Abstract
An 8-mo-old male African pygmy hedgehog was anorectic and ataxic; physical examination revealed tetraparesis and a gangrenous left hindlimb. Analgesic and supportive care were administered, but the animal died 3 d after presentation. Postmortem examination revealed a histiocytic sarcoma in a mesenteric lymph node with metastasis to several organs, multifocal vacuolation in the cerebral and cerebellar white matter, and a meningioma in the left lateral ventricle. We diagnosed wobbly hedgehog syndrome (WHS) with disseminated histiocytic sarcoma and lateral ventricular meningioma. Ventricular meningioma, a rare neoplasm in veterinary and human patients, has not been reported previously in hedgehogs, to our knowledge. The neurologic signs in our case were probably caused by the WHS-related vacuolar lesions and are consistent with those of reported WHS cases. Duration of illness was shorter than is typical of WHS cases, which might be related to the disseminated histiocytic sarcoma. Clinical relevance of the lateral ventricular meningioma was not evident because the ventricular mass was localized and not invasive.
An 8-mo-old male African pygmy hedgehog (Atelerix albiventris) was presented to the Veterinary Teaching Hospital of Hokkaido University, Japan, with a 3-wk history of illness. Initial clinical signs were of reduced appetite and ataxia, and the animal’s condition deteriorated. The animal was tetraparetic and unable to stand or roll into a ball. Withdrawal reflexes were absent in the left hindlimb, and the soft tissues and skin of the hindlimb were malodorous, desiccated, and blackened, consistent with gangrene (Fig. 1A). No abnormalities were detected on radiography. Wobbly hedgehog syndrome (WHS) was suspected, and a poor prognosis was given. Despite supportive care, the hedgehog died 3 d later.
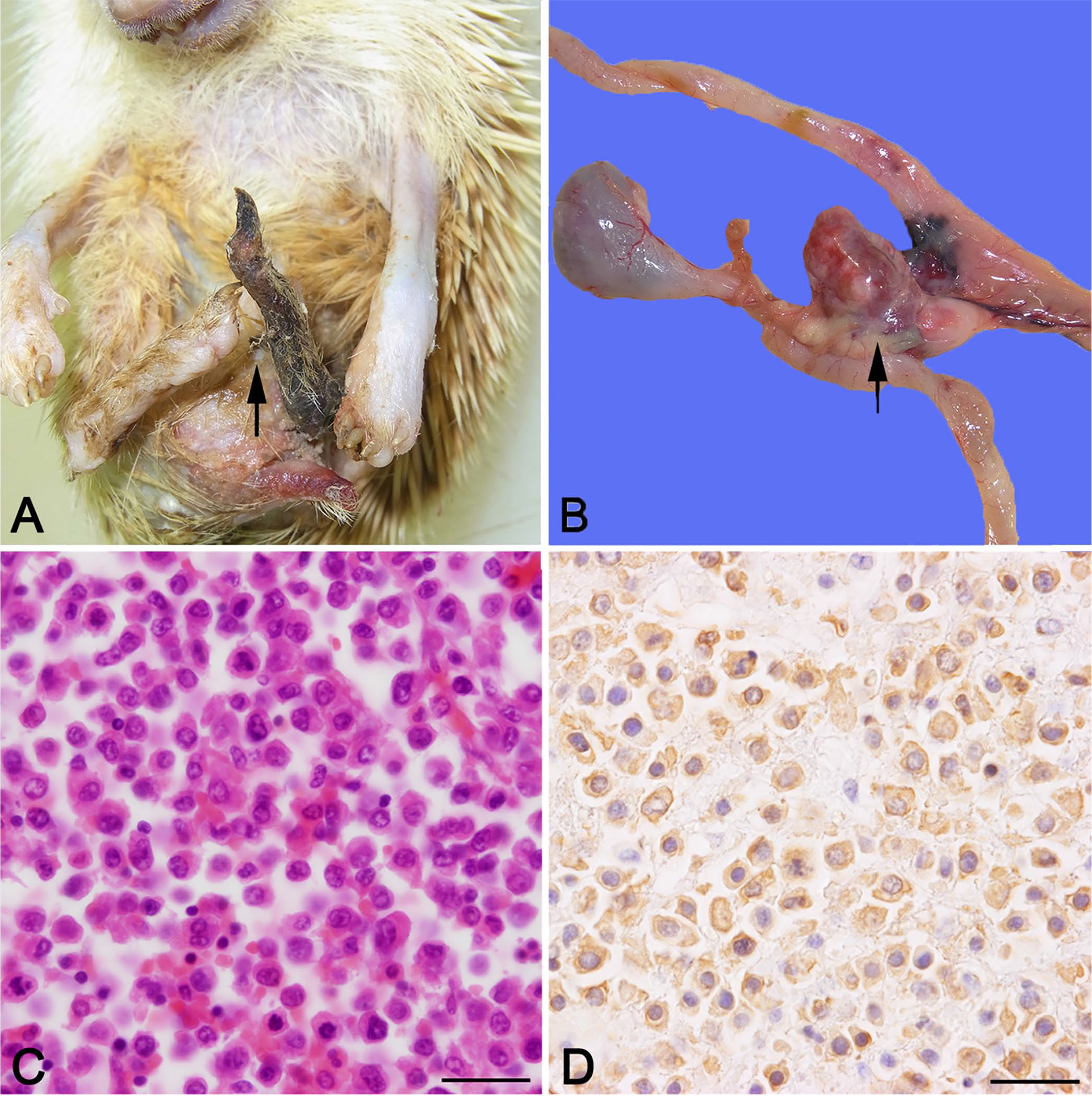
Gross images at autopsy and histologic images of the mesenteric mass in an African pygmy hedgehog.
