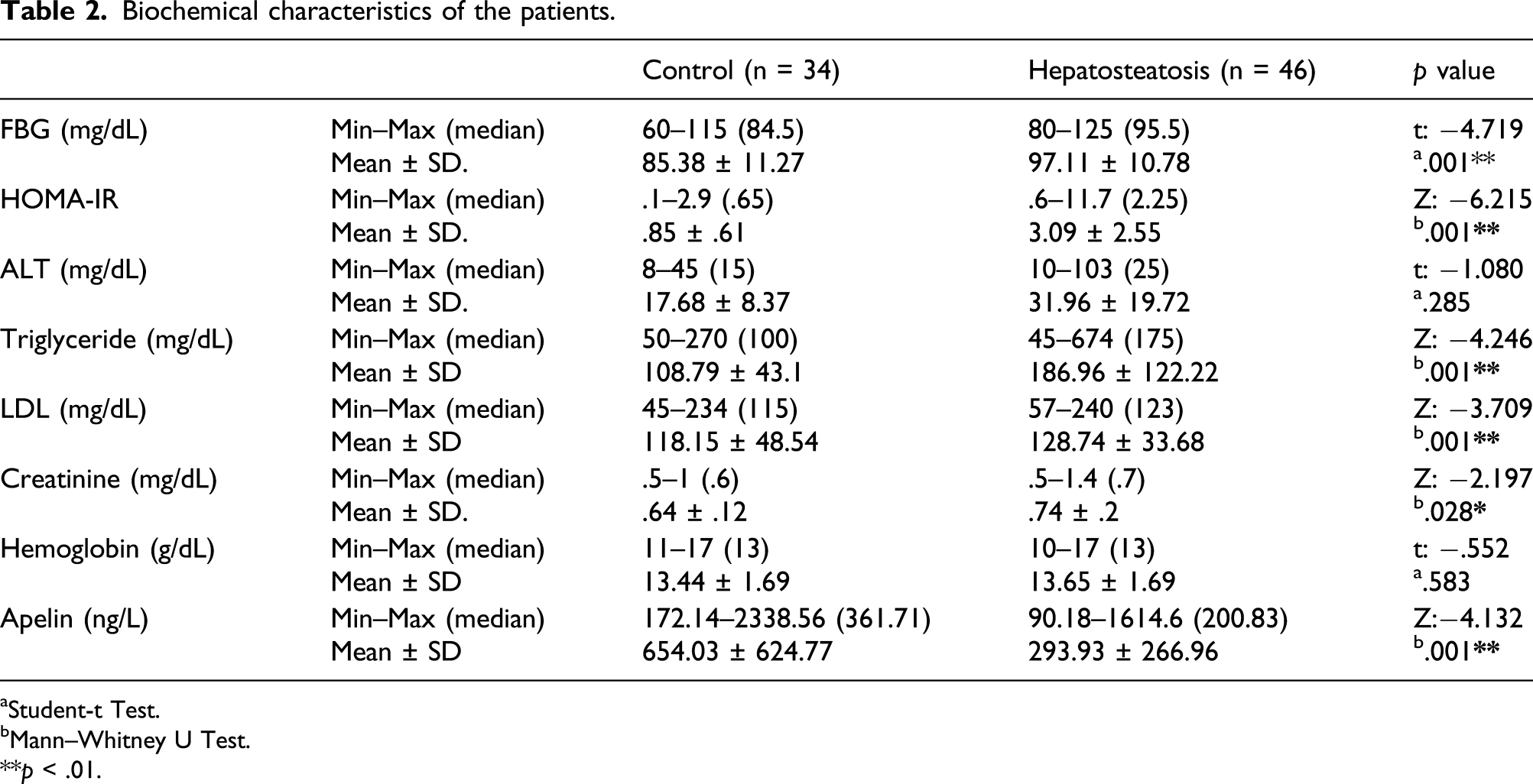
aStudent-t Test.
bMann–Whitney U Test.
**p < .01.
Logistic regression results of factors affecting steatosis.

**p < .01.
Correlation of between apelin and BMI, FBG, HOMA-IR, LDL, and triglycerides.

r = Spearman’s korelasyon katsayısı; *p < .05 **p < .01.
Discussion
NAFLD is characterized by accumulation of fat in the liver. There are several adipokines that exert an influence on NAFLD, such as resistin, PAI-1, visfatin, RBP-4, chemerin, adipsin, obestatin, omentin, vaspin, adiponectin, and leptin, and there are various studies that evaluated these as biomarkers. 7
Another adipokine, apelin, which plays an important role in glucose metabolism with its insulin-sensitizing effect, is synthesized and secreted from many tissues. Insulin regulates apelin synthesis and apelin increases the uptake of glucose from peripheral tissue. In addition, a small number of studies on apelin, which is also associated with inflammation and angiogenesis, have been conducted with diabetic patients in general.
While a positive correlation was found between the plasma apelin level, and insulin resistance, and HbA1C in some previous clinical studies, some studies showed that the plasma apelin level paradoxically decreased in diabetic patients.8-11 For example, Chang et al. found that the apelin level was lower in female patients with polycystic ovary and insulin resistance compared to a control group. 12 Similarly, in our study, the apelin level in non-diabetic patients was found to have a negative correlation with FPG and HOMA-IR levels. These different data suggest that the real role of apelin in insulin resistance remains uncertain.
Montazerifar and Aktas found a positive correlation between apelin and BMI in two separate studies and reported that the serum apelin level was higher in those with NAFLD.13,14 Another study performed with only male patients found no difference between apelin levels in patients with NAFLD and a control group in the same age group. 15 In our study, which is one of the uncommon studies in the literature conducted in patients with hepatosteatosis including both genders, a negative correlation was found between BMI and apelin level in non-diabetic patients. Moreover, the apelin level of patients with hepatosteatosis was lower than that of patients without hepatosteatosis.
On the other hand, Hashım et al. showed a relationship between increased apelin levels and the histological severity of NAFLD. 16 Since there was no histological verification in our study, which is the limiting part of our study, we do not know how many of the patients we included in the study were having simple steatosis and how many had steatohepatitis.
While the apelin level is increased in diabetic patients because of low insulin levels, for cases with insulin resistance, such as polycystic ovary, the level of apelin may be lower because the insulin level is high. Likewise, in our study, the apelin level may have been found to be low because of fatty liver and insulin resistance. Its negative correlation with BMI and FBG also suggests the presence of excess insulin. A low apelin level, which increases insulin sensitivity in non-diabetic patients with fatty liver, suggests that problems such as insulin resistance and diabetes may arise in these patients.
Limitation
This study had three potential limitations, one being the small number of patients. The second was that it was based on data from a single center. The third limitation was the absence of liver biopsies. Multicenter studies with more patients and liver biopsies could yield more valid results.
Conclusion
In light of the results of all these studies, it is seen that there may be other factors affecting apelin levels other than insulin that are not yet clear. Therefore, there is still a need for studies involving more cases in which the apelin level is examined in non-diabetic NAFLD cases with histopathological differentiation, which could clarify the relationship between apelin and hepatosteatosis and shed light on these unknowns, as well as detect other possible molecules affecting apelin.
Footnotes
Acknowledgments
We would like to thank Emire Bor (EMPIAR) for statistical consultation and data analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Ethical approval for this study was obtained from Prof. Dr Cemil Taşcıoğlu City Hospital’s ethics committee, dated 31.03.2020 and numbered 48670771/73
Informed consent
Informed consent was obtained from all individual participants included in the study.
