Abstract
Prion diseases are neurodegenerative diseases characterized by the accumulation of misfolded prion protein (PrPSc) in the brain and other tissues. Animal prion diseases include scrapie in sheep, chronic wasting disease (CWD) in cervids, and transmissible mink encephalopathy (TME) in ranch-raised mink. We investigated the susceptibility of raccoons to various prion disease agents and compared the clinicopathologic features of the resulting disease. Raccoon kits were inoculated intracranially with the agents of raccoon-passaged TME (TMERac), bovine-passaged TME (TMEBov), hamster-adapted drowsy (TMEDY) or hyper TME (TMEHY), CWD from white-tailed deer (CWDWtd) or elk (CWDElk), or atypical (Nor98) scrapie. Raccoons were euthanized when they developed clinical signs of prion disease or at study endpoint (<82 mo post-inoculation). Brain was examined for the presence of spongiform change, and disease-associated PrPSc was detected using an enzyme immunoassay, western blot, and immunohistochemistry. All raccoons inoculated with the agents of TMERac and TMEBov developed clinical disease at ~6.6 mo post-inoculation, with widespread PrPSc accumulation in central nervous system tissues. PrPSc was detected in the brain of 1 of 4 raccoons in each of the CWDWtd-, CWDElk-, and TMEHY-inoculated groups. None of the raccoons inoculated with TMEDY or atypical scrapie agents developed clinical disease or detectable PrPSc accumulation. Our results indicate that raccoons are highly susceptible to infection with raccoon- and bovine-passaged TME agents, whereas CWD isolates from white-tailed deer or elk and hamster-adapted TMEHY transmit poorly. Raccoons appear to be resistant to infection with hamster-adapted TMEDY and atypical scrapie agents.
Introduction
The transmissible spongiform encephalopathies (TSEs) are fatal neurodegenerative diseases that include kuru and Creutzfeldt-Jakob disease in humans, scrapie in sheep, bovine spongiform encephalopathy (BSE) in cattle, chronic wasting disease (CWD) of cervids, and transmissible mink encephalopathy (TME). A central event in the pathogenesis of TSEs is the conversion of the cellular form of the prion protein (PrPC), which is encoded by the PRNP gene, to a misfolded, pathogenic form (PrPSc). For this reason, TSEs may also be termed prion diseases.
When TSEs are transmitted from one species to another, the success of transmission, also known as the “transmission barrier” or “species barrier,” can be unpredictable and appears to be the result of complex interactions between the TSE agent and host factors, mainly host PrPC.7,39,47 Naturally occurring TSEs have not been reported in raccoons (Procyon lotor), although the habitat of these ubiquitous omnivores likely overlaps with that of CWD-infected deer and scrapie-infected sheep. In addition, raccoons have been identified as one of the primary scavengers of deer carcasses in a CWD-affected area of Wisconsin. 25 Previous studies demonstrated that raccoons develop clinical disease and PrPSc accumulation in tissues following intracranial (Eckroade RJ. Neuropathology and experimental transmission to other species of transmissible mink encephalopathy (TME). PhD thesis. Madison, WI: University of Wisconsin, 1972)9,22 or oral (Eckroade RJ, 1972) 9 inoculation of mink-origin TME, intracranial inoculation of classical sheep scrapie, 20 and intracranial inoculation of raccoon-passaged TME or scrapie agents. 19
TME is a rare, noncontagious prion disease of ranch-raised mink (Mustela vison) that is thought to be caused by a food-borne pathogen, most likely through feeding of raw tissues from BSE-affected cattle (Marsh RF, Hartsough GR. Evidence that transmissible mink encephalopathy results from feeding infected cattle. In: Proc 4th International Sci Congr Fur Anim Prod; Toronto, Canada; 1988). 28 We have previously reported the successful transmission of mink-origin TME to raccoons 22 and demonstrated that this isolate retains its biological characteristics on second passage in raccoons. 19
Inoculation of the TME agent from the Stetsonville, WI outbreak of TME in 1985 into Syrian hamsters, followed by serial passage in Syrian hamsters, resulted in the emergence of 2 clinical forms of hamster-adapted TME: the “hyper” (HY) form was characterized by hyperexcitability and cerebellar ataxia, whereas hamsters with the ‘drowsy’ (DY) form of the disease had complete lethargy and no hyperexcitability or cerebellar ataxia. 27 To date, the potential for raccoons to serve as a host for the HY or DY hamster-adapted TME agent is unknown.
Two forms of scrapie are recognized in sheep: classical scrapie causes disease in multiple animals within a flock, sheep develop clinical scrapie at 2–5 y of age, and affected animals have widespread spongiform change and PrPSc accumulation in the brain and lymphoid tissues. In contrast, atypical (Nor98) scrapie is characterized by sporadic incidence within a flock, disease in older animals, and lesions and PrPSc accumulation restricted to the central nervous system (CNS) tissues.4,5 We have previously reported the successful transmission of classical scrapie isolate US 13-7 to raccoons 20 and maintenance of phenotypic characteristics on second passage. 19 To date, the potential for raccoons to serve as a host for atypical scrapie is unknown.