At autopsy, an ulcerated area on the inner left forelimb and a large ulcer with suppurative exudate on the upper left hindlimb were noted. The left hindlimb was blackened and necrotic in the area distal to the stifle joint as noted clinically. The lungs (right cranial and left lobes) had 2 dark-red foci; splenomegaly was present. A large pale red-brown smooth lobulated soft mass (2.5 × 1.5 × 1.5 cm) in the mesentery was adherent to the pancreas and intestine (Fig. 1B). No gross abnormalities were noted in the brain or spinal cord. Representative samples of liver, spleen, kidney, heart, lung, brain, cervical spinal cord, mesenteric mass, and left hindlimb were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin or Luxol fast blue.
For immunohistochemistry, antigen retrieval was performed with citrate buffer at 95°C for 20 min. The antibodies used in immunohistochemical analysis were anti-cytokeratin AE1/AE3 (Dako), anti-vimentin (Abcam), anti-olig2 (Merck Millipore), anti–glial fibrillary acidic protein (GFAP; Dako), anti-Iba1 (Fujifilm Wako), anti-neurofilament (Dako), anti-CD3 (Dako), and anti-CD20 (Thermo Fisher Scientific). Immunolabeling was performed according to the manufacturer’s instructions. Labeled antigen was visualized with 3,3′-diaminobenzidine substrate (Dojindo Molecular Technologies), and sections were counterstained with hematoxylin.
Histologic evaluation of the left hindlimb showed that the epidermis, dermis, subcutis, and subcutaneous muscle were necrotic with marked infiltration of neutrophils, lymphocytes, plasma cells, and macrophages. The mesenteric mass corresponded to a mesenteric lymph node, which was replaced by sheets of neoplastic cells within a thin fibrovascular stroma. Neoplastic cells were large with abundant eosinophilic cytoplasm, indistinct cell borders, and oval nuclei (Fig. 1C). Anisokaryosis and anisocytosis were marked, and multinucleate giant cells were observed occasionally. Mitotic count was 50 per 2.37 mm2. Metastases of the neoplastic cells were recognized in the jejunum, ileum, liver, lung, spleen, and epicardium. Using immunohistochemistry, the neoplastic cells were positive for Iba1 (Fig. 1D) and negative for CD3 or CD20.
