Abstract
Gangliogliomas are composed of neoplastic glial and neuronal cells and are extremely rare tumors of the central nervous system of domestic animals. The present report describes the clinical presentation and the pathomorphological and immunophenotypical characteristics of a desmoplastic ganglioglioma in the spinal cord of a 3-year-old male western European hedgehog (Erinaceus europaeus). Clinically, the hedgehog exhibited a skin wound and therapy-resistant paresis of the left hind limb. Necropsy showed dilatation of the urinary bladder. Histologic examination of the thoracic spinal cord revealed a focally extensive infiltrative mass, which consisted of multiple nodules of smaller bipolar or oligopolar glial cells and variably sized polygonal, ganglionic, neuron-like cells embedded in variable amounts of microcystic neuropilic matrix. An area of spindle-shaped cells arranged in interwoven fascicles and surrounded by a prominent network of reticulin fibers was interpreted as desmoplastic leptomeningeal stroma. Immunohistochemistry revealed a moderate number of glial fibrillary acidic protein and S-100-positive cells and processes. In addition, the ganglionic neuron-like cells expressed neurofilament, microtubule-associated protein-2, and neuron-specific enolase. In summary, this spinal cord tumor was composed of astroglial and neuronal cellular elements, justifying the diagnosis of a desmoplastic ganglioglioma.
The western European hedgehog (Erinaceus europaeus) is a common pan-European free-ranging animal protected by wildlife conservation laws, which, in contrast to its relative, the middle-African hedgehog (Atelerix albiventris), is generally not kept as a pet. Therefore, little is known about the type and frequency of its naturally occurring neoplasms. 5 The reported tumors of the nervous system of the western European hedgehog include 1 malignant peripheral nerve sheath tumor and 1 undifferentiated brain tumor. 5 Gangliogliomas are usually benign tumors of the central nervous system composed of neoplastic glial and neuronal cells and are extremely rare tumors in all species of domestic animals. 9 Reported cases include 1 case from the spinal cord of a 4-month-old calf 15 and 1 case from the intracranial and extracranial facial nerve and medulla oblongata region in a 7-month-old heifer. 13 Four cases have been reported in dogs, including 1 juvenile 18 and 2 adult cases involving the brain 10 and 1 juvenile case involving the eye. 16 Similarly, gangliogliomas and gangliocytomas are rare tumors of humans, accounting for approximately 0.4% of all primary central nervous system neoplasms. 2 The most commonly affected areas are the temporal lobes and the cerebellum with rare occurrence within the spinal cord, with a predilection for the cervical region. 8,11 Notably, there is a higher incidence of the morphologically related neoplastic entities, termed desmoplastic infantile astrocytoma and desmoplastic infantile ganglioglioma, which compose up to 16.0% of all intracranial tumors in infants. 3 These large cystic tumors of infants involve the superficial leptomeninges and cerebral cortex and are frequently attached to the dura mater. These tumors are composed of a prominent desmoplastic stroma with a neuroepithelial population, restricted to neoplastic astrocytes or astrocytes combined with a variable neuronal component, respectively. 3,4 The objective of the current report is to describe the clinical, pathomorphological, and immunohistochemical appearance of a desmoplastic variant of ganglioglioma in an adult western European hedgehog.
A male western European hedgehog of unreported age was presented for paresis and a skin wound on the dorsal aspect of the left metatarsal region. Since radiographs of the hind limbs failed to display pathological alterations of the skeleton, an underlying neurological problem was suspected. The skin wound was surgically corrected, and the animal was treated for 4 weeks with antibiotics and vitamin B complex. Because of a lack of clinical improvement and the poor prognosis, the hedgehog was euthanized and submitted for necropsy. Tissue specimens were fixed in 10% neutral buffered formalin. The vertebral column containing the spinal cord was decalcified in 10% ethylenediamine tetra-acetic acid (EDTA) solution, as described. 19 Subsequently, tissue specimens were processed, embedded in paraffin wax, sectioned at 2–4 μm, and stained with hematoxylin and eosin, periodic acid-Schiff (PAS), Luxol fast blue, cresyl violet, Azan, and Gomori and Bielschowsky silver stains. Immunohistochemical examination of serial sections was performed with the avidinbiotin-peroxidase complex method a using monoclonal antibodies specific for α-smooth muscle actin (clone 1A4, diluted 1:250), b pan-cytokeratin (clone AE1/AE3, diluted 1:500), b microtubule-associated protein-2 (clone AP20, diluted 1:50), c neurofilament (clone 2F11, diluted 1:200), b neuronal nuclei (NeuN; clone A60, diluted 1:100), d neuron-specific enolase (clone VI-H14, diluted 1:100), b vimentin (clone V9, diluted 1:20), b and Ki-67 (clone MIB-1, diluted 1:100), e as described. 5,20 Furthermore, polyclonal antibodies specific for glial fibrillary protein (GFAP; diluted 1:2000), b factor VIII-related antigen (diluted 1:200), b myelin basic protein (diluted 1:800), c and S-100 protein (diluted 1:800) f were used. The specificity of the immunohistochemical reactions was confirmed by the use of normal western European hedgehog brain, muscle, and skin. As negative controls, serial sections were treated with a mouse immunoglobulin Gl-negative isotype control antibody (CBL600) c or rabbit serum f instead of the primary monoclonal or polyclonal antibodies, respectively.