CWD is a naturally occurring contagious prion disease that affects cervid species including mule deer (Odocoileus hemionus),29,46,48 Rocky Mountain elk (Cervus elaphus nelsoni),46,49 and white-tailed deer (Odocoileus virginianus).29,46 Previous attempts to transmit CWD from mule deer to raccoons via the intracranial route were not successful.18,20
We report herein the successful transmission of the agents of bovine-passaged TME, third-passage raccoon TME, CWD from elk, CWD from white-tailed deer, and hamster-adapted HY TME to raccoons. Misfolded prion protein or clinical signs were not detected following intracranial inoculation of the agents of atypical scrapie or hamster-adapted DY TME.
Materials and methods
Ethics statement
Our experiment was carried out in accordance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Academy of Sciences, Washington, DC) and the Guide for the Care and Use of Agricultural Animals in Research and Teaching (Federation of Animal Science Societies, Champaign, IL). The Institutional Animal Care and Use Committee at the National Animal Disease Center reviewed and approved the animal use protocols (protocols 2514, 3765, 3849, 3913).
Inoculum preparation and animal procedures
Inocula used to challenge raccoons were selected to represent a wide range of natural and experimental TSE isolates from different donor species. Where possible, we selected TSE isolates that had caused disease in previous animal transmission studies.
Transmissible mink encephalopathy inocula
Raccoon-passaged TME (TMERac) inoculum was prepared from the brain of a raccoon that developed clinical TSE in a prior experiment 19 after inoculation of brain material from a raccoon that received the mink-passaged agent of TME 22 from the Stetsonville, WI outbreak of TME in farmed mink in 1985. 27 The TMERac inoculum is therefore from the second passage of the mink TME agent in raccoons. Bovine-passaged TME (TMEBov) inoculum was prepared from a Holstein steer that developed clinical signs of prion disease 19 mo after intracranial inoculation of mink brain homogenate from the Stetsonville farm. 27 The hamster-adapted drowsy (TMEDY) and hyper (TMEHY) inocula were prepared from Syrian hamsters that developed clinical TSE after intracranial inoculation of brain homogenate from hamsters in which the TME agent from the Stetsonville outbreak had been serially passaged. 6
Chronic wasting disease inocula
White-tailed deer (CWDWtd) inoculum was prepared from a pool of 11 CWD-affected white-tailed deer brainstems from Wisconsin. 23 All donor white-tailed deer were homozygous for glycine at PRNP codon 96. Elk (CWDElk) inoculum was prepared from a pool of 2 CWD-positive elk brainstems from South Dakota (equal parts MM132 and LM132 donor tissue). 23 The animals from which the inocula were prepared were all positive by immunohistochemistry (IHC) for PrPSc, and the pooled samples of both inocula were positive for PrPSc by western blot (WB).
Atypical scrapie inoculum
Atypical scrapie inoculum was prepared from a pool of cerebrum from 2 atypical scrapie-affected sheep from Norway (PRNP genotypes A136H154Q171/ARH and A136F141R154Q171/AFRQ). The brains of donor sheep were positive for PrPSc using validated statutory laboratory tests and in-house WBs.
Sourcing and husbandry of raccoons has been described previously. 20 Briefly, 8-wk-old raccoon kits were obtained from a commercial breeder. All raccoons were vaccinated against canine distemper virus. Raccoons were assigned to TME-inoculated (n = 10 TMERac [n = 5 intracranial, n = 5 intralingual], n = 4 TMEBov, n = 5 TMEDY, n = 5 TMEHY), CWD-inoculated (n = 4 CWDWtd, n = 4 CWDElk), atypical scrapie inoculated (n = 5), or non-inoculated control (n = 8) groups. All raccoons were inoculated intracranially with 0.1 mL of a 10% w/v inoculum as described previously, 20 except for 5 raccoons in the TMERac challenge group that were inoculated with 0.1 mL of the same 10% w/v inoculum injected into the cranial one-third of the tongue (intralingual). Non-inoculated negative control raccoons (n = 8) were housed in the same room as the inoculated raccoons but in separate pens. Raccoons were housed in a biosafety level 2 containment facility at the National Animal Disease Center (Ames, IA) and monitored daily for clinical signs of prion disease. Raccoons were euthanized when they showed unequivocal signs of prion disease, including ataxia, inability to climb, or recumbency; when euthanasia was necessary as a result of intercurrent illness or injury that could not be remedied by veterinary care; or at the end of the experiment at 82 mo (6.8 y) post-inoculation (MPI).
Postmortem examination was performed on all raccoons, including collection of 2 sets of tissue samples. One set of tissues included representative sections of: brain, eye (retina), optic nerve, sciatic nerve, trigeminal ganglion, lymph nodes (retropharyngeal, mesenteric, popliteal, prescapular), palatine tonsil, third eyelid, stomach, duodenum, jejunum, ileum, cecum, colon, recto-anal lymphoid tissue, salivary gland, liver, pancreas, kidney, urinary bladder, spleen, adrenal, pituitary, thyroid, skeletal muscles (diaphragm, biceps femoris, masseter, psoas major, triceps), heart muscle, tongue, turbinate, lung, trachea, foot pad, nasal planum, and anal sac gland. These tissues were fixed in 10% buffered formalin, embedded in paraffin wax, and sectioned at 5 μm for microscopy examination after hematoxylin and eosin (H&E) staining. The second set of tissues, comprising subsamples of all tissues collected into formalin, was frozen.
Prion protein gene polymorphisms
Frozen spleens from experimental raccoons were used for characterization of the raccoon prion protein gene polymorphisms as described previously. 22
Western blot
Samples were collected from the brainstem at the level of the obex, and WB was performed as reported previously. 11 WBs were developed using mouse anti-PrP monoclonal antibody 6H4 at a 1:10,000 dilution.
