Abstract
Gastrinoma, an infrequent diagnosis in middle-aged dogs, occurs with nonspecific gastrointestinal morbidity. Laboratory tests can yield a presumptive diagnosis, but definitive diagnosis depends on histopathology and immunohistochemistry. We describe a malignant pancreatic gastrinoma with lymph node metastases and corresponding Zollinger–Ellison syndrome in a Mexican gray wolf (Canis lupus baileyi) and review this endocrine neoplasm in domestic dogs. A 12-y-old, captive, male Mexican gray wolf developed inappetence and weight loss. Abdominal ultrasonography revealed a thickened duodenum and peritoneal effusion. Two duodenal perforations were noted on exploratory celiotomy and were repaired. Persisting clinical signs led to a second celiotomy that revealed a mesenteric mass, which was diagnosed histologically as a neuroendocrine carcinoma. During the following 16 mo, the wolf received a combination of H2-receptor antagonists, proton-pump inhibitors, gastroprotectants, and anti-emetics, but had recurrent episodes of anorexia, nausea, acid reflux, and remained underweight. Worsening clinical signs and weakness prompted euthanasia. The antemortem serum gastrin concentration of 414 ng/L (reference interval: 10–40 ng/L) corroborated hypergastrinemia. Autopsy revealed a mass expanding the right pancreatic limb; 3 parapancreatic mesenteric masses; duodenal ulcers; focal duodenal perforation with septic fibrinosuppurative peritonitis; chronic-active ulcerative esophagitis; and poor body condition. The pancreatic mass was diagnosed histologically as a neuroendocrine carcinoma and the parapancreatic masses as lymph node metastases. Immunohistochemistry of the pancreatic mass was positive for gastrin and negative for glucagon, insulin, pancreatic polypeptide, serotonin, somatostatin, and vasoactive intestinal peptide.
The Mexican gray wolf (Canis lupus baileyi) is a federally protected endangered carnivore listed under the Endangered Species Act since 1976, when the species was nearly extinct. 5 Between 1977 and 1980, the Mexican Wolf Species Survival Plan (MWSSP) established a breeding population, which in 2014 numbered 248 animals distributed among 55 facilities. 5 Because of captive-breeding and reintroduction, as per the U.S. Fish & Wildlife Service Ecological Services of the Southwest Region (https://www.fws.gov/southwest/es/mexicanwolf/reintroproj.html), by the end of 2016, there were 113 wolves distributed among 23 packs in the wild.
In December 2014, a captive-born 12-y-old intact male Mexican gray wolf, housed at a facility involved in the MWSSP, was presented to staff veterinarians because of inappetence and weight loss of 5.5 kg over the course of 15 mo. Abdominal radiographs were unrewarding. The animal was sedated for abdominal ultrasonography, which revealed a thickened duodenum and peritoneal effusion. An exploratory celiotomy allowed detection and repair of 2 duodenal perforations. Persisting clinical signs led to a second celiotomy that revealed a 4-cm mesenteric mass, which was diagnosed histologically as a suspected neuroendocrine carcinoma. Over the next 16 mo, the wolf was maintained on famotidine and omeprazole, and inconsistently received sucralfate, pantoprazole (Protonix, Pfizer, Philadelphia, PA), and maropitant (Cerenia, Zoetis, Kalamazoo, MI). During this time, the animal was intermittently anorexic and would chronically lick his lips causing caretakers to suspect nausea and acid reflux. Progressive anorexia, suspected chronic acid reflux, and lethargy prompted euthanasia.
At autopsy, the animal had moderate generalized muscle atrophy, scant-to-moderate adipose stores, and was dehydrated. There was multisegmental chronic-active ulcerative esophagitis. The abdominal cavity contained 200 mL of dark-brown fluid, and peritoneal surfaces were roughened with fibrin and reactive fibrovascular tissue consistent with peritonitis. Segmentally, the duodenum and the proximal jejunum were uniformly dilated up to 6 cm in circumference, and 13 cm distal to the pylorus, on the antimesenteric border, the duodenum was focally perforated. The serosal defect was stained with bile, and covered by fibrin, edema, and hemorrhage. The duodenal mucosa, extending for 25 cm, had multifocal, <0.5 cm diameter ulcers. The lumen contained bile-stained digesta and blood clots. Focally expanding the right pancreatic limb, 6 cm from the right and left pancreatic junction, was a 3.5 × 3 × 2 cm firm mass that was moderately well demarcated and mottled white-to-gray to dark-red on cross-section (Fig. 1). Within the mesentery adjacent to the pancreatic limbs, there were 3 firm masses, which measured 2.5 cm3, 7.5 × 3.5 × 2.5 cm, and 4.5 × 3 × 2.5 cm (Fig. 1). On cross-section, all masses were similar to the pancreatic mass.