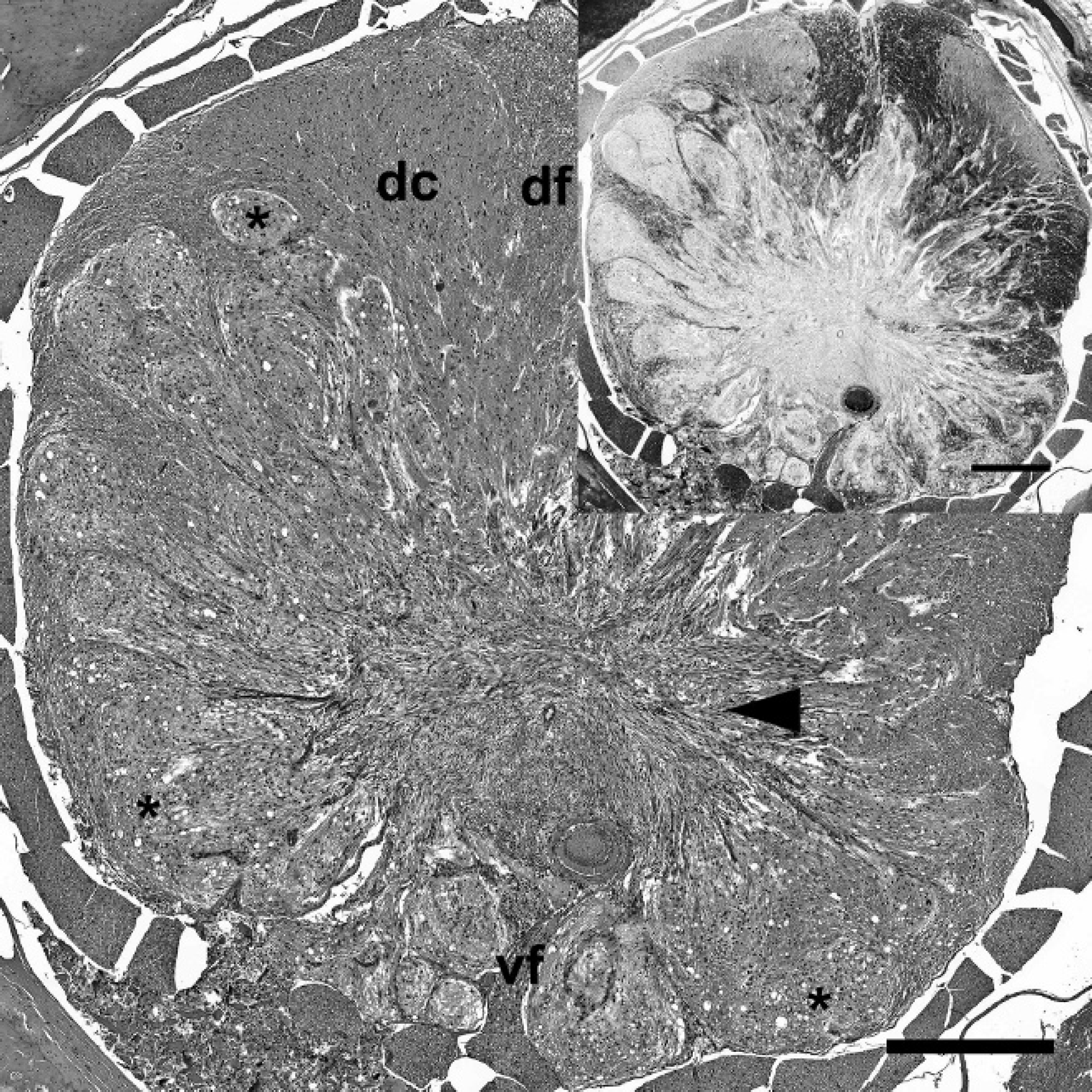
Spinal cord; western European hedgehog (Erinaceus europaeus). Desmoplastic ganglioglioma showing focally extensive infiltrative growth. The mass is composed of multiple nodules with microcystic neuropilic matrix (asterisks) and desmoplastic leptomeningeal stroma (arrowhead). dc = dorsal column of the gray matter; df = dorsal funiculus of the white matter; vf = ventral fissure. Hematoxylin and eosin. Bar = 1,000 μm. Inset, serial section. No Luxol fast blue-positive myelin sheaths or cells are present within the mass. Luxol fast blue. Bar = 500 μm.
On postmortem examination, the hedgehog was in good body condition and weighed 1.08 kg. The urinary bladder was severely dilated and filled with approximately 70 ml of straw-colored, clear, dilute urine. Histopathological examination of the thoracic spinal cord revealed an infiltrative, partially demarcated, multinodular mass, focally effacing more than 70% of the cross-sectional area, including the entire ventral and lateral funiculi of the white matter, the ventral columns of the gray matter, and the central canal (Fig. 1). The mass consisted of multiple low to moderately cellular nodules extending into the normal location of the ventral and lateral funiculi and a centrally located, moderately cellular, stroma-rich area at the dorsal end of the ventral fissure (Fig. 1). The nodules were composed of heterogeneous populations of smaller bipolar or oligopolar cells intermixed with single or nests of variably sized polygonal cells embedded in variable amounts of micro-cystic neuropilic matrix (Fig. 2). The smaller bipolar or oligopolar cells had ill-defined cellular borders; small to moderate amounts of occasionally vacuolated eosinophilic cytoplasm; centrally located, oval to elongated nuclei with finely granulated chromatin; and indistinct nucleoli. Some of the bipolar or oligopolar cells had brightly eosinophilic, elongate cytoplasmic inclusions, interpreted as Rosenthal fibers (Fig. 3). The intermixed polygonal cells were interpreted as ganglionic neuron-like cells. These cells were up to approximately 50 μm in diameter, with distinct borders and moderate to abundant amounts of eosinophilic cytoplasm. Nuclei were slightly eccentric, large, round, and euchromatic, with 1 or 2 prominent nucleoli (Fig. 2). Between the multifocal nodules described above and the ventral fissure, a moderately cellular area composed of interwoven fascicles of parallel-oriented spindle-shaped cells within a moderately increased amount of a fibrillar extracellular matrix was interpreted as desmoplastic leptomeningeal stromal component (Fig. 4). Overall, there was a moderate to high degree of anisocytosis and anisokaryosis. No mitoses were seen. The adjacent thoracic spinal cord displayed marked compression atrophy and a moderate diffuse lymphohistiocytic meningoleukomyelitis with prominent spheroids, dilated myelin sheaths, and gitter cells. Luxol fast blue-positive myelin sheaths were not detected within the mass (Fig. 1). Cresyl violet-stained sections displayed coarsely granular cytoplasmic components resembling Nissl substance within the larger polygonal cells. Bielschowsky-stained sections displayed intensely labeled neuronal processes, sometimes arranged in haphazard orientation or forming brush-like bundles, especially in the nodular areas (Fig. 5). Most of the larger polygonal cells displayed a moderate to intense, intracytoplasmic staining pattern. In contrast, Gomori silver stain revealed a prominent reticulin network surrounding most of the cells in the desmoplastic area and slightly weaker in the nodular areas (Fig. 6). Furthermore, Azan-stained sections demonstrated the presence of a delicate fibrillar extracellular matrix within the desmoplastic area only. The PAS reaction mainly stained the intralesional perivascular basal laminae. A moderate number of uniformly, pan-cytoplasmically GFAP-positive as well as S-100-positive bipolar or oligopolar cells was detected in the nodular areas (Fig. 7). In contrast, only few longitudinally oriented, GFAP-positive processes were detected within the desmoplastic area. Furthermore, expression of neurofilament (Fig. 8), microtubule-associated protein-2 (Fig. 9), and neuron-specific enolase was detected in multiple small to medium-sized axons, spheroids, and the large polygonal ganglionic neuron-like cells in the nodular areas. An average of 16.8 Ki-67-positive nuclei per mm 2 (21 Ki-67-positive nuclei within 20 high-power fields) was observed within the mass, compared with no positive nuclei per mm 2 in the unaffected cervical spinal cord. Some of these Ki-67-positive nuclei displayed a round to oval shape, a diameter of up to 20 μm, a moderately condensed, granulated chromatin, indistinct nucleoli, and they were situated within cell bodies with rather indistinct cytoplasm and cellular borders, interpreted as bipolar or oligopolar glial cells (Fig. 10). Furthermore, other Ki-67-positive nuclei displayed a round to elongated shape, a smaller diameter of up to 10 μm, a more condensed chromatin pattern, and scant cytoplasm, interpreted as invading leukocytes or microglia. There was a slightly increased amount of factor VIII-related antigen and α-smooth muscle actin-positive vascular stromal elements. Single spindle-shaped cells and processes within both areas of the mass were vimentin positive. No vimentin-positive glial cells were detected in the normal-appearing spinal cord and brain. In contrast, weak multifocal vimentin-labeling was observed in the meninges, besides a more pronounced signal in endothelial cells and multiple different mesoderm-derived cell types in the subcutis. The neoplastic cells were negative for myelin basic protein and pan-cytokeratin. The applied monoclonal anti-NeuN antibody revealed no immunoreactivity within EDTA-decalcified spinal cord sections, despite a normal neuronal labeling pattern in simultaneously processed, nondecalcified sections of the brain. Further histological findings in the central nervous system included a mild multifocal lymphohistiocytic meningoleukomyelitis in the cervical spinal cord. The brain and both sciatic nerves appeared normal. In addition, mild lymphohistiocytic interstitial pneumonia, periportal hepatitis, and pyelonephritis of unknown origin were observed. The subperiosteal bone of the mandibles displayed 3 incremental rings, indicative of the animal's age.