Antigen-capture enzyme immunoassay
Frozen brainstem samples were homogenized in 1× PBS at a concentration of 20% w/v and assayed with a commercial enzyme immunoassay (EIA; HerdChek BSE-Scrapie Ag test kit, IDEXX Laboratories, Westbrook, ME) as described previously. 45
Immunohistochemistry
All paraffin-embedded tissues were immunostained by an automated immunohistochemical method for detection of PrPSc as described previously 12 using the anti-PrP monoclonal antibody F99/96.7.1. 33
Results
Prion protein gene polymorphisms
Nucleotide polymorphisms were detected at 16 sites in the raccoon prion protein open-reading frame between codons 23 and 243, but only 2 of these changes resulted in amino acid substitutions: serine for glycine at codon 75 (n = 6) and asparagine for serine at codon 107 (n = 2).
Incubation time and clinical presentation
Raccoons in the TMERac and TMEBov (cases 1–4) challenge groups developed clinical disease at 6–8 mo after intracranial or intralingual inoculation (Table 1). Clinical signs included lethargy, lack of response to external stimuli, inability to climb up to their sleeping quarters, and recumbency.
Summary of results of experimental inoculation of raccoons with different prion isolates. Results from previous studies on the experimental inoculation of raccoons with the agent of chronic wasting disease from mule deer and classical scrapie from sheep are included for completeness.
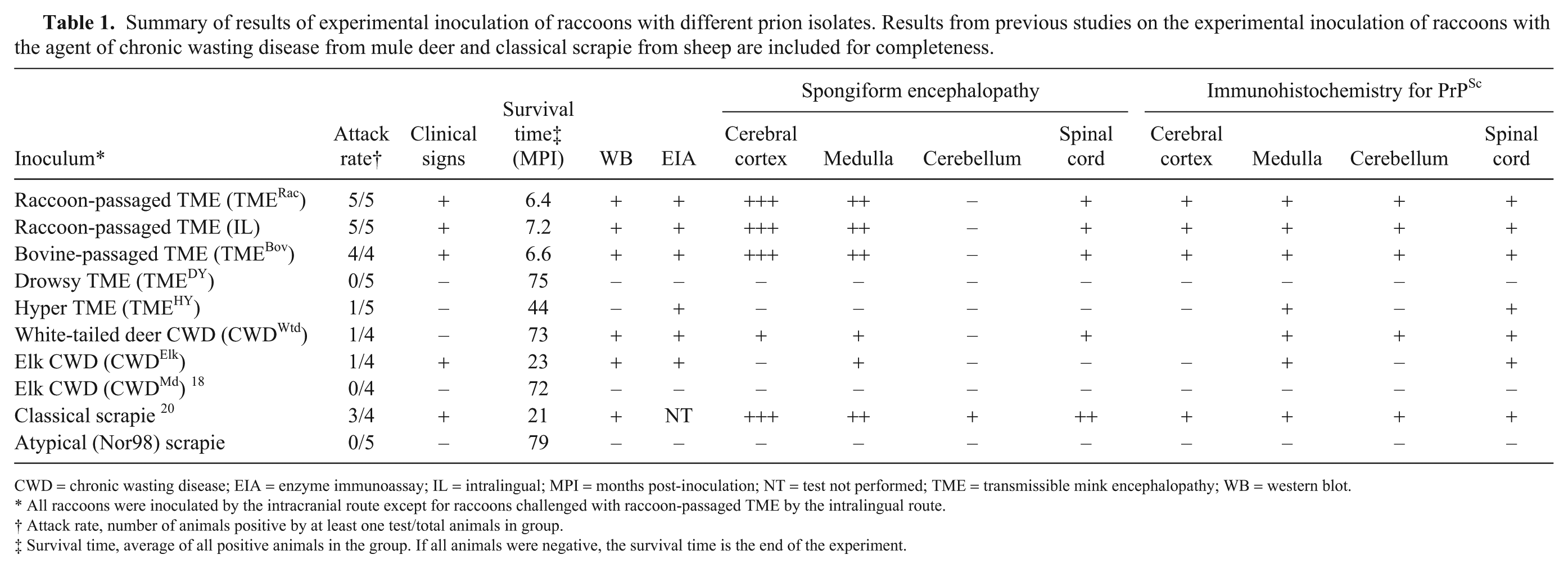
CWD = chronic wasting disease; EIA = enzyme immunoassay; IL = intralingual; MPI = months post-inoculation; NT = test not performed; TME = transmissible mink encephalopathy; WB = western blot.
All raccoons were inoculated by the intracranial route except for raccoons challenged with raccoon-passaged TME by the intralingual route.
Attack rate, number of animals positive by at least one test/total animals in group.
Survival time, average of all positive animals in the group. If all animals were negative, the survival time is the end of the experiment.
None of the animals inoculated with TMEDY, TMEHY, CWDWtd, or atypical scrapie agents developed clinical signs consistent with prion disease (Table 1), although PrPSc was detected in the brain of one of the raccoons inoculated with CWDWtd (case 5). Causes of intercurrent deaths in these groups included urinary obstruction (n = 2), hemorrhagic cystitis (n = 2), chronic skin lesions that were not responsive to treatment (n = 2), and uterine endometrial hyperplasia with multifocal cyst formation (n = 2). The remaining 3 raccoons in the TMEDY challenge group and 1 raccoon in the CWDWtd challenge group were euthanized at 75 or 82 MPI, respectively, when the experiment was terminated.
