Abstract
A 4-year-old female alpaca was referred to The Queen's Veterinary School Hospital (Cambridge, United Kingdom) with a history of abortion 6 days previous, at approximately 245 days in gestation. The patient had developed watery diarrhea 24 hr after abortion and exhibited bruxism, progressive depression, and ultimately collapse. On presentation to the referral institute, the alpaca was unable to stand and exhibited severe tachypnea, dyspnea, and tachycardia. The patient was considered to be moribund and was euthanized. Macroscopic postmortem assessment and subsequent histological investigations revealed multifocal ulceration of the third gastric compartment, with vascular mycotic invasion and disseminated multifocal embolic mycotic pneumonia. Real-time polymerase chain reaction targeting the large ribosomal subunit (28S) performed on paraffin-embedded lung tissue successfully amplified 28S RNA specific for Aspergillus species. The primary cause of the depression and collapse of this animal was considered to be a combination of respiratory compromise due to severe embolic mycotic pneumonia and endotoxic shock associated with the ulcerative gastritis of the third gastric compartment. To the authors' knowledge, this is the first report of ulcerative gastritis of the third gastric compartment histologically illustrated as a portal of entry for Aspergillus species causing embolic mycotic pneumonia in an alpaca.
Mycotic pneumonia has been previously reported in the alpaca 4,5 ; however, the route of entry of the fungus in this species remains histologically unconfirmed. The disease has been more fully characterized in a single llama 6 and in horses, in which there is a well-established link between gastrointestinal disease, mycotic invasion, and embolic mycotic disease in a variety of organs including the lungs. 8 The current report describes a case of mycotic pneumonia in an alpaca, in which fungal-colonized ulcerative lesions in the third gastric compartment clearly indicate a gastrointestinal portal of entry for the fungus.
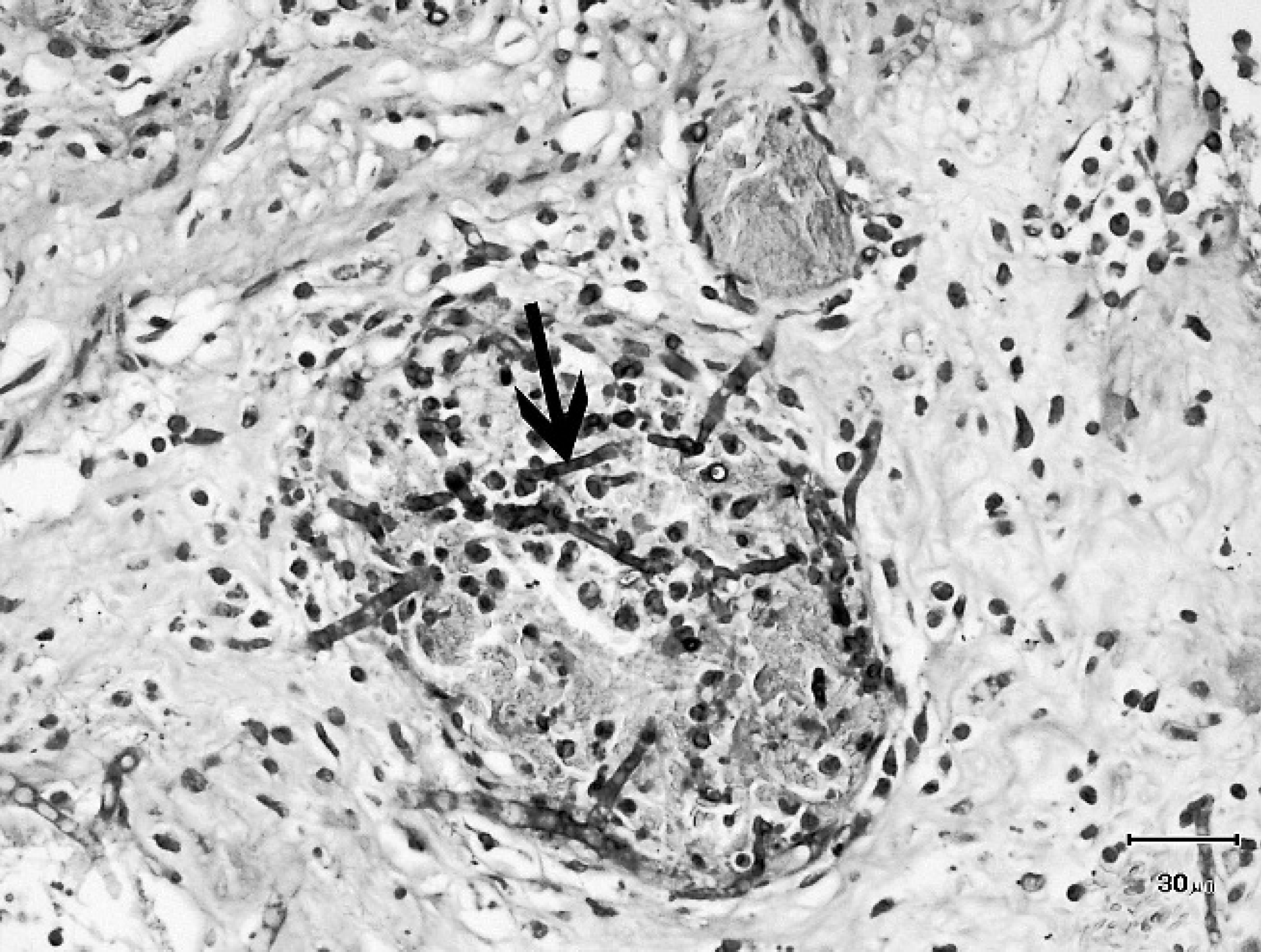
Submucosa third gastric compartment; thrombosed blood vessel with invasive fungal hyphae (arrow). Gomori methenamine-silver counterstained with hematoxylin and eosin stain. Bar = 30 μm.