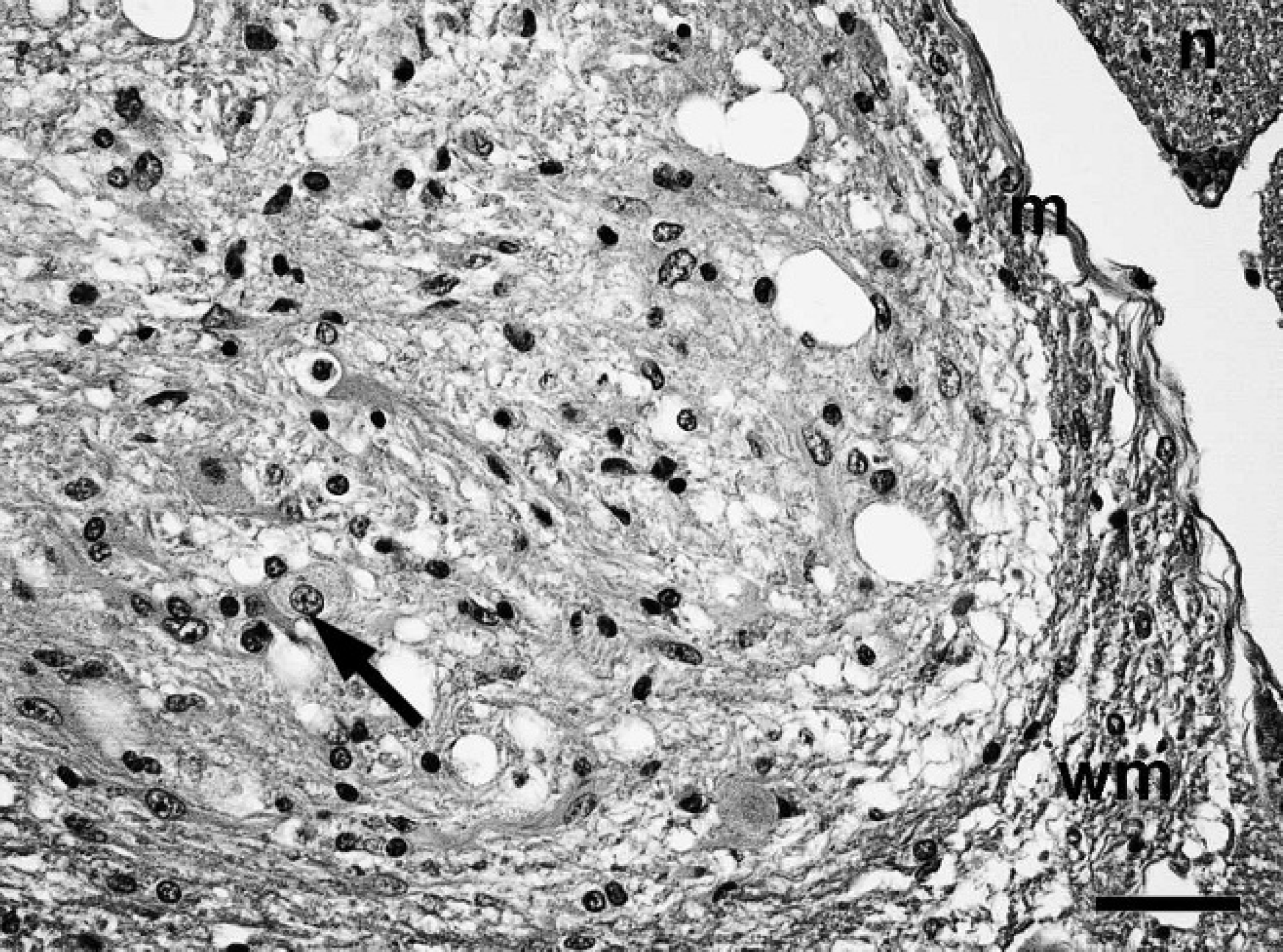
Spinal cord; western European hedgehog (Erinaceus europaeus). Nodules consisting of bipolar or oligopolar glial cells and polygonal ganglionic neuron-like cells (arrow) in a microcystic neuropilic matrix. m = meninx; n = spinal nerve root, wm = white matter. Hematoxylin and eosin. Bar = 40 μm.
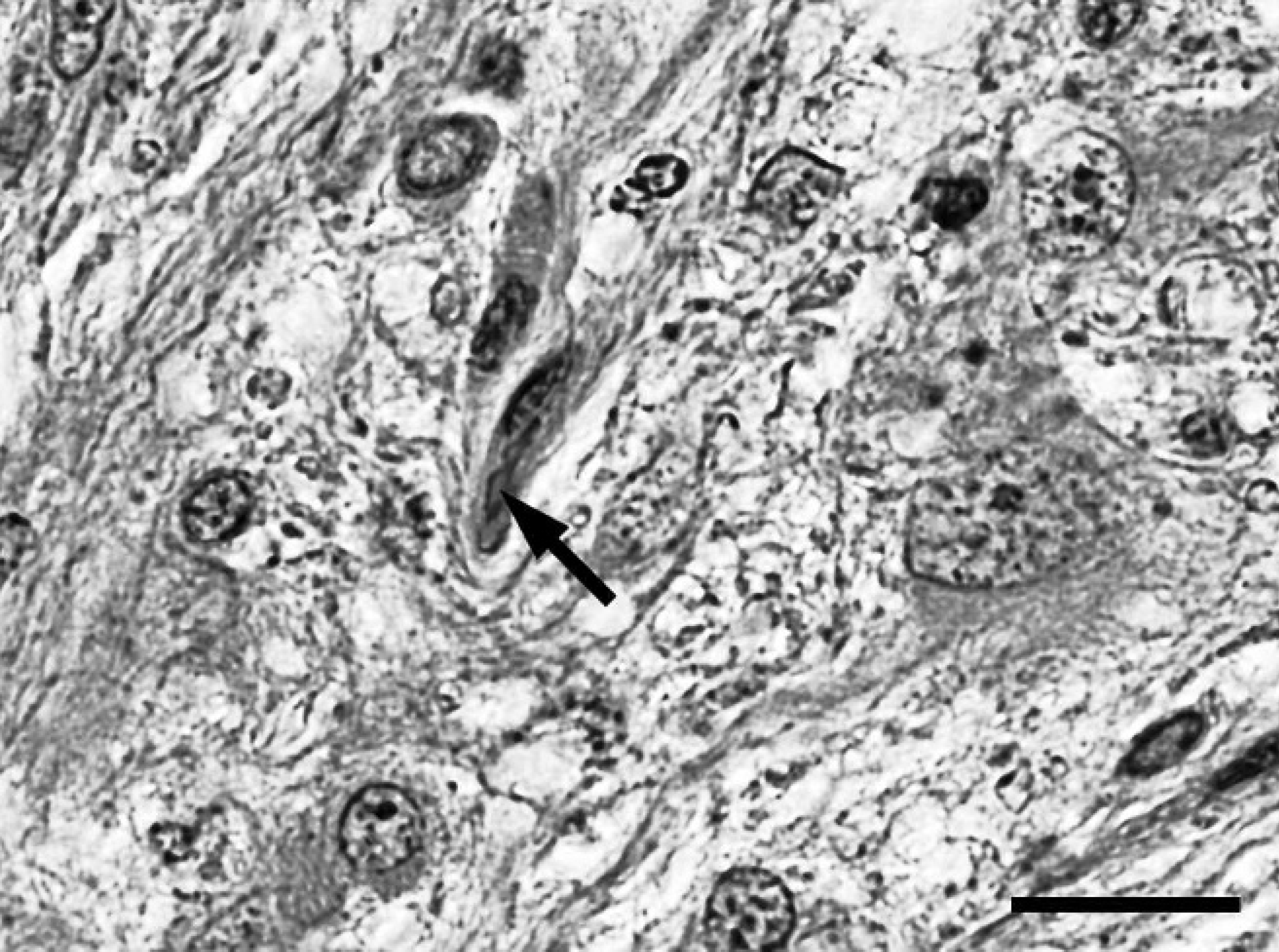
Spinal cord; western European hedgehog (Erinaceus europaeus). A bipolar astroglial cell containing an elongated intracytoplasmic brightly eosinophilic inclusion interpreted as Rosenthal fiber (arrow). Hematoxylin and eosin. Bar = 20 μm.