At 23 MPI, 2 raccoons in the CWDElk challenge group became recumbent and were euthanized. No significant lesions were observed at postmortem examination, but PrPSc was detected in the brain of one of these animals (case 6). The 2 remaining raccoons in the CWDElk challenge group were euthanized at the end of the study at 82 MPI.
Detection of PrPSc
To determine if raccoons inoculated with TSE agents accumulate misfolded prion protein in the CNS, we assayed the brainstem by WB, EIA, and IHC.
Western blot
Using WB, PrPSc was detected in brain samples from all raccoons inoculated with TMERac (data not shown) or TMEBov agents (cases 1–4; Fig. 1). PrPSc glycoform patterns were similar in raccoons inoculated with TMERac or TMEBov agents, and also similar to the TME inocula derived from raccoon and bovine, respectively. PrPSc was detected in one CWDWtd- (case 5) and one CWDElk- (case 6) inoculated raccoon, and the molecular profile was similar regardless of inoculum (Fig. 2). PrPSc was not detected on WB in any raccoons inoculated with TMEHY, TMEDY, or atypical scrapie.
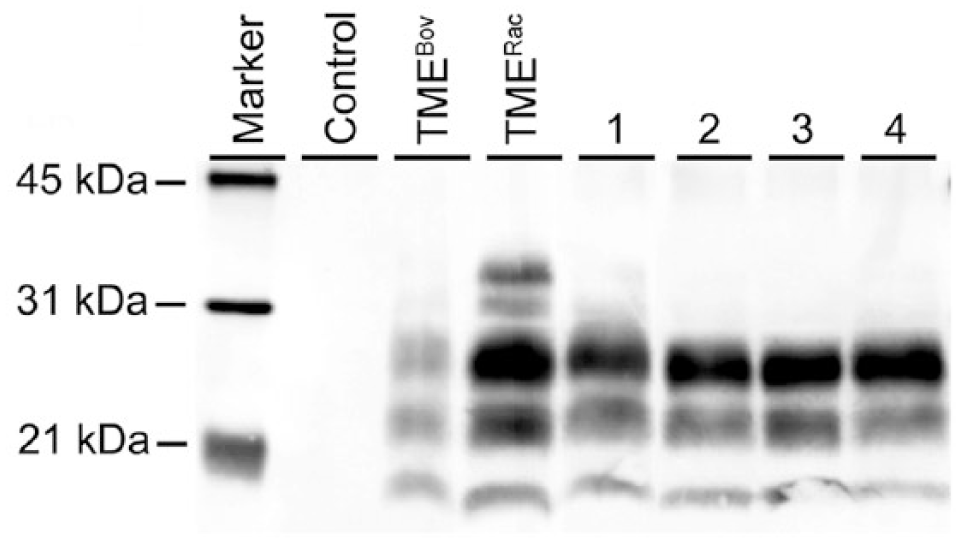
Western blot detection of PrPSc in brain homogenates from raccoons (1–4) inoculated intracranially with bovine-passaged transmissible mink encephalopathy (TMEBov). Control = non-inoculated control raccoon; TMERac = raccoon-passaged TME inoculum. Anti-PrP antibody 6H4.
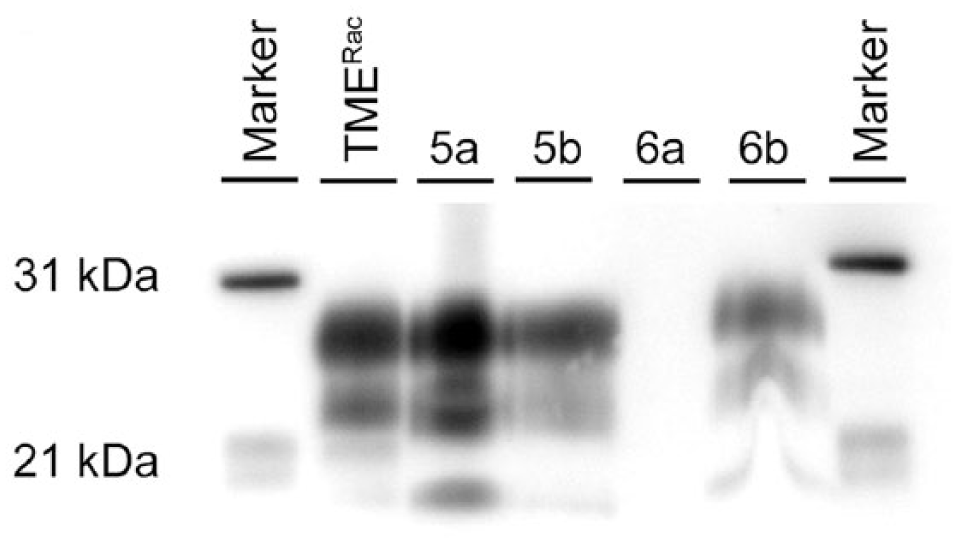
Western blot detection of PrPSc in brain homogenates from raccoons inoculated intracranially with the agent of chronic wasting disease from white-tailed deer (case 5) or elk (case 6). TMERac = positive control material from a raccoon inoculated intracranially with raccoon-passaged transmissible mink encephalopathy; a = cerebrum; b = obex. Monoclonal antibody 6H4.