A 4-year-old female alpaca was referred to The Queen's Veterinary School Hospital (Cambridge, United Kingdom) with a history of abortion 6 days previous, at approximately 245 days in gestation. Investigations by the referring veterinary surgeon included fetal fluid serology for Neospora caninum, Toxoplasma gondii, and Bovine viral diarrhea virus, all of which were negative. Fetal fluids were also negative for nitrates. Bacterial culture of placenta and fetal stomach contents yielded no significant growth.
The referring veterinary surgeon had treated the alpaca prophylactically with procaine penicillin and dihydrostreptomycin sulfate prior to referral, but the patient developed watery diarrhea 24 hr after abortion and exhibited bruxism, progressive depression, and ultimately collapse while receiving symptomatic treatment with enrofloxacin a and butylscopolamine bromide and metamizole. b
On arrival at The Queen's Veterinary School Hospital, the alpaca was unable to stand and exhibited severe tachypnea, dyspnea, and tachycardia. The patient was considered to be moribund, and per agreement with the owner, the alpaca was euthanized with pentobarbitone sodium c without further clinical investigations.
At necropsy, the animal was found to be in a poor body condition and was scored 1 out of 5 on a scale developed in ruminants, in which 1 = emaciated and 5 = obese. 2 Four liters of straw-colored peritoneal fluid were present in the abdomen, and the peritoneal surfaces appeared roughened, with fibrin tags on the serosal surface of the third gastric compartment. The mucosal surface along the greater curvature and adjacent to the pylorus exhibited multifocal red, depressed, spherical ulcers ∼15 mm in diameter.
Well-circumscribed, multifocal, white, firm foci measuring ∼4 mm in diameter were present throughout the lung, involving both the pleural surfaces and the parenchyma. Petechial hemorrhages were evident on the tracheal mucosa, musculature surrounding the trachea, pleural surfaces, splenic capsule, and uterine mucosa. All other organ systems, including the uterus, were examined and were found to be macroscopically unremarkable.