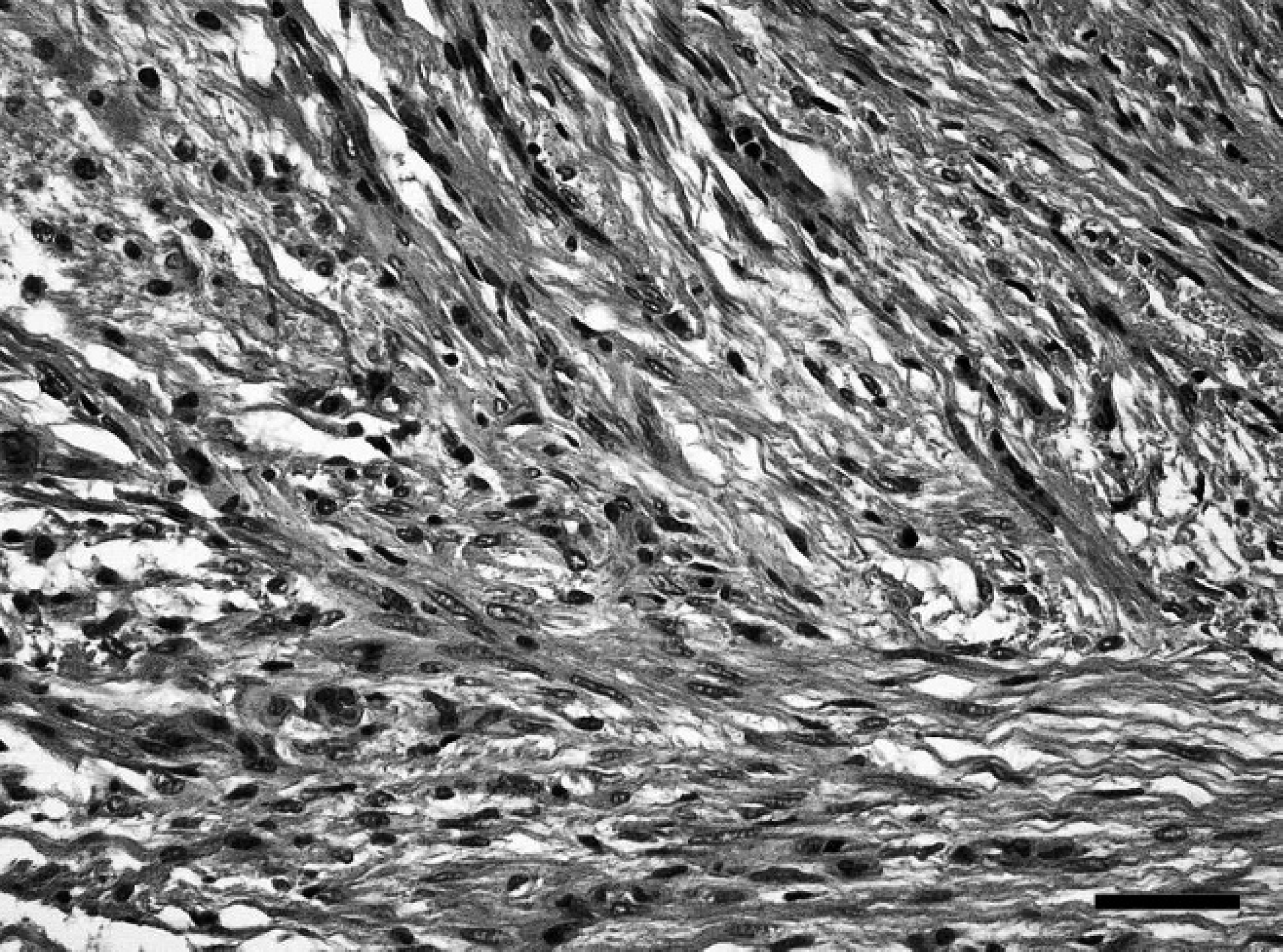
Spinal cord; western European hedgehog (Erinaceus europaeus). Area of spindle-shaped cells arranged in interwoven fascicles within moderate amounts of fibrillar matrix, consistent with desmoplastic stroma. Hematoxylin and eosin. Bar = 40 μm.