Antigen-capture enzyme immunoassay
All raccoons that had a positive WB result also had a strong positive EIA result (optical density [OD] reading = 4.00; Table 1). One raccoon inoculated with TMEHY (case 7) was EIA-positive but with an OD = 0.447, which was much lower than the other EIA-positive raccoons. All remaining raccoons were negative by EIA with ODs of 0.094–0.173.
To determine if raccoons inoculated with various TSE agents develop spongiform lesions or accumulate misfolded prion protein in the brain, coronal brain sections were examined by light microscopy after H&E staining and by IHC.
Histopathology
Scattered large vacuoles were observed in the white matter of all raccoons examined, including non-inoculated control animals. This type of vacuolation was particularly prominent in the cerebellum and cerebrum. Disease-associated spongiform change was observed in all raccoons in the TMERac and TMEBov challenge groups, and raccoons 6 and 7 in the CWDWtd and CWDElk challenge groups, respectively.
In raccoons in the TMERac and TMEBov challenge groups, microscopic spongiform lesions were severe and diffuse. Spongiform change was most severe in the cerebral cortex, characterized by marked vacuolation of the neuropil and moderate gliosis without neuronal vacuolation or degeneration (Fig. 3). Vacuolation of the neuronal perikarya was most commonly observed in the brainstem and spinal cord.
The distribution of spongiform change in raccoon 5, which had evidence of PrPSc accumulation after inoculation with CWDWtd, was similar to raccoons in the TMERac and TMEBov challenge groups, but the severity of the spongiform change was generally mild-to-moderate. Spongiform change in the cerebral cortex was more severe rostrally. Vacuolation of the neuronal perikarya was observed in the deep gray matter of the midbrain, paraventricular nuclei of the thalamus and cerebral neocortex, and was most prominent in the red nucleus of the midbrain (Fig. 4). Intraneuronal vacuolation was not observed in the red nucleus in any other inoculated or non-inoculated control raccoons.
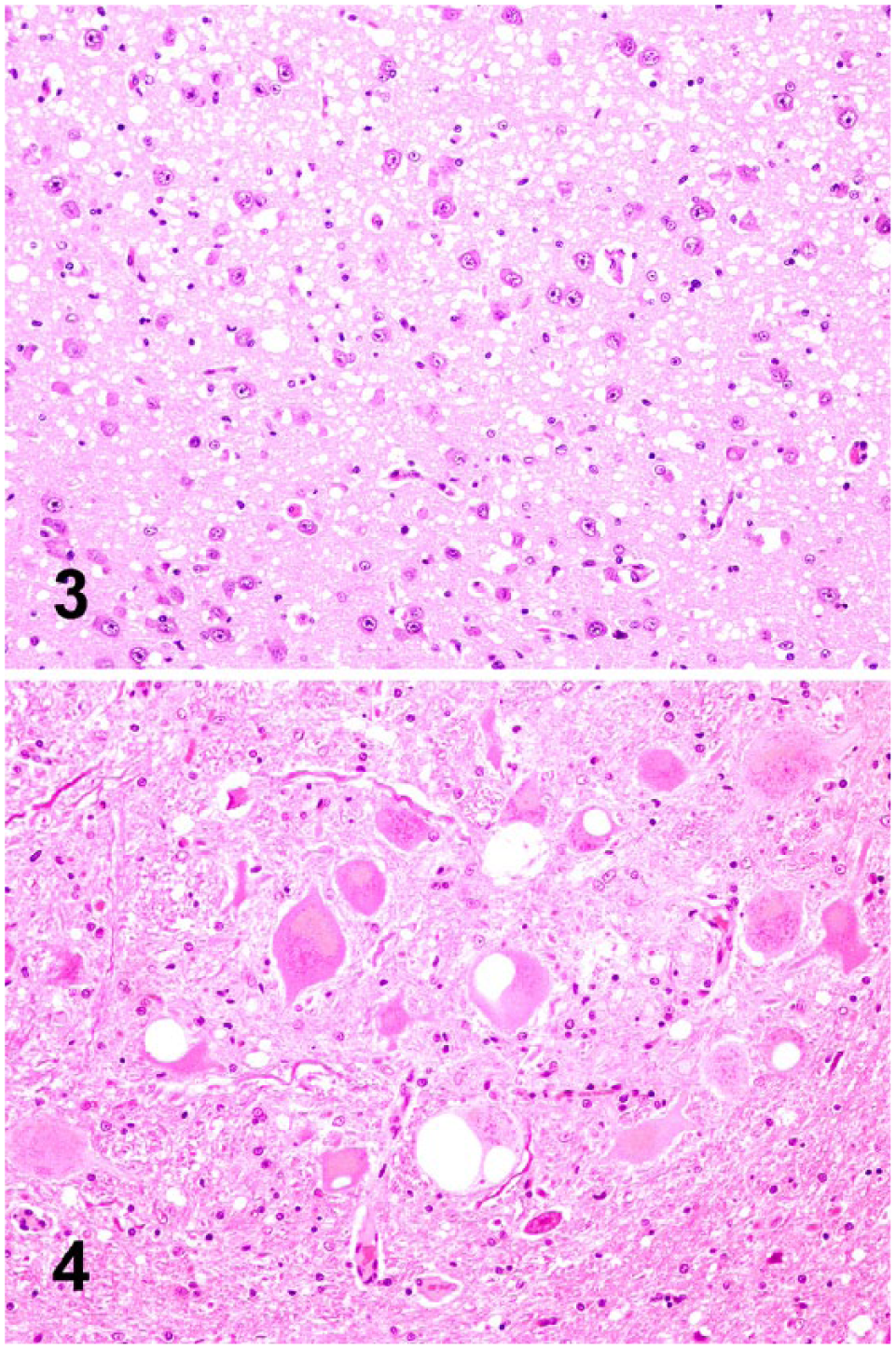
Spongiform change in the brains of experimentally inoculated raccoons. H&E.
