Abstract
In this retrospective study, we describe the clinicopathologic and immunohistochemical findings in a series of primary central nervous system (CNS) neoplasms in African hedgehogs (Atelerix albiventris). Twelve CNS neoplasms were found among 762 African hedgehog submissions (1.6%) to a private diagnostic laboratory in an 18-y period. The median age of affected hedgehogs was 3.5 y. No sex predilection was found. Hindlimb paresis, weakness, and ataxia were the most commonly reported clinical signs. Gangliogliomas (n = 6) and astrocytomas (n = 5) were the most commonly observed neoplasms; one oligodendroglioma was found. Gangliogliomas were found in the cerebellar white matter (2 of 6), brainstem (4 of 6), cervical spinal cord (1 of 6), and frontal lobe (1 of 6); one metastasized to the tongue. Gangliogliomas were immunoreactive for neurofilament protein (NFP), glial fibrillary acidic protein (GFAP), S100, and CD34. All astrocytomas were gemistocytic, located in the cerebrum, and none of these neoplasms metastasized. Astrocytomas were positive for GFAP, S100, and CD34, but negative for NFP. The oligodendroglioma was located in the cerebrum, and was positive for S100, but negative for GFAP and NFP.
Introduction
African (Atelerix albiventris) and European (Erinaceus europaeus) hedgehogs are popular pets and zoological exhibit animals in the Americas, Europe, and some Asian countries. Primary neoplasms of multiple systems occur in hedgehogs and, according to a retrospective study, mammary carcinoma, lymphoma, and oral squamous cell carcinoma are most common in these species.9,17 To our knowledge, documentation of clinicopathologic features of primary central nervous system (CNS) neoplasms in African hedgehogs is limited to only a few single-case reports. These include an anaplastic astrocytoma in the spinal cord, 6 a desmoplastic ganglioglioma in the spinal cord, 23 and a cerebellar mixed glioma. 1 Thus, the objectives of our study were: 1) to describe the clinical and pathologic features of a series of CNS neoplasms in African hedgehogs; and 2) to describe the usefulness of currently available immunohistochemical markers to distinguish CNS neoplasms in the aforementioned species.
Materials and methods
Animals
Cases submitted to Northwest ZooPath (Monroe, WA) in 1997–2015 were considered for our study. Criteria for inclusion in the study consisted of African hedgehog cases with primary CNS neoplasms identified by microscopic examination.
Histopathology
All tissues received for histopathology were collected during postmortem examination performed by clinical veterinarians. Tissues were preserved in 10% neutral-buffered formalin for up to 5 d prior to being processed routinely. Tissues were sectioned at 5 μm, mounted on frosted glass slides, and stained with hematoxylin and eosin. Tumors were assessed using the World Health Organization histological classification of tumors of the nervous system of domestic animals. 11 A high-power (40×) objective with a field number of 26.5 was used to count mitotic figures in 7 representative fields (i.e., densely populated areas of the neoplasm, excluding areas of necrosis, hemorrhage, histologic artifacts, and edema). Thus, the diameter of the field in which mitotic figures were counted was 2.38 mm. 15 In one case (case 10), the neoplasm was very small, and the diameter of the field was limited to 1.34 mm.
Immunohistochemistry
An immunohistochemistry (IHC) panel was performed on 9 haphazardly selected cases that met the criteria of inclusion (cases 1, 3–5, 7–9, 11, and 12). The panel included antibodies to detect glial fibrillary acidic protein (GFAP), neurofilament protein (NFP), and S100 protein. Some cases (cases 4, 5, 8, 9, and 11) were labeled with CD34. Also, one ganglioglioma (case 1) was labeled with synaptophysin and vimentin. IHC was performed on a BOND-MAX automated staining system (Vision BioSystems; Leica, Bannockburn, Illinois) using the Bond polymer detection system (Vision BioSystems) with 3,3’-diaminobenzidine (DAB) as the chromogen for S100, NFP, and synaptophysin or on the Discovery Ultra automated staining system (Ventana Medical Systems, Tucson, AZ) using the UltraMap alkaline phosphatase red detection system (Ventana) for CD34, GFAP, and vimentin. All slides were counterstained with hematoxylin. Positive controls are shown in Supplementary Table 1. For negative controls, antibodies were replaced with homologous non-immune sera. CD34-positive immunoreactivity was interpreted in light of the expected positive staining results on control tissue. Sequence comparison between human CD34 and African hedgehog CD34 cannot be performed because of lack of sequence data for A. albiventris; however, based on staining patterns, the cross immunoreactivity is appropriate. Further information regarding immunohistochemical reagents can be found in Supplementary Table 1.