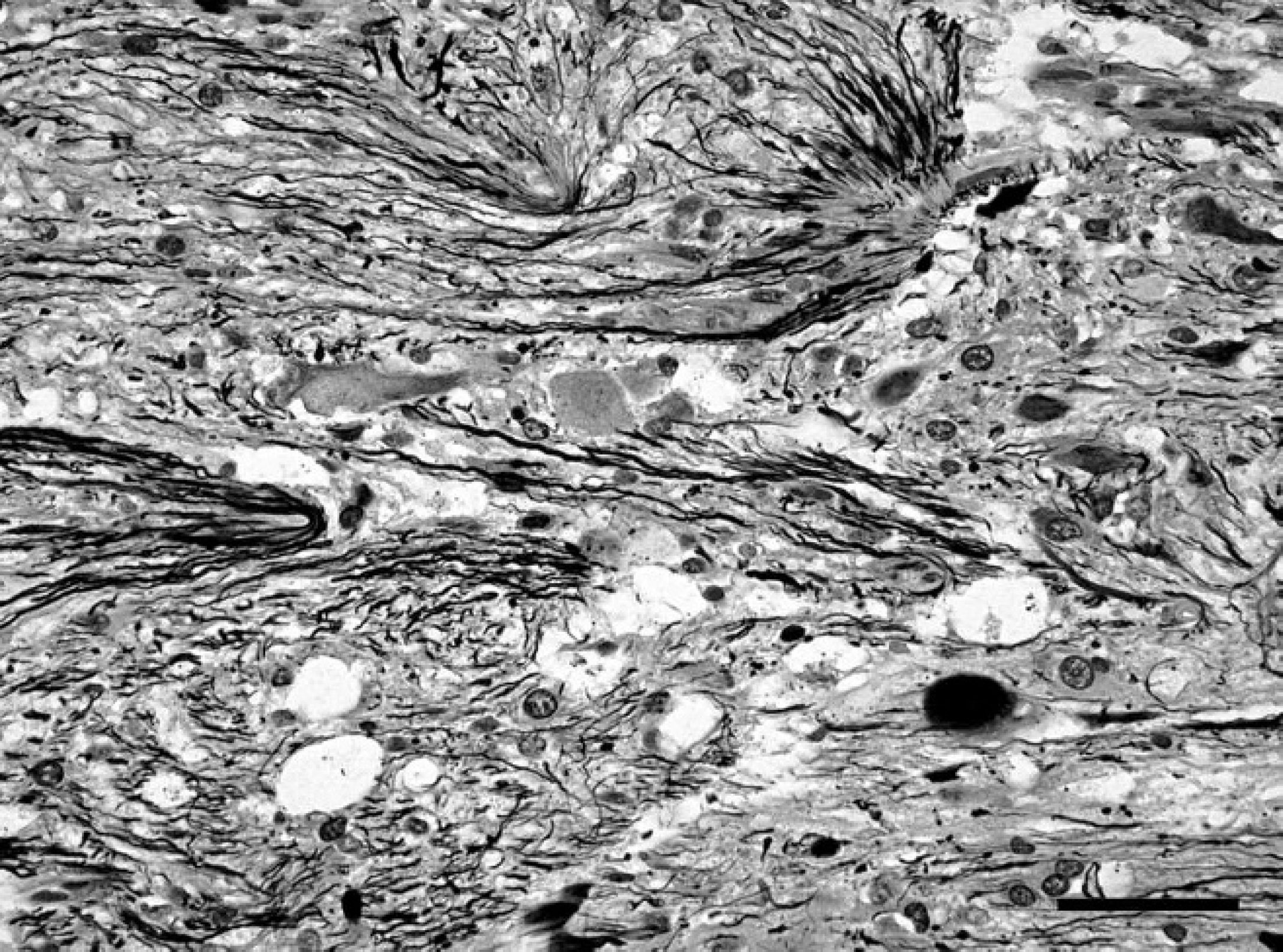
Spinal cord; western European hedgehog (Erinaceus europaeus). Bielschowsky silver stain shows intensely stained neuronal processes sometimes arranged in haphazard orientation or forming brush-like bundles especially in the nodular areas of the mass. Bielschowsky silver stain. Bar = 40 μm.
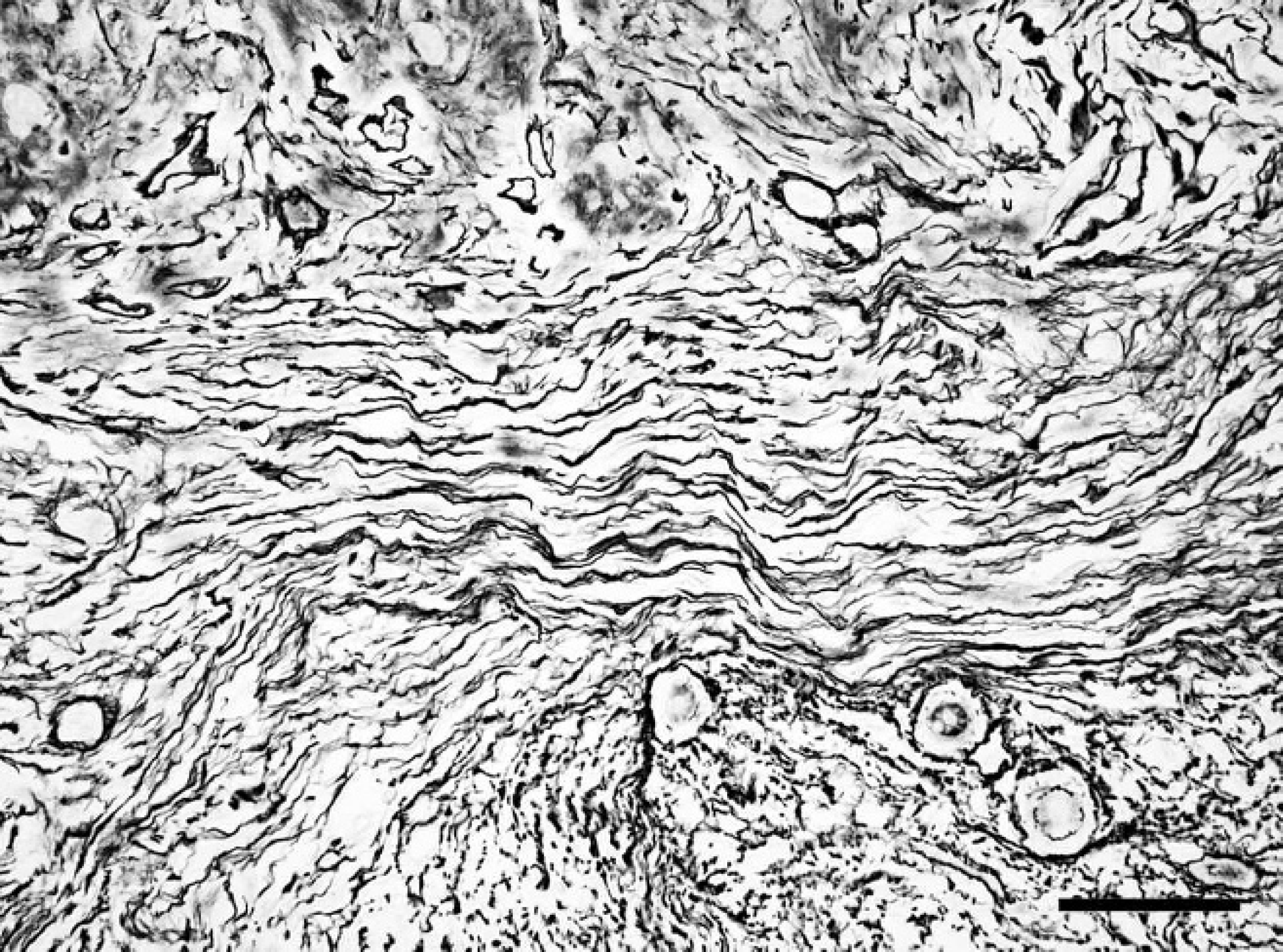
Spinal cord; western European hedgehog (Erinaceus europaeus). Gomori silver stain shows a prominent reticulin network surrounding almost every cell in the desmoplastic area of the mass. Gomori silver stain. Bar = 40 μm.