In raccoon 6 (inoculated with CWDElk), spongiform change was mild-to-moderate with distribution observed only in the olivary nuclei in the medulla at the level of the obex, central gray matter of the midbrain, paraventricular nuclei and rostral ventral nucleus of the thalamus, hypothalamus, and caudate nucleus. No disease-associated spongiform change was observed in raccoon 7, which was inoculated with the TMEHY agent and had a positive EIA result on medulla at the level of the obex.
Immunohistopathology
Immunoreactivity for PrPSc was observed throughout the brain and spinal cord of all raccoons in the TMERac and TMEBov challenge groups. The overall amount of immunolabeling was greatest in raccoons inoculated with the TMERac agent via the intracranial route, but the anatomic distribution and range of morphologic immunolabeling types was similar across all 3 groups. Particulate, intraglial, and intraneuronal immunolabeling was present throughout the gray matter of the brain and spinal cord. Intraneuronal immunolabeling was more common caudally and was most prominent in the reticular formation, olivary nuclei (Fig. 5), pontine nuclei, and cerebellar roof nuclei. Perineuronal and linear immunolabeling was observed in a restricted number of neuroanatomic regions, including the reticular formation, central gray matter, basal ganglia (Fig. 6), olfactory cortex, and deep layers of the cerebral cortex (internal pyramidal and multiform layers). In the cerebral cortex, particulate and intraglial immunolabeling was present across all cortical layers, but the amount of particulate immunolabeling was greatest in the internal pyramidal layer (Fig. 7). In the cerebellar cortex, particulate and intraglial immunolabeling was present in the granular and molecular layers (Fig. 8). Particulate immunolabeling in the molecular layer was frequently present as linear formations extending from the Purkinje cell layer toward the superficial molecular layer.
The anatomic distribution of PrPSc in raccoon 5 (inoculated with CWDWtd) was similar to raccoons in the TMERac and TMEBov challenge groups, but there were some differences in the relative amount of PrPSc and the morphologic types of immunolabeling observed. Particulate and intraneuronal immunolabeling were common in the brainstem, whereas intraglial immunolabeling was rarely observed. Intraneuronal immunolabeling co-localized with intraneuronal vacuolation in the red nucleus of the midbrain (Fig. 9). Perineuronal and linear immunolabeling was widely distributed (Fig. 10, caudate nucleus). In the cerebral cortex, immunolabeling was mild-to-moderate. Intraglial, intraneuronal, and particulate labeling was widely distributed, whereas perineuronal and linear labeling (Fig. 11) was variably present and more common rostrally. Scant PrPSc was observed in the cerebellar cortex and, where present, consisted of particulate immunolabeling and occasional aggregates that were largely confined to the granule cell layer (Fig. 12). Marked intraneuronal, particulate, and linear immunolabeling was observed in the cerebellar roof nuclei.
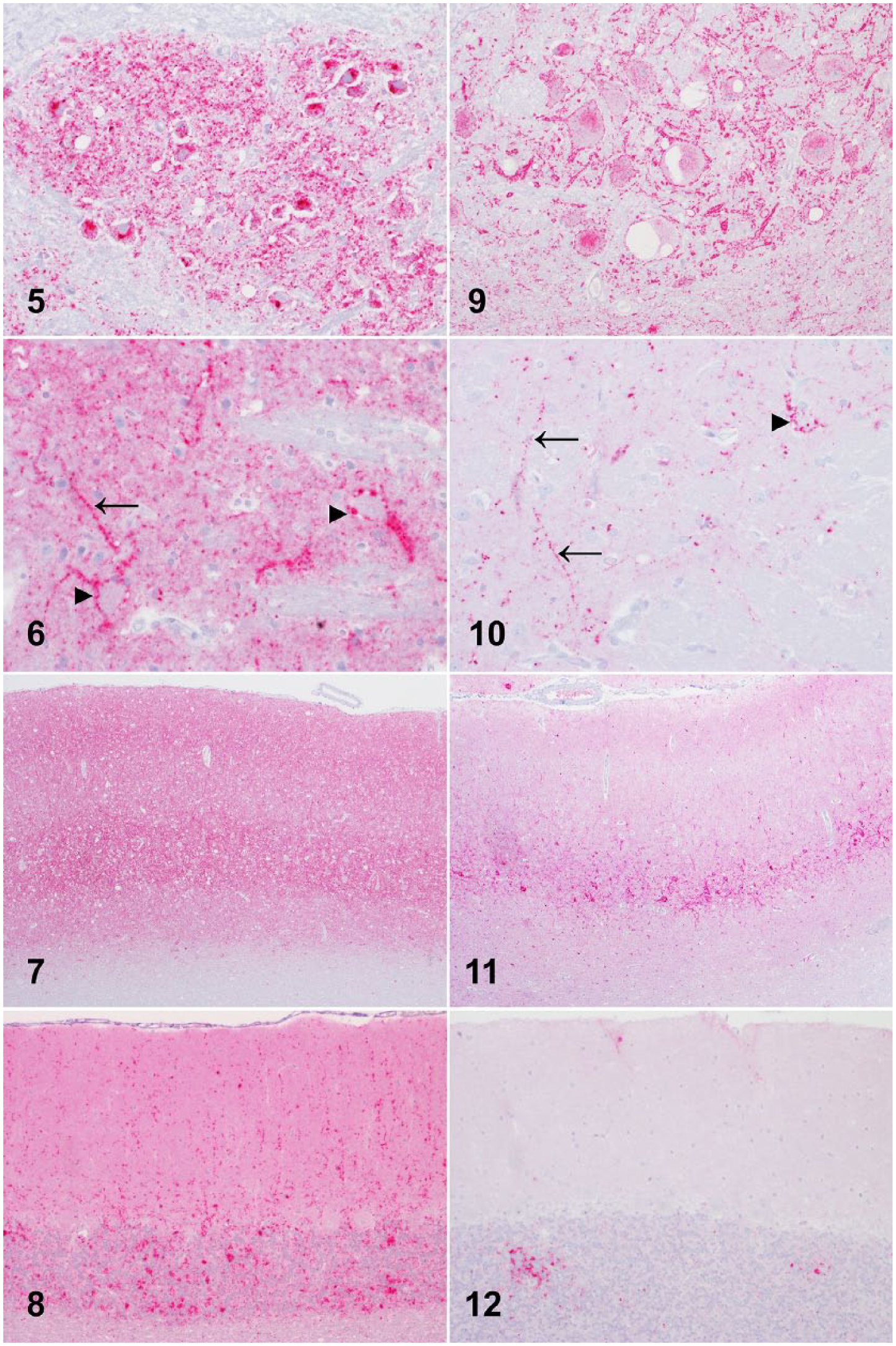
Immunoreactivity for PrPSc in the brains of experimentally inoculated raccoons. PrP detection using anti-PrP monoclonal antibody F99/97.