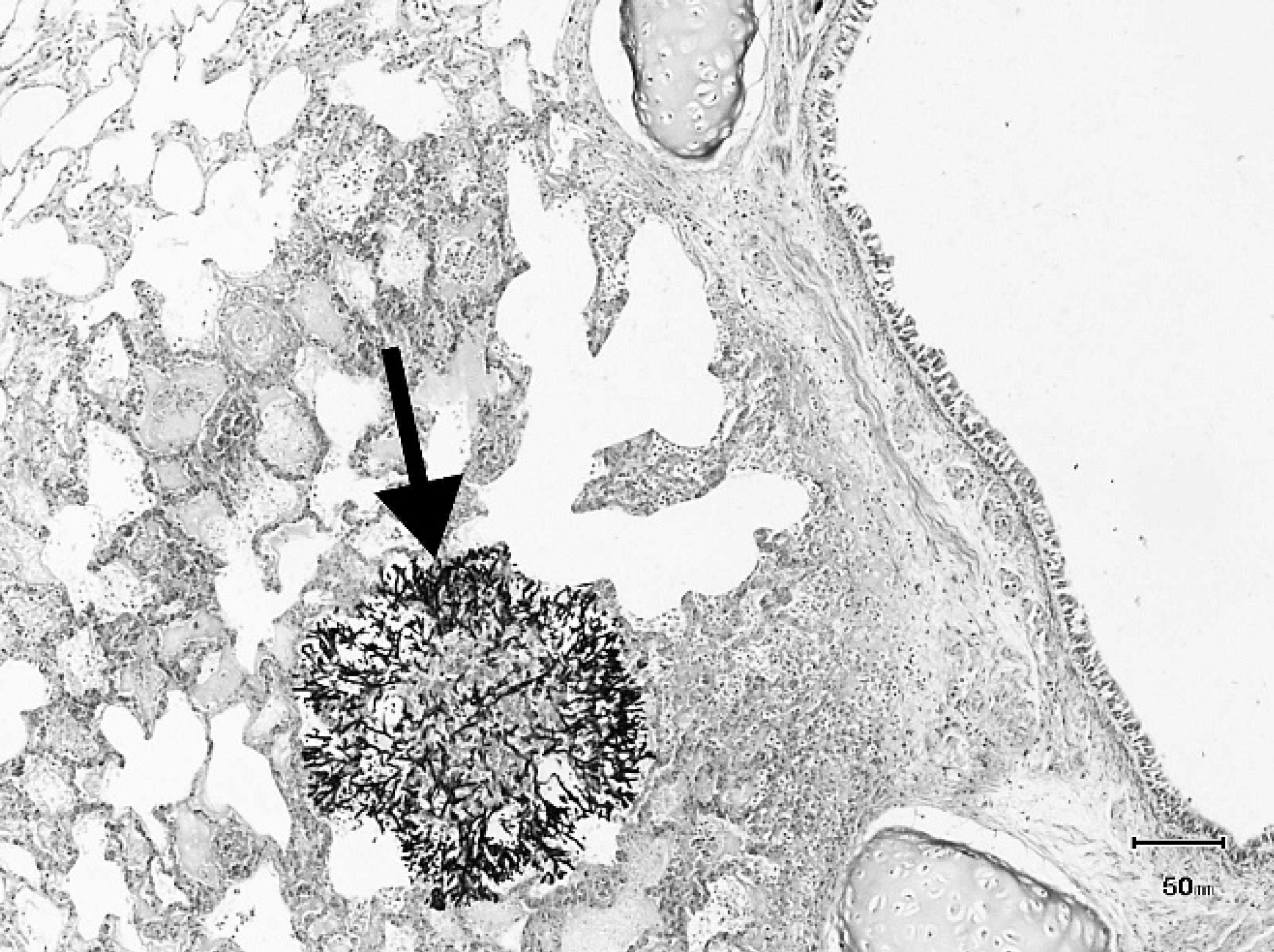
Lung; large aggregate of fungal hyphae (arrow) adjacent to a bronchus. Gomori methenamine-silver counter-stained with hematoxylin and eosin stain. Bar = 50 mm.
For histologic investigation, tissue samples from all major organs were fixed in 10% neutral buffered formalin solution and then submitted for routine processing and paraffin embedding. Three-micrometer sections were mounted on glass slides and stained with hematoxylin and eosin.
Histological examination of the third gastric compartment revealed multifocal ulceration of the mucosal epithelium, with accumulation of necrotic debris and a diffuse infiltrate of moderate numbers of lymphocytes, plasma cells, and viable and degenerate neutrophils throughout the remaining mucosa, submucosa, and muscularis. Many fungal hyphae were prominent within the exposed submucosa in areas of ulceration. Hyphae were septate, exhibited acute dichotomous branching, had parallel walls, and measured ∼5 μm min diameter. Multiple submucosal vessels were thrombosed and contained fungal hyphae within the vessel lumen and in close association with the vascular wall (Fig. 1).