Results
A total of 762 African hedgehog submissions were identified from 1997 to 2015 at Northwest ZooPath. Submissions from 12 (1.6%) hedgehogs met the criteria of inclusion for our study (Table 1). Ten cases were submitted from zoological hedgehog collections, and 2 cases were from private veterinary clinics. Five hedgehogs were female, 5 male, and the sex was not known for 2 animals. The 10 hedgehogs were 1–5-y-old (mean: 3.5 y). The clinical history was known for 9 animals and included hindlimb paresis (n = 4), weakness (n = 3), ataxia (n = 2), and single animals with one of each: weight loss, forelimb paresis, lateral recumbency, inability to flex the forelimbs, quadriplegia, and blindness. Five hedgehogs were euthanized because of poor prognosis associated with severe ataxia, poor response to treatment, inability to walk, and progressive hindlimb paresis (cases 3, 5, 6, 9, and 11). Three animals were found dead (cases 4, 8, and 10). Concurrent diseases in hedgehogs with CNS neoplasms included nephritis (n = 6), spongiform change of the cerebellar white matter and brainstem (4), enteritis (3), myocarditis (2), cortical adrenal hyperplasia (2), cystitis (2), dermatitis (2), degenerative disc disease (2), and one of each of pancreatitis, hepatitis, hepatic lipidosis, encephalitis, cutaneous sarcoma, myocardial fibrosis, or sialadenitis.
Signalment and clinicopathologic findings in African hedgehogs with central nervous system neoplasms.

F = female; M = male; NA = not available.
Spayed.
Six gangliogliomas, 5 astrocytomas, and 1 oligodendroglioma were identified via histopathology. Gangliogliomas were recorded in the white matter of cerebellum (2 of 6), brainstem (3 of 6), cervical spinal cord (1 of 6), and cerebrum (1 of 6; Fig. 1). Gangliogliomas were composed of intermixed populations of neoplastic glial and neuronal cells (Fig. 2). Glial cells predominated in these neoplasms (~70–90% of the neoplastic cell population) and were polygonal to spindloid, with indistinct margins and scant-to-moderate amounts of eosinophilic cytoplasm. Their nuclei were round-to-oval, with finely stippled chromatin and a small basophilic nucleolus. No mitoses were observed in 2 cases (cases 5 and 6). For the remaining cases, the mitotic count was low (1–4 mitoses; cases 1–4). Neoplastic neuronal cells were polyhedral-to-polygonal with distinct margins and moderate-to-abundant cytoplasm. Some neoplastic neuronal cells contained variable amounts of lipofuscin. Nuclei were round-to-oval with finely stippled chromatin and had a small nucleolus. No mitoses were observed in the neoplastic neuronal cell population. A few of the neoplastic neuronal cells were binucleate. One ganglioglioma (case 4) metastasized to the tongue. Neoplastic neuronal cells were diffusely positive for NFP (Fig. 3, Table 2). The glial cell component of these tumors was diffusely positive for GFAP (Fig. 4, Table 2) and S100. In these tumors, the pattern of CD34 immunoreactivity was diffuse with positive cells having intense labeling of their surface membranes and distal processes, and included both glial and neuronal cell components (Fig. 5). Neoplastic neuronal cells were positive for synaptophysin (Fig. 6).