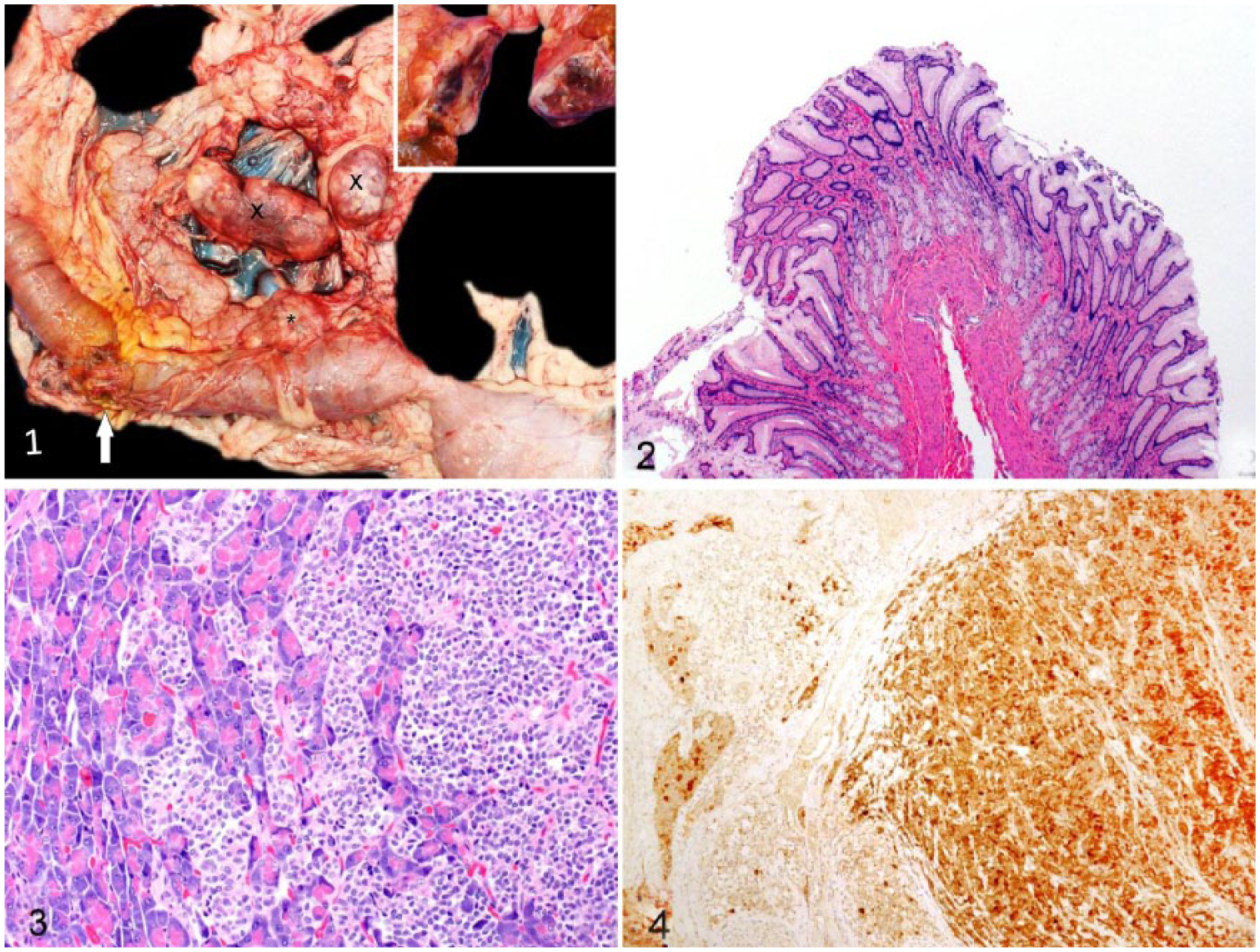
Pancreatic gastrinoma and Zollinger–Ellison syndrome in a Mexican gray wolf (Canis lupus baileyi).