In raccoon 6 (inoculated with CWDElk), PrPSc was present throughout the brainstem, midbrain, and thalamus, and was restricted to very mild punctate, intraglial, and intraneuronal accumulations. Moderate amounts of PrPSc were observed in the ventral central gray matter of the midbrain, ventral hypothalamus, caudate nucleus (including linear and perineuronal types), and putamen (including aggregates). Disease-associated PrPSc immunoreactivity was not observed in the cerebellum or cerebral cortex. In raccoon 7 (inoculated with TMEHY), PrPSc accumulation was restricted to very mild particulate immunolabeling in the intermediolateral column of the spinal cord and the ventrolateral part of the nucleus of the spinal tract of the trigeminal nerve.
In addition to PrPSc accumulation observed in the brain and spinal cord, PrPSc immunoreactivity was detected in the retina, optic nerve, adrenal medulla, and occasionally the enteric nervous system of raccoons inoculated with the TMERac agent. The tissue distribution of PrPSc was similar for raccoons inoculated by the intracranial or intralingual routes. In raccoons inoculated with TMEBov, PrPSc immunoreactivity was seen in the retina and pituitary. PrPSc accumulation was not observed in any non-CNS tissues in raccoons inoculated with TMEHY, TMEDY, CWDWtd, CWDElk, or atypical scrapie agents.
Discussion
We demonstrated that raccoons inoculated intracranially with the agent of TME passaged in cattle (TMEBov) or raccoons (TMERac) readily develop clinical disease and accumulate disease-associated prion protein with incubation times of ~7 MPI. In contrast, inoculation of CWD isolates from white-tailed deer (CWDWtd) or elk (CWDElk), or hamster-passaged hyper TME (TMEHy), resulted in very low attack rates, extended incubation times, and variable clinical presentations and PrPSc accumulation. Intracranial inoculation of raccoons with TMEDY or atypical scrapie agents did not result in clinical disease or PrPSc accumulation up to study termination at 80 MPI (6.6 y post-inoculation).
The Stetsonville, WI outbreak of TME in farmed mink in 1985 27 provided the original challenge material used in bovine, raccoon, and hamster passage studies. Raccoons inoculated with TMEBov or TMERac agents showed a similar disease phenotype, whereas the disease phenotype in raccoons inoculated with hamster-passaged TME strains was very different, especially with regard to attack rate and PrPSc accumulation. Therefore, it would appear that serial passage of the mink-derived TME agent in hamsters results in a change in the biological characteristics of the TME isolate, leading to an increase in the species barrier to raccoons.
The disease phenotype in raccoons inoculated intracranially with the TMERac agent was similar to that reported for the 2 previous intracranial passages of this strain,19,22 thus demonstrating that the disease phenotype for TMERac remains stable through 3 serial passages in raccoons.
No differences were observed in the disease phenotype in raccoons inoculated with the TMERac agent by the intracranial or intralingual routes, which is consistent with results for intracranial versus intralingual challenge in sheep with scrapie. 15 In contrast, in hamsters inoculated with HY TME3,32 or scrapie 35 agents, intralingual inoculation tends to result in extended incubation times compared to intracranial inoculation. These discordant results may be related to different prion strain properties or host-dependent differences in, for example, prion trafficking.