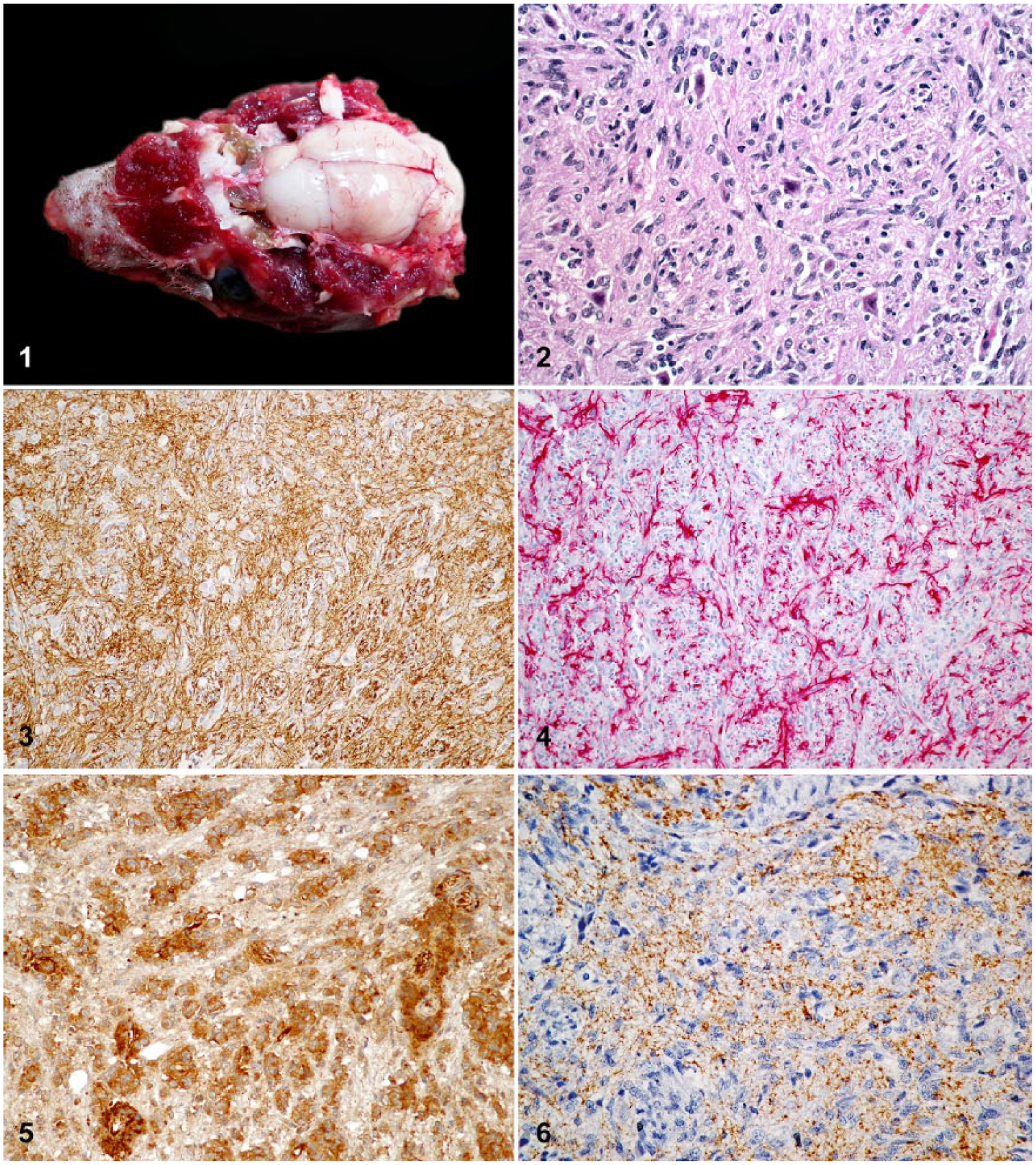
Gangliogliomas in African hedgehogs (Atelerix albiventris).
Immunohistochemical profiles of central nervous system neoplasms in African hedgehogs.

GFAP = glial fibrillary acidic protein; NFP = neurofilament protein; + = positive; − = negative.
All astrocytomas were located in the cerebrum. These neoplasms were composed of polygonal-to-polyhedral cells arranged in short fascicles and perivascular whorls (Fig. 7). Thus, these tumors were categorized as gemistocytic. Cells in astrocytomas had abundant eosinophilic cytoplasm with round-to-oval vesicular nucleus. No mitoses were detected. In one astrocytoma (case 7), there were broad areas of lytic necrosis that obscured ~40% of the neoplastic tissue. Astrocytomas were negative for NFP (Fig. 8) and diffusely immunoreactive for GFAP, S100 (Figs. 9, 10; Table 2), and CD34.