Although there were no gross findings in the central nervous system (CNS), histologic analysis showed bilateral vacuolation and demyelination in the white matter of the cerebrum and cerebellum, striatum, pons, and cervical spinal cord (Suppl. Fig. 1A). Vacuolation was evident also in the reticular formation, middle cerebellar peduncle, superior olive and periolivary nucleus in the pons, and in the ventral and lateral funiculi of the cervical spinal cord. In addition, neuronal degeneration was also observed in the ventral horn of the cervical spinal cord (Suppl. Fig. 1B). Surrounding the vacuolated regions, Iba1-positive microglia (Suppl. Fig. 1C) and GFAP-positive astrocytes (Suppl. Fig. 1D) were mildly increased. These lesions are consistent with WHS.
The dorsal wall of the left lateral ventricle was focally expanded by a well-demarcated mass that elevated the overlying ependymal cells (Fig. 2A). The mass was composed of sheets of neoplastic cells with faint stroma. The neoplastic cells had polygonal to spindle-shaped eosinophilic cytoplasm, indistinct cell borders, and oval clear nuclei (Fig. 2B). Anisokaryosis, anisocytosis, and mitosis were not observed. The neoplastic cells were positive for cytokeratin (Fig. 2C) and vimentin (Fig. 2D), but negative for GFAP, olig2, Iba1, and neurofilament. The neoplastic tissue was localized in the periventricular area and did not connect with meninges of the brain surface. These findings are consistent with lateral ventricular meningioma (meningothelial subtype).
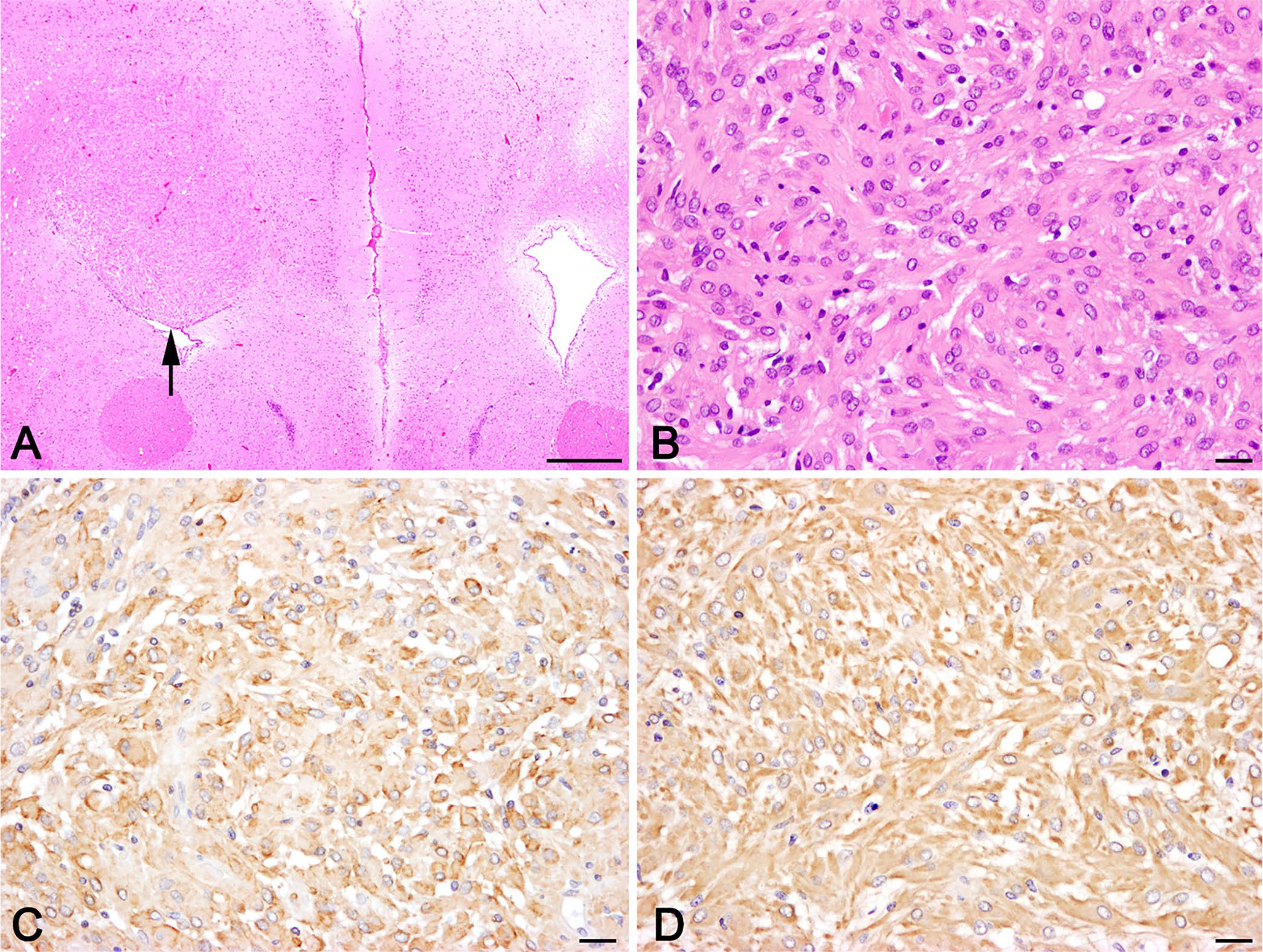
Histologic images of the lateral ventricular mass in an African pygmy hedgehog.