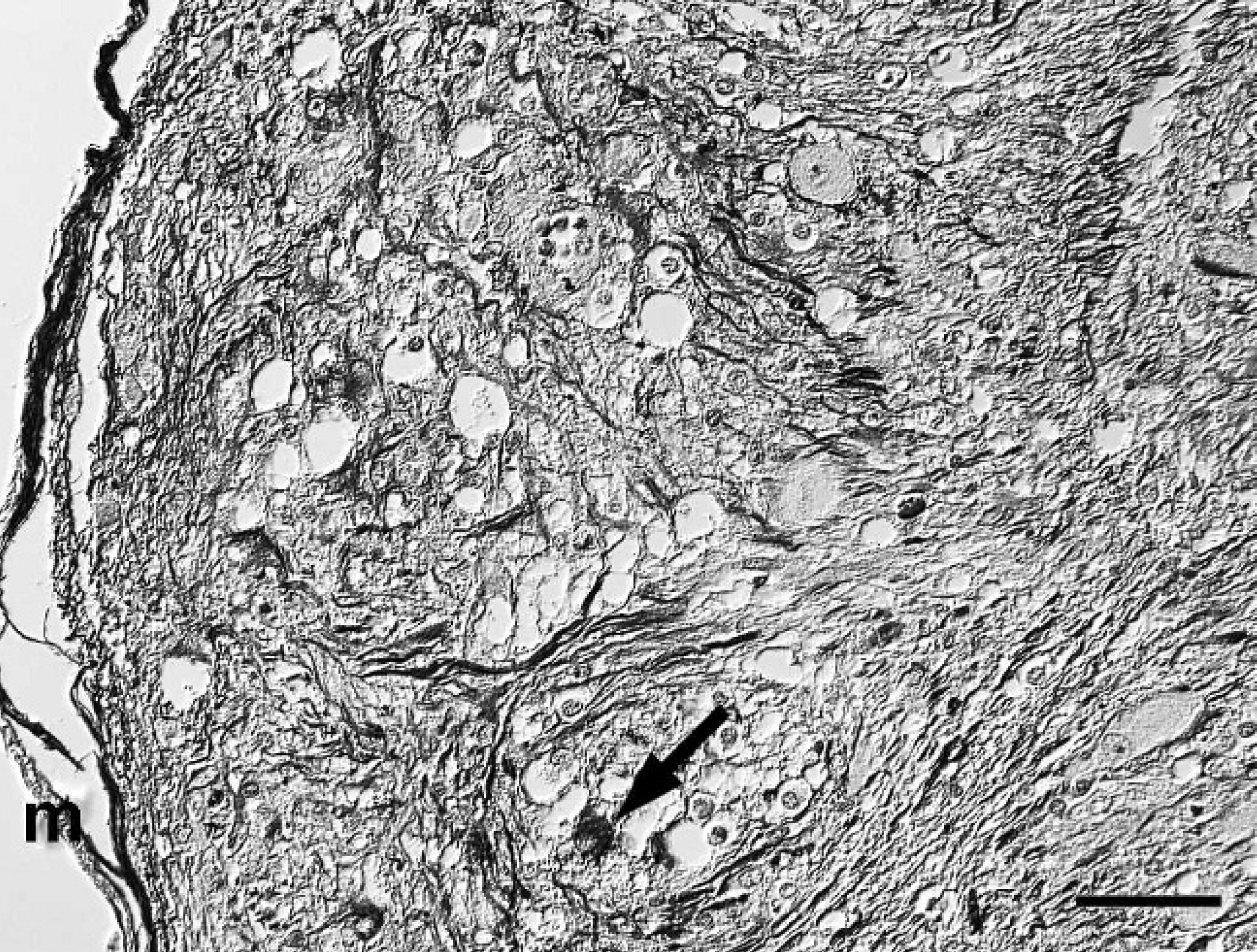
Spinal cord; western European hedgehog (Erinaceus europaeus). Numerous cells (arrow) and cell processes within nodular areas of the mass are strongly positive for glial fibrillary acidic protein (GFAP). m = meninx. Avidin-biotin-peroxidase complex method, hematoxylin counterstain, Nomarski differential interference contrast. Bar = 50 μm.
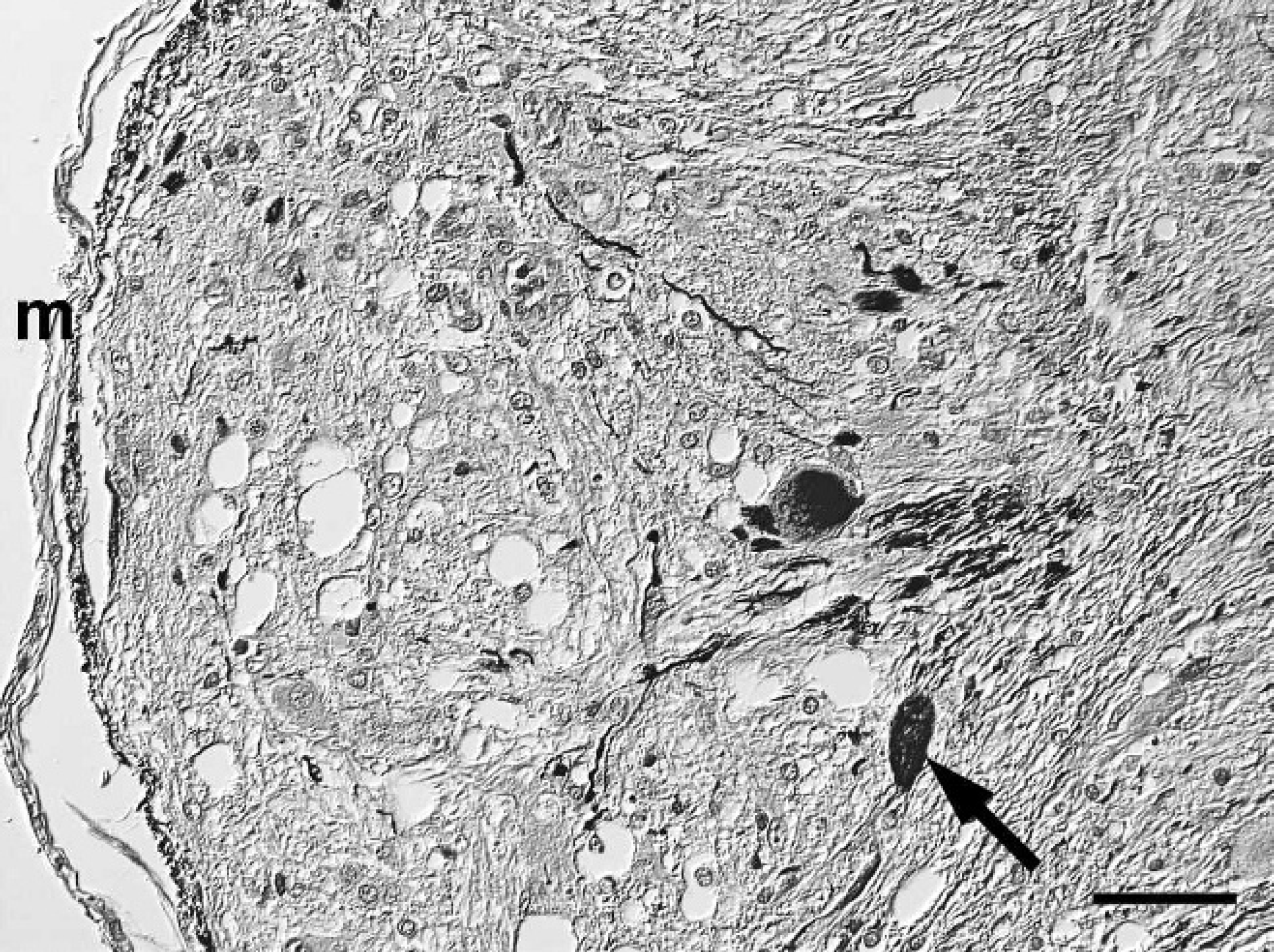
Spinal cord; western European hedgehog (Erinaceus europaeus). Polygonal, ganglionic, neuron-like cells (arrow) within a nodular area of the mass have mild to moderate neurofilament immunoreactivity. m = meninx. Avidin-biotin-peroxidase complex method, hematoxylin counterstain, Nomarski differential interference contrast. Bar = 50 μm.