Additional findings included moderate leptomeningeal fibrosis, splenic contraction, focal adrenal gland cortical nodular hyperplasia, hepatic nodular hyperplasia with glycogen-type vacuolation, and gastric mucosal hyperplasia (Fig. 2). The pancreatic mass and the mesenteric masses were histologically similar. Adjacent to the opening of the common bile duct was a highly cellular and infiltrative multilobular neuroendocrine neoplasm dissected by thick fibrous connective tissue (Fig. 3). The mass disrupted and replaced pancreatic acini, parapancreatic adipose tissue, and the duodenal serosa and muscularis externa. Neoplastic cells were arranged in packets and rosettes separated by thin fibrovascular tissue. The cells had poorly defined borders, eosinophilic loosely granular cytoplasm, and round-to-ovoid to notched nuclei with coarse chromatin. The mitotic count was 3 per ten 400× high-power fields. There was infrequent karyomegaly. In the adjacent stroma, there was chronic hemorrhage. The mesenteric masses contained similar neoplastic cells with necrosis, and the masses replaced and compressed residual lymphoid tissue, indicating that these masses were lymph node metastases. Rare clusters of similar neoplastic cells occupied hepatic sinusoids.
Immunohistochemistry for gastrin, glucagon, insulin, pancreatic polypeptide, serotonin, somatostatin, and vasoactive intestinal peptide was performed on the pancreatic mass as described previously.4,17 The mass was negative for all hormones except for strong multifocal cytoplasmic gastrin immunoreactivity (Fig. 4), which confirmed the neoplastic cells to be gastrin-producing. 13
Gastrin concentration of 414 ng/L was determined in the wolf’s serum collected prior to euthanasia (125I radioimmunoassay; Endocrine Diagnostic Laboratory [EDL], Michigan State University, East Lansing, MI). 16 Although a serum gastrin reference interval (RI) is lacking for the Mexican gray wolf, we presume that the levels are similar to those reported in dogs. In dogs, the baseline serum gastrin concentration varies among reports, likely because of different assays, and is described as a range of 10–40 ng/L, 16 66–76 pg/mL, 18 23–104 pg/mL, 18 20–100 pg/mL, 18 27–85 pg/mL, 6 45–98 pg/mL, 6 or as a mean of 40 pg/mL 19 or 71 pg/mL. 3 In the case of our wolf, the gastrin level was ~10 times the higher end of the canine RI (10–40 ng/L) used by the EDL. In dogs with gastrinoma, serum gastrin concentrations are typically >3 times the upper end of the RI, but more specific values are lacking given the rarity of reported cases. 21 The therapeutic use of H2-receptor antagonists and proton-pump inhibitors can falsely increase serum gastrin, but studies in healthy dogs have concluded that gastrin concentrations rarely exceed 3 times the upper end of the RI (10–40 ng/L in that study); therefore, therapy-related fluctuations of serum gastrin are unlikely to be confounded with a functional gastrinoma. 16
Autopsy, histopathology, immunohistochemistry, and serum gastrin levels in the wolf in our study confirmed a diagnosis of malignant pancreatic gastrinoma with metastasis to regional lymph nodes, rare tumor microemboli in hepatic sinusoids, and secondary duodenal peptic ulcers as a result of Zollinger–Ellison syndrome. One ulcer was transmural and had resulted in septic suppurative peritonitis. The clinical suspicion of chronic acid reflux was corroborated by the presence of chronic-active ulcerative esophagitis.