Intracranial inoculation of raccoons with the agent of CWD resulted in variable disease phenotypes; the agent of CWD from mule deer (CWDMd) did not transmit, 18 whereas CWDWtd and CWDElk did transmit but with low attack rates (25%) and variable PrPSc accumulation. These differences in disease expression could be related to a number of factors, including variations in the amount of PrPSc or infectivity in the challenge inocula, PRNP genotype of donor and recipient animals, or biological variation in the characteristics of individual TSE isolates.
Sequencing of raccoon PRNP from experimental animals in our study revealed nucleotide polymorphisms resulting in amino acid substitutions at codons 75 (serine for glycine) and 107 (asparagine for serine). Codon 107 polymorphism has been reported previously and was considered relatively charge-neutral. 22 The same authors reported the substitution of asparagine for serine at codon 46, 22 but we did not detect this polymorphism in any of the raccoons in our study. There did not appear to be an association between PRNP polymorphisms and disease outcome, although statistical analysis was not possible given the small number of animals in each group.
Pooled samples of all 3 CWD inocula were positive for PrPSc by WB,21,23,40 indicating that PrPSc was present in all inocula. Furthermore, all 3 inocula have successfully produced prion disease when inoculated in cervid and non-cervid species (in the following description, all animals were inoculated by the intracranial route except when noted otherwise): the CWDMd inoculum in mule deer (oral inoculation), 40 cattle, 16 sheep, 17 white-tailed deer, 23 fallow deer, 14 and reindeer 30 ; the CWDWtd inoculum in cattle 21 and white-tailed deer 23 ; and the CWDElk inoculum in white-tailed deer, 23 fallow deer, 14 reindeer, 30 and elk 31 (oral inoculation).
All inocula used in our study were prepared from pooled brain material from multiple CWD-affected animals. Using bioassay, it has been shown that multiple CWD strains can be isolated from a single CWD-affected animal,1,8,36,38 so it is possible that the challenge inocula contained multiple prion strains that may have differed in their ability to amplify in different host species. For all 3 inocula, transmission to cervid species (mule deer, white-tailed deer, fallow deer, reindeer) was more efficient than transmission to non-cervid species (cattle, sheep), so the poor cross-species transmission of these inocula to raccoons is not surprising. In experiments in which multiple inocula were used to challenge groups of animals of a single species (white-tailed deer 23 or reindeer 30 ), no significant differences were observed in survival times, attack rates, or clinicopathologic features. However, in cattle experiments, the CWDWtd inoculum 21 was observed to produce higher attack rates and shorter incubation times than the CWDMd inoculum. 16 Experimental challenge of raccoons with single-source inocula from cervids of different species and PRNP genotypes may help to elucidate the relative contribution of these factors to transmission of the agent of CWD from cervids to raccoons.
Intracranial inoculation of raccoons with the atypical scrapie inoculum did not produce clinical disease or detectable PrPSc before study termination at 80 MPI. In contrast, classical scrapie readily transmits to raccoons following intracranial inoculation with average incubation times of 21.7 MPI and 14.3 MPI for first 20 and second 19 passages, respectively, and there is widespread spongiform change and PrPSc accumulation throughout the brain.
The atypical scrapie challenge inoculum was prepared from the cerebra of sheep that were positive for PrPSc using validated statutory laboratory tests and in-house WB. It has previously been demonstrated that, in cross-species transmissions, a lack of detectable PrPSc using WB or IHC is not necessarily indicative of a lack of infectivity.24,37 In raccoons inoculated with atypical scrapie, the EIA OD readings from all brain samples were at the low end of the negative range, which suggests that, if PrPSc is present, it is in very small amounts. Mouse bioassay is underway to investigate whether infectivity is present.
Atypical and classical scrapie in sheep differ in a number of features, including genetic factors involved in susceptibility and molecular and biochemical characteristics of PrPSc.4,5 Atypical scrapie isolates have been transmitted to sheep by intracranial34,41,42,44 or oral43,44 inoculation, and to transgenic mice expressing ovine2,13,26 or porcine 10 PRNP sequences. The lack of transmission of atypical scrapie to raccoons in our study indicates a strong transmission barrier and provides an additional point of difference in the pathobiologic characteristics of classical and atypical scrapie.
We demonstrated that bovine- and raccoon-passaged TME isolates transmit readily to raccoons, CWD isolates from white-tailed deer and elk and hamster-adapted hyper TME transmit poorly, and hamster-adapted drowsy TME and atypical scrapie agents do not transmit. Although intracranial inoculation is an unnatural route for exposure of raccoons to CWD infection, our experiment shows that the agent of CWD from various cervid sources has the potential for transmission to raccoons. Given that raccoons develop disease after intralingual inoculation with the raccoon-passaged TME agent, it is possible that this route of inoculation could also result in successful transmission of other prion isolates in this species (e.g., CWD). Oral transmission studies are necessary to investigate the potential susceptibility of naturally exposed raccoons to disease and determine if shedding in excreta occurs at any time during the incubation period.
Footnotes
Acknowledgements
We thank Martha Church, Joe Lesan, Leisa Mandell, Kevin Hassall, and Trudy Tatum for excellent technical support. We acknowledge the contributions of Drs. Amir Hamir and Richard Bessen to this study. The atypical scrapie inoculum was kindly provided by Sylvie Benestad. Disclaimer: Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. The USDA is an equal opportunity provider and employer.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded in its entirety by congressionally appropriated funds to the U.S. Department of Agriculture, Agricultural Research Service. The funders of the work did not influence the study design, data collection and analysis, decision to publish, or the preparation of the manuscript.