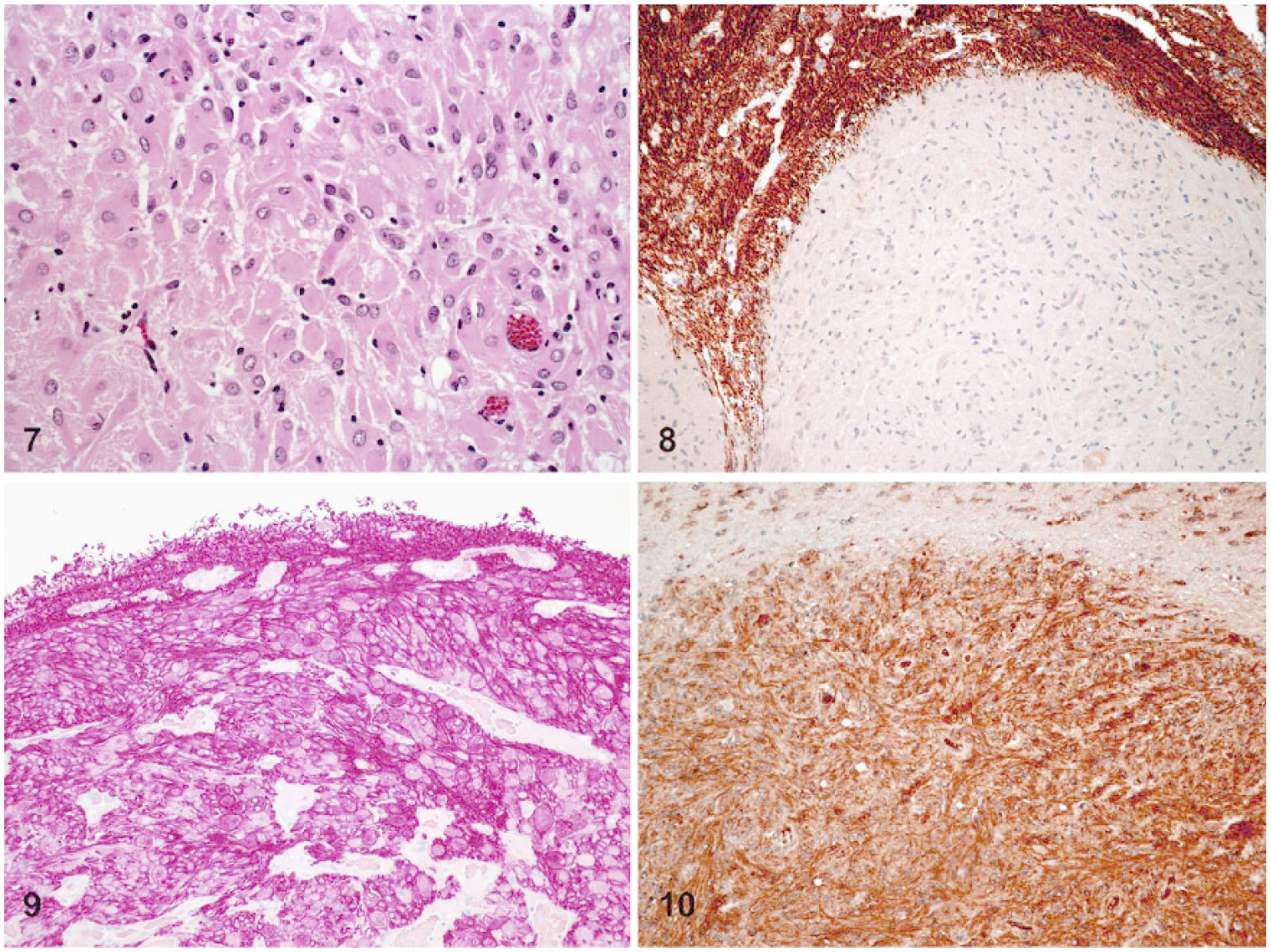
Gemistocytic astrocytomas in African hedgehogs (Atelerix albiventris).
The oligodendroglioma was located in the cerebrum (Fig. 11) and consisted of densely packed sheets of round-to-polygonal cells, with distinct margins and moderate amounts of eosinophilic cytoplasm with a clear perinuclear halo (fixation artifact; Fig. 12). Nuclei were round-to-oval and hyperchromatic. No mitoses were observed. There were multiple foci of mucinous degeneration and a few foci of microvascular proliferation throughout this tumor. This neoplasm was diffusely positive for S100 and negative for GFAP and NFP (Table 2).