Gastrinoma is a rarely reported neuroendocrine tumor that is responsible for Zollinger–Ellison syndrome, which is characterized by gastric acid hypersecretion, upper gastrointestinal ulceration, and non-beta islet cell tumors. 18 The syndrome was first described in a dog in 1976, 18 and has been reported mostly in domestic dogs, 21 a few cats, 21 and now in a Mexican gray wolf. Affected dogs are middle-aged (range: 3.5–12 y; mean: 8.2 y) without a breed predisposition, and females may be overrepresented. 21 The tumor typically involves the pancreatic islets, 14 but has also been reported in the duodenal wall at the level of the major duodenal papilla, 20 in the liver, 13 in the mesenteric root, 2 and in parapancreatic lymph nodes, 10 sometimes without a detected pancreatic mass, although autopsies were not always performed. The predilection of this tumor for the pancreas is interesting given that the adult canine pancreas lacks gastrin-producing cells. 13 However, gastrin-producing cells are present in the pancreas of the developing fetus and neonate, and in the adult duodenum and gastric antrum. 13 Theories to explain how gastrin-producing neoplasms develop in pancreatic islets are multiple and include: the translocation of gastrin-producing cells from the duodenum or gastric antrum to the pancreatic islets during fetal development; the differentiation of pluripotent cells, or nesidioblasts, within the pancreatic islets to gastrin-producing cells or G cells; and de-differentiation from an existing islet pancreatic endocrine cell into a gastrin-producing cell. 13 Gastrinoma tumor cells are reported to have cytoplasmic granules characteristic of those in delta cells. 1
Patients with functional gastrinomas develop hypergastrinemia that results in nonspecific signs of gastrointestinal disease. The pathophysiology involved is a perturbed gastric pH regulatory feedback loop. Normally, gastrin-producing cells or G cells of the distal part of the stomach produce controlled amounts of gastrin that are essential for gastrointestinal system function. 12 The anticipation of eating or gastric distension or an ingested meal (e.g., protein or high stomach pH) leads to gastrin release into the blood stream, which stimulates parietal cells to increase gastric acid production, thus causing a decrease of gastric pH.12,18 To avoid gastric hyperacidity, G cells closely monitor the pH of the distal part of the stomach, and once the pH reaches a critical point (pH of 1–2), 12 the release of gastrin is inhibited. Consequently, the stimulus to produce gastric acid is removed, and the stomach pH stabilizes. 12 This gastrin-stomach pH regulatory feedback loop is a major mechanism that protects against acid-induced mucosal damage. In instances of abnormal and uncontrolled hypergastrinemia, such as occurs with a functional gastrinoma, the regulatory feedback loop is perturbed and rendered ineffective at stabilizing stomach pH. Therefore, highly acidic stomach contents may empty into the duodenum or may be expelled through the esophagus. Duodenal acidification likely activates a second protective mechanism, the intestinal negative feedback loop, which relies on neuronal reflexes and secretin to suppress gastric acid production and delay gastric emptying. 12 The sequelae of hypergastrinemia are gastric acid hypersecretion and gastric mucosal hyperplasia.18,21
Common clinical signs are vomiting, weight loss, depression, lethargy, anorexia, and diarrhea; 21 in those patients with gastrointestinal ulceration, which is the majority, 21 there may be hematemesis, melena, hematochezia, abdominal pain, anemia, fever, leukocytosis, elevated urea, hypoalbuminemia, polydipsia, and electrolyte abnormalities.8–10,15,18 Other patients, including the wolf in our study, may develop ulcerative esophagitis related to acid reflux favored by a delay in gastric emptying and chronic regurgitation.8,18,19,21 Less often, patients have steatorrhea 18 or extraluminal bile duct obstruction. 20 Because of the frequency of gastrointestinal ulceration, perforation and peritonitis are common. 21
Diagnosing gastrinoma in a patient is complicated by nonspecific clinical findings, the rarity of the tumor, the small size of the tumor, and the variable usefulness of imaging and endoscopy. The clinical signs, physical exam, and the minimum database (complete blood count, biochemistry) may suggest gastrointestinal disease, but even so, the differential diagnosis remains broad. 9 Abdominal radiographs are usually unremarkable unless there is perforation.10,15 Contrast-enhanced radiographs may show gastric wall thickening, complete pyloric obstruction, gastroduodenal ulceration, or increased small bowel transit time. 21 Abdominal ultrasonography may identify a pancreatic mass or metastases, 1 but more often it is unrewarding given the usual small tumor size.9,10,15 More advanced imaging modalities, such as computed tomography, will increase sensitivity and may visualize neoplastic masses that will require further investigation. 21