WHS in African pygmy hedgehogs was first described in 2006. 4 WHS has not been described in wild individuals but occurs frequently in pet hedgehogs, perhaps as a result of the low genetic diversity in pet hedgehogs. WHS is characterized by various neurologic clinical signs such as falling to one side, seizures, and paresis 4 ; these signs progress from mild ataxia to complete paralysis. Typical lesions of spongy myelinopathy are seen throughout the CNS, particularly in the cerebellum, medulla oblongata, and cervical and thoracic spinal cord. 3 Definitive diagnosis of WHS is made by postmortem histologic assessment. The neurologic signs in our case (i.e., ataxia, tetraparesis, and astasia) were probably caused by the WHS-related vacuolar lesions and not by the lateral ventricular meningioma because the ventricular mass was localized and not invasive. The neurologic lesions in our case might account for the gangrene in the distal part of the left hindlimb and ulcers on the left forelimb and hindlimb, which probably resulted from recumbency and chronic ischemia.
The neurologic signs and histologic findings in our case are consistent with those of reported WHS cases. Minor exceptions might be preservation of the thalamus and mild astrogliosis in our case. 3 The duration of illness from onset to death in our case was shorter than that of typical WHS cases, which may be related to the disseminated histiocytic sarcoma that was present in liver, spleen, lung, epicardium, and alimentary tract. Although histiocytic sarcoma is not a common tumor in African pygmy hedgehogs, several cases have been reported.5,9,10
To our knowledge, lateral ventricular meningioma has not been reported previously in hedgehogs. Ventricular meningioma is rare in the veterinary field as well as in human patients. Although the histopathologic features of the ventricular meningioma in our case resemble those of meningioma in the brain parenchyma of an African pigmy hedgehog, 7 the neoplastic tissue was localized in the periventricular area in our case, and a connection between the neoplastic tissue and meninges was not observed. The origin of ventricular meningioma is considered to be arachnoid cells in the choroid plexus stroma.6,13 During development, the choroid plexus is formed by invagination of the mesenchyme of the myelencephalon, and arachnoid cells are transported together with the invaginants. 13 Although meningioma-related neurologic signs were not evident in our case, ventricular meningioma increases intracranial pressure and can cause hydrocephalus. 1 Pet African pygmy hedgehogs frequently succumb to neoplasms at relatively young ages, and the CNS is often affected.2,5,8,11,12 Therefore, neoplasms should be included in differential diagnoses of hedgehogs showing neurologic signs, and thorough examination of the CNS is important even in cases with apparent WHS-related lesions.
Supplemental Material
Supplemental_material – Supplemental material for Wobbly hedgehog syndrome with disseminated histiocytic sarcoma and lateral ventricular meningioma in an African pygmy hedgehog
Supplemental material, Supplemental_material for Wobbly hedgehog syndrome with disseminated histiocytic sarcoma and lateral ventricular meningioma in an African pygmy hedgehog by Lesa A. Thompson, Atsuya Morita, Shoko Murakami, Noboru Sasaki, Miou Murashita, Ryou Yamazaki, Atsushi Kobayashi, Takashi Kimura and Mitsuyoshi Takiguchi in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Veterinary Teaching Hospital, Hokkaido University.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