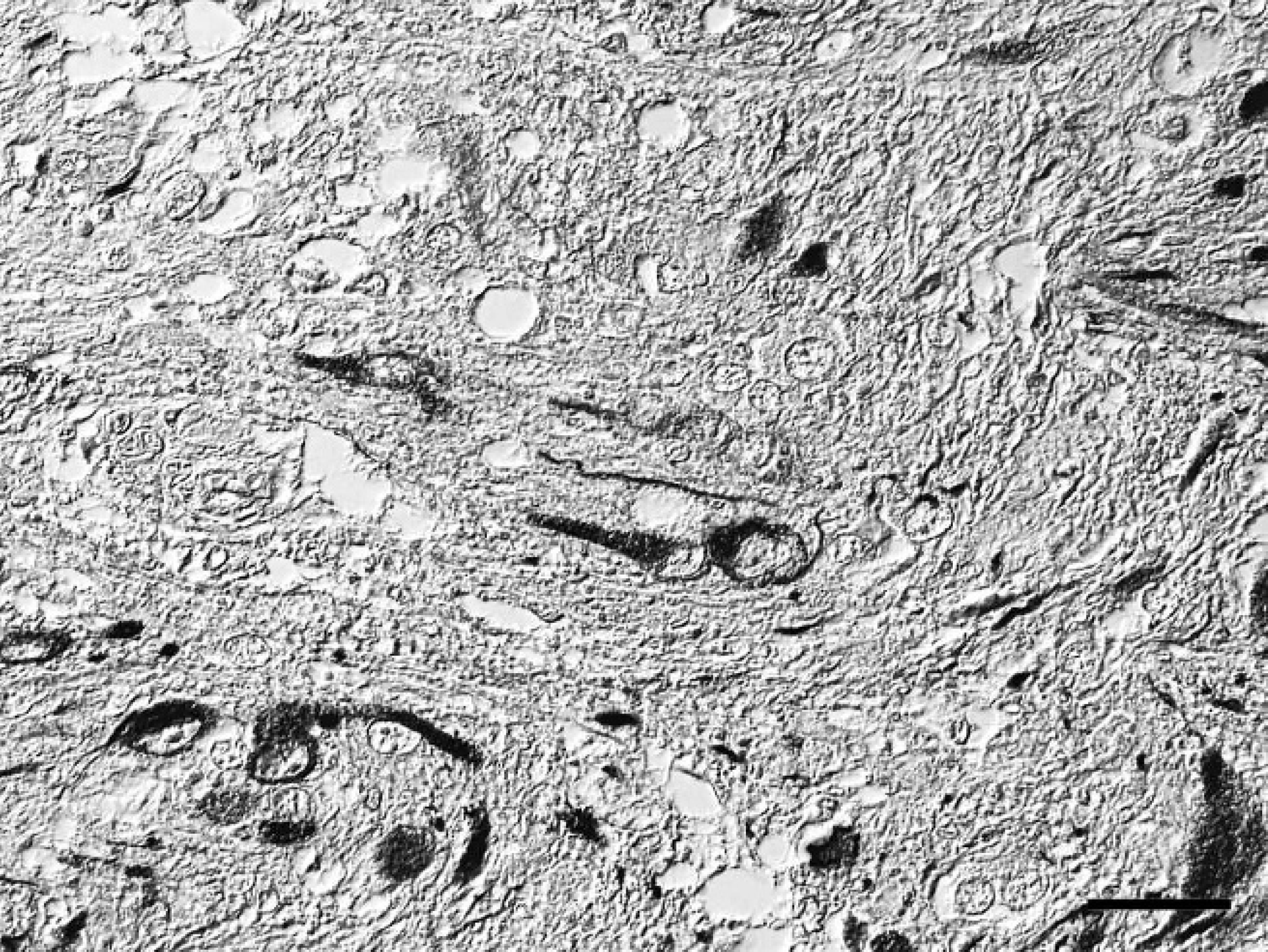
Spinal cord; western European hedgehog (Erinaceus europaeus). Disseminated polygonal, ganglionic, neuron-like cells within the nodular area of the mass have strong immunoreactivity for microtubule associated protein-2 (MAP2), indicating neuronal differentiation. Avidin-biotin-peroxidase complex method, hematoxylin counterstain, Nomarski differential interference contrast. Bar = 25 μm.