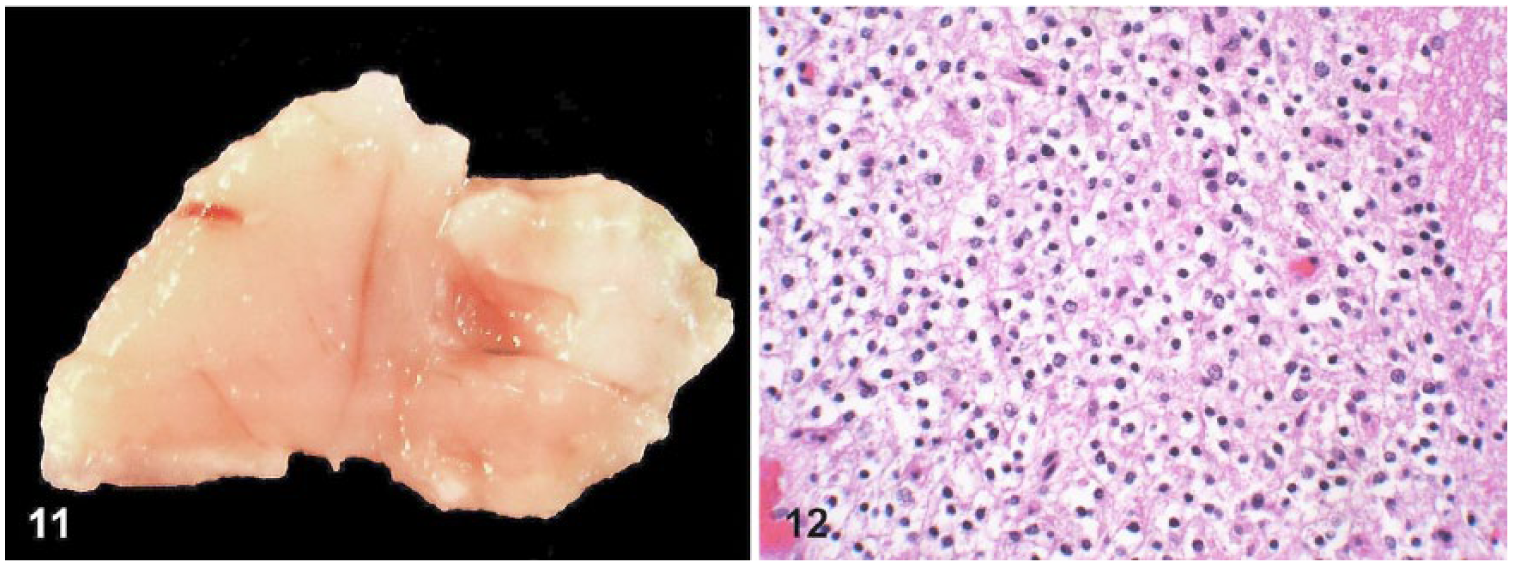
An oligodendroglioma in an African hedgehog (Atelerix albiventris).
Discussion
In our retrospective study, the relative prevalence of primary CNS tumors was low (~1.6% of the hedgehog submissions to Northwest ZooPath in an 18-y period). This finding is perhaps not surprising given that primary CNS neoplasms are considered rare in veterinary species. However, this relative prevalence is greater than expected based on brain tumor submissions in the mammalian archive at Northwest ZooPath, suggesting a possible predilection in hedgehogs for development of brain neoplasms (MM Garner, pers. comm., 2017). Although the incidence of primary brain neoplasms for veterinary species is undetermined, the prevalence of CNS neoplasms in dogs has been reported to be 2–4.5%.5,14,21 In our study, most CNS neoplasms were seen in adult animals, and only one astrocytoma was seen in a younger (1-y-old) hedgehog. The sex of most of these cases was evenly distributed, and sex predisposition appears to be unlikely. Hindlimb paresis, weakness, and ataxia were the most commonly reported clinical signs. Importantly, these clinical signs overlap with those seen in cases of wobbly hedgehog syndrome 7 and intervertebral disc disease. 16 Thus, for 5 of the cases reported in our study (cases 6–8, 10, and 11) it is unclear whether these signs were solely associated with the presence of a CNS neoplasm or to the result of other contributing factors (i.e., spongiform degeneration and intervertebral disc disease). Furthermore, practitioners should be aware that CNS neoplasms might clinically mimic other CNS diseases in African hedgehogs. The most common concurrent microscopic lesion was nephritis, which is unlikely to be directly associated with a CNS neoplasm.