Endoscopy, with endoscopic biopsies, is commonly used and can detect pyloric antrum hypertrophy, pyloric stenosis, gastrointestinal ulceration, hemorrhage, and inflammation. This approach is unlikely to diagnose gastrinoma and may result in an erroneous diagnosis of inflammatory bowel disease.6,10,11,15 In at least 3 cases, endoscopic biopsies revealed ulcerative inflammation followed by an exploratory celiotomy that detected a pancreatic gastrinoma, 8 a pancreatic gastrinoma with hepatic metastasis, 19 or a metastatic gastrinoma. 10
Although celiotomy is an invasive procedure, exploratory celiotomy for biopsy collection and immunohistochemistry may be the best diagnostic approach. This approach was used successfully in at least 9 cases.1,7,8,10,13,20 Other useful laboratory tests include a measure of basal gastric acid secretion, a measure of serum gastrin concentration, and provocative testing of gastrin secretion with intravenous secretin or calcium gluconate.9,21 Provocative testing is particularly useful in cases of suspected gastrinoma in which serum gastrin levels are normal-to-borderline.10,15
Although such laboratory tests may aid in diagnosis, in addition to documenting hypergastrinemia, a definitive diagnosis of gastrinoma is based on histologic evidence of a pancreatic islet cell tumor with neoplastic cells immunoreactive for gastrin. 21 Hypergastrinemia alone is insufficient to diagnose a gastrinoma, because elevated basal serum gastrin may accompany renal failure, gastric outflow obstruction, hypochlorhydria, atrophic gastritis, small intestinal resection, gastric dilation and volvulus, chronic gastritis, liver disease, immunoproliferative enteropathy of Basenji dogs, and certain therapeutic drugs.15,21 Although immunohistochemistry or radioimmunoassay of tumor tissue extracts can confirm the presence of gastrin in the tumors, it fails to show that the hormone is being secreted in excess into the circulation. 21 Compatible clinical signs and lesions can correlate with a functional gastrinoma, but documenting concomitant hypergastrinemia is more convincing.
Treatment of canine gastrinoma is difficult and palliative, and clinicians resort to a combination of surgical intervention and long-term medical therapy. 15 Early surgical intervention has the advantage of making a final diagnosis and beginning treatment, but because of the likelihood that surgical resection will be incomplete, medical management is important and depends on drugs that reduce gastric acid secretion and treat gastrointestinal ulceration, while correcting fluid and electrolyte losses. 21 Proton-pump inhibitors such as omeprazole, alone or in combination, appear to be a good choice for medical management.2,14
Following a diagnosis of gastrinoma, canine patients have a guarded-to-poor prognosis, in part because metastasis to the liver and regional lymph nodes has already occurred in 85% of cases.10,18,21 Canine patients receiving combined treatment are reported to survive up to 18 mo, 21 although 1 patient treated indefinitely with omeprazole was asymptomatic and survived for at least 24 mo, 2 and another receiving ranitidine, omeprazole, and sucralfate survived for 26 mo. 14 Patients were often euthanized upon diagnosis or following the recurrence of treated disease, some died before a diagnosis was made, 11 and others died from complications of the disease, including septic peritonitis from intestinal perforation.6,18
The disease progression of the wolf in our study mirrored what is described in domestic dogs diagnosed with gastrinoma. The wolf had nonspecific clinical signs, and initial bloodwork and imaging suggested gastrointestinal disease. Two exploratory celiotomies followed, which revealed duodenal ulcers and a mesenteric mass suspicious of a neuroendocrine carcinoma. The animal was medically managed until it was euthanized because of declining health. Autopsy and ancillary testing resulted in a definitive diagnosis of malignant gastrinoma, which had caused longstanding morbidity that included multiple duodenal ulcers, 2 instances of duodenal perforation, gastric mucosal hyperplasia, and reflux esophagitis. It is believed that the primary gastrinoma was the focal mass in the right pancreatic limb adjacent to the common bile duct. This location appears to be a frequent site in dogs, 21 and its proximity to the opening of the bile duct favors one of the elaborated theories of the origin of pancreatic gastrinomas within the “gastrinoma triangle”. 13 This area is delimited by the common and cystic bile ducts superiorly, the junction of the body and the tail of the pancreas medially, and the junction of the second and third segments of the duodenum inferiorly. 13 In humans, the development of gastrin-producing tumors in this area is thought to originate, in part, from ectopic gastrin-producing cells. 13
Footnotes
Acknowledgements
We thank Southwest Wildlife Conservation Center for their role in the MWSSP and their wildlife conservation efforts. We thank Dr. Miranda Frohlich, Dr. Russell T. Greene, and Dr. Adam Kincaid for their assistance with this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support from the Midwestern University College of Veterinary Medicine for the immunohistochemistry studies performed. The authors received no other specific financial support for the research, authorship, and/or publication of this article.