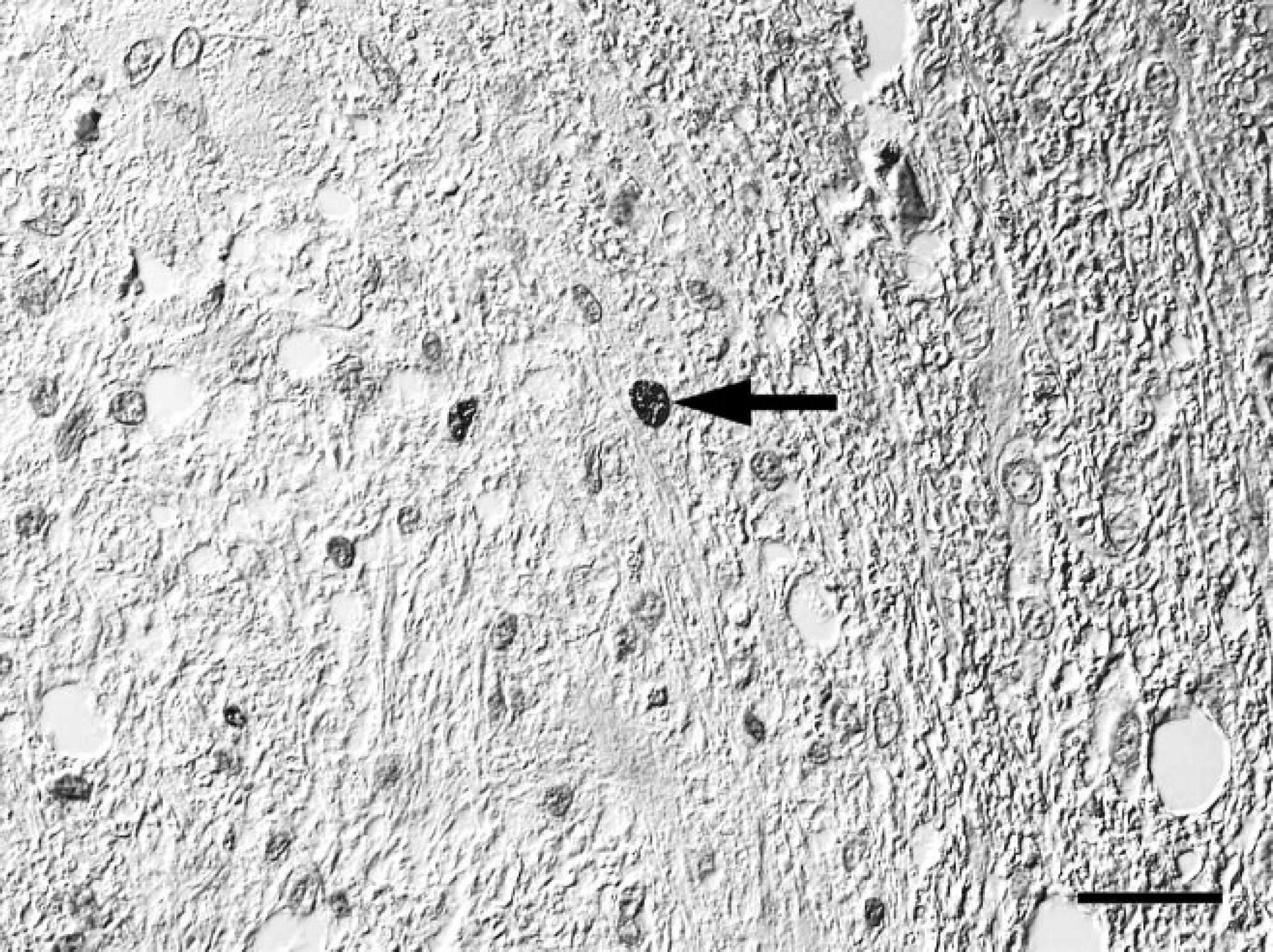
Spinal cord; western European hedgehog (Erinaceus europaeus). Few Ki-67 immunoreactive nuclei (arrow) within bipolar or oligopolar glial cells within a nodular area of the mass. Avidin-biotin-peroxidase complex method, hematoxylin counterstain, Nomarski differential interference contrast. Bar = 25 μm.
The present communication encompasses the clinical, pathomorphological, and immunophenotypical characterization of a desmoplastic ganglioglioma in the spinal cord of an adult male western European hedgehog. The detection of 3 subperiosteal incremental lines in the mandibular bone suggested that this hedgehog was at least 3 years of age. 12 Among the tumors of the central nervous system of domestic animals with neuronal differentiation (except the more frequent embryonal tumors), the current World Health Organization histologic classification system differentiates the primarily neuroblastic olfactory neuroblastoma (esthesioneuroblastoma) and the more differentiated gangliocytomas and gangliogliomas. 9 Gangliocytomas are an almost uniform proliferation of well-differentiated neuronal cells, whereas gangliogliomas include an additional population of neoplastic glial cells. 2,9 In human beings, gangliogliomas are mostly benign tumors, with the degree of glial differentiation determining their biological behavior. 2 Among the glial elements, both astrocytic and oligodendrocytic elements can be found mostly intermingled. 10 The present case included a GFAP-positive astrocytic component with occasional Rosenthal fibers, which are a characteristic feature of pilocytic astrocytomas. 21 Pilocytic astrocytomas are known to be less aggressive and have a better prognosis in humans, and although they are not included in the current World Health Organization histologic classification of tumors of the central nervous system of domestic animals, they have been recently described in dogs. 17 The demonstration of pleomorphic neurofilament-positive ganglionic neuron-like cells within parameningeal locations where normally no gray matter exists excluded the differential diagnosis of pre-existing neurons within an astrocytoma. In the present case, the immunohistological demonstration of GFAP-positive and S-100-positive glial cells as well as neurofilament-positive, microtubule-associated protein-2-positive, and neuron-specific enolase-positive neuronal cells within the tumor confirmed the diagnosis of ganglioglioma and is in accordance with previous reports from bovine and canine cases. 10,13,15,18 The generally weak vimentin immunoreactivity and the lack of NeuN immunoreactivity solely within the spinal cord point toward a less than optimal antigen preservation, which is suggested to be the result of the applied fixation and decalcification protocol. 1 A remarkable histologic feature of the present case was a prominent desmoplastic leptomeningeal component, which seemed to connect the multiple microcystic nodules with the meninges. A comparable desmoplastic component including an Azan-positive and Gomori silver stain-positive reticulin network was either not present or not assessed in detail in most previous veterinary case reports. 10,15,18 However, there is one case description from the medulla oblongata region in a 7-month-old heifer, which includes an area dominated by spindle cells arranged in a parallel orientation, 13 and 1 report from the eye of a juvenile dog, which contains randomly arranged or whorled cells embedded in dense stromal connective tissue, 16 possibly representing a comparable desmoplastic component. A similar desmoplastic leptomeningeal component with a prominent reticulin network, which may mimic a mesenchymal tumor, is described as a characteristic component of desmoplastic infantile gangliogliomas in humans. Despite the name, several noninfantile human cases of this tumor have been reported. 3
The origin of gangliogliomas remains enigmatic. Human gangliogliomas are believed to originate from neuroglial precursor cells. 2 An alternative origin from specialized subpial astrocytes was suggested for human desmoplastic infantile astrocytomas and gangliogliomas due to their abundant expression of basal lamina-associated proteins. 3 Both variants are in agreement with the finding of a limited number of Ki-67-positive nuclei within bipolar or oligopolar glial cells in the present case. The lack of Ki-67-positive nuclei within the ganglionic neuron-like cells does not exclude a neoplastic origin of these cells and has also been described for human cases of ganglioglioma. 6 It is suggested that a neuronal differentiation including the expression of neuron-specific antigens such as synaptophysin, microtubule-associated protein-2, neurofilament, and NeuN begins only after the cessation of mitotic activity. However, because of the well-differentiated histological appearance, a hamartomatous histogenesis cannot be excluded. 3,18 In the present case, the spinal cord neoplasia was considered to be the cause of the therapy-resistant ataxia, paresis, and urinary bladder dilatation. This adds a new entity to the list of clinical differential diagnoses for ataxia and paresis in hedgehogs, which is more frequently caused by trauma or a demyelinating disease known as wobbly hedgehog syndrome. 7,14 Similar to the present case, hind limb paresis and urinary bladder malfunction can be the leading symptoms of cases of intraspinal ganglioglioma in human beings. 8
Acknowledgements. The authors are grateful to Bettina Buck, Petra Grünig, and Annika Lehmbecker for excellent technical assistance.
Footnotes
a.
Vector Laboratories, Burlingame, CA.
b.
Dako Deutschland GmbH, Hamburg, Germany.
c.
Chemicon Europe, Hofheim/Taunus, Germany.
d.
Millipore Corp., Billerica, MA.
e.
Dianova, Hamburg, Germany.
f.
Sigma-Aldrich Chemie GmBH, Munich, Germany.