Gangliogliomas and astrocytomas were the most common neoplasms identified in our study. Only one oligodendroglioma was found, and no embryonal, ependymal, choroidal, or meningeal tumors were diagnosed. Gangliogliomas are rarely documented in veterinary species with the only reported cases in the spinal cord of a calf and a European hedgehog, and the eyeball of a puppy.18,19,23 These tumors are composed of glial and neuronal cells that display various degrees of cellular atypia, but their precise cellular origin is unclear. It has been suggested that, in humans, gangliogliomas originate from a developmentally compromised or dysplastic precursor lesion.3,4 In humans, gangliogliomas are generally benign and uncommon; they primarily occur in children and young adults, and have a predominant supratentorial topographic distribution.4,13 In our study, all but one were located within the infratentorial region and were diagnosed in adult animals. The reason for the overrepresentation of gangliogliomas in adult African hedgehogs and the predominant infratentorial location of these neoplasms is undetermined. Given the age of the hedgehogs at which these gangliogliomas were diagnosed, it could be speculated that the cumulative effect of a carcinogenic agent (e.g., chemicals, infectious agents) or a hereditary component may play a role in the development of these neoplasms. Also, assuming that a precursor lesion is associated with the development of gangliogliomas in African hedgehogs (as suggested for their human counterpart)3,4 it could be speculated that such precursor lesion occurs more frequently within the infratentorial region. Further investigation, however, is required to determine a possible cause for these neoplasms. The immunohistochemical profile of gangliogliomas in African hedgehogs is similar to that reported for humans.4,13 Thus, immunohistochemistry for NFP, GFAP, and S100 may be used to support a diagnosis of ganglioglioma in African hedgehogs with CNS neoplasms composed of a combination of neuronal and glial cells.
Astrocytomas are considered to be the second most common primary CNS neoplasm in dogs, representing 17–28% of all neoplasms,8,20 and the telencephalon is the most common site for canine astrocytomas. 22 In our study, astrocytomas were the second most common primary CNS neoplasm, and all were found in the cerebral hemispheres. Also, the gemistocytic variant was the only one seen among these cases, and no medium-grade (anaplastic) or high-grade (glioblastoma) astrocytic neoplasms were observed. Precursor cells of the subventricular zone have been suggested as the most likely cell origin of gliomas. 24 The precise cell origin of astrocytomas in African hedgehogs is undetermined. In our study, microscopic examination was sufficient for the diagnosis of gemistocytic astrocytomas, but IHC for GFAP and S100 may be helpful for supporting a diagnosis of other histologic variants of astrocytic neoplasia (e.g., fibrillary and protoplasmic) as well as medium- and high-grade astrocytomas in African hedgehogs.
The oligodendroglioma found in our study largely recapitulates the histologic features of its canine counterpart. To date, no specific immunohistochemical markers have been found for oligodendrogliomas. Negative immunoreactivity for GFAP along with positive immunoreactivity for 2’,3’-cyclic nucleotide 3’-phosphodiesterase (CNPase) and oligodendrocyte transcription factor 2 (Olig2) has been used to characterize oligodendrogliomas in dogs. 10 In our study, S100 labeled neoplastic oligodendrocytes, but neoplastic cells were negative for GFAP and NFP. Thus, this IHC panel may be used to support the diagnosis of oligodendroglioma in African hedgehogs in challenging cases.
All astrocytomas and gangliogliomas tested for CD34 immunoreactivity in our study were positive. CD34 is a stem cell epitope that is not observed in normal human brain tissue, but is highly expressed in gangliogliomas and malformative CNS lesions,2,3,12 suggesting that these lesions arise from precursor cells that undergo abnormal glioneuronal differentiation. It would be interesting to investigate if CD34 is expressed in infratentorial, non-neoplastic lesions associated with CNS signs in African hedgehogs.
Supplemental Material
DS1_JVD_10.1177_1040638718793687 – Supplemental material for Primary central nervous system neoplasms in African hedgehogs
Supplemental material, DS1_JVD_10.1177_1040638718793687 for Primary central nervous system neoplasms in African hedgehogs by Juan F. Muñoz-Gutiérrez, Michael M. Garner and Matti Kiupel in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