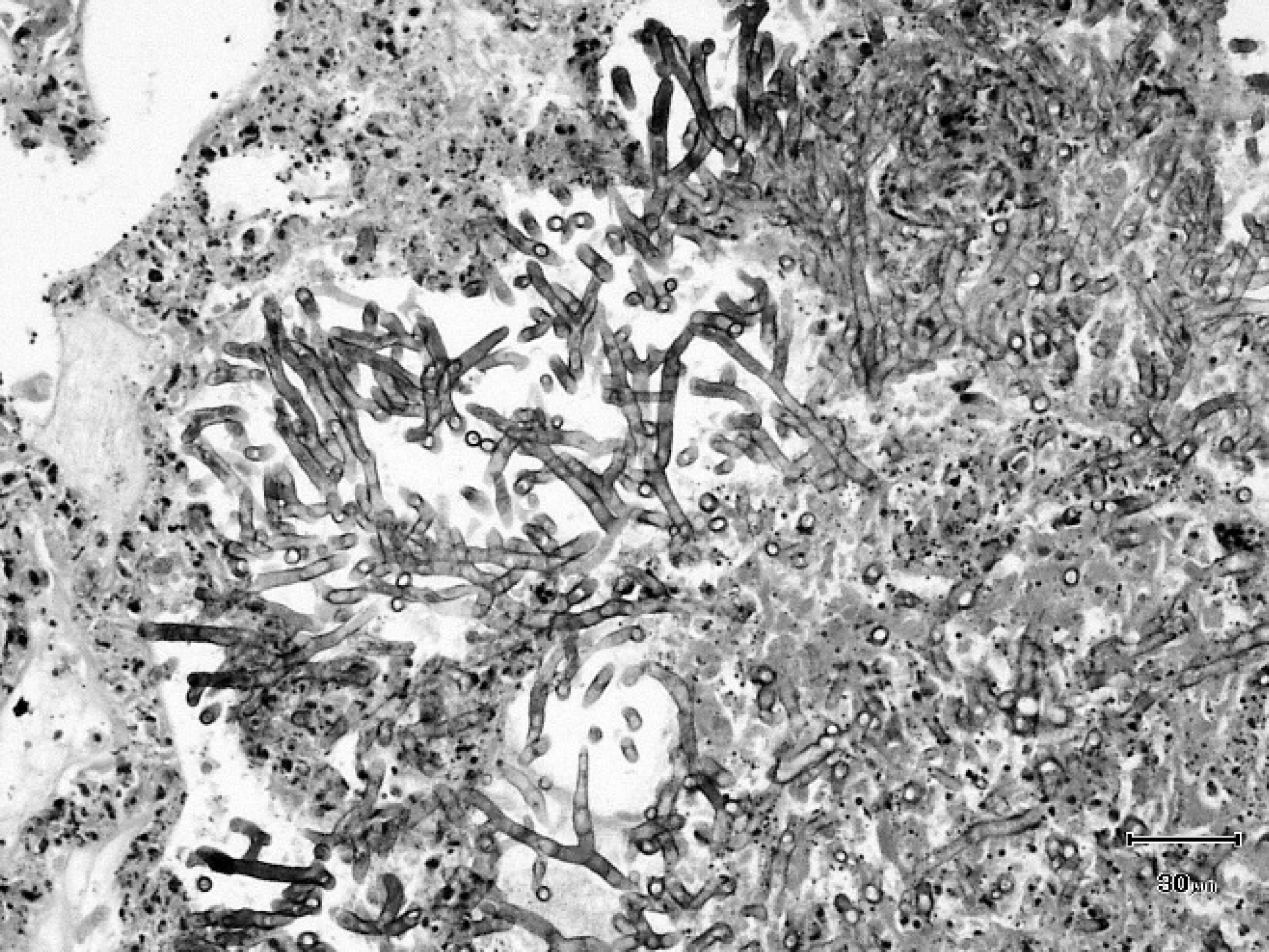
Lung; fungal hyphae consistent with Aspergillus spp. Note that the hyphae are septate and exhibit acute, dichotomous branching. Hematoxylin and eosin stain. Bar = 30 μm.
In the lungs, a multifocal, marked, embolic pneumonia with intralesional fungi was noted (Fig. 2) and was characterized by the presence of large numbers of viable and degenerate neutrophils with dense accumulations of detritus. Radiating from these foci were large numbers of fungal hyphae morphologically identical to those seen in the wall ofthe third gastric compartment (Fig. 3). The lumen of several large pulmonary arteries were thrombosed and contained fungal hyphae enmeshed within amorphous eosinophilic material and karyorrhectic debris.
Fungal hyphae with identical morphology were seen throughout the liver, where they were present in association with areas of hemorrhage, individual hepatocellular necrosis, and moderate infiltrates of macrophages admixed with fewer lymphocytes and plasma cells. Many hyphae were present immediately beneath the hepatic capsule.
Paraffin-embedded lung tissue was sent to the Mycology Reference Laboratory of the Southwest Regional Laboratory (United Kingdom) for detection of the genus-specific Aspergillus large ribosomal subunit. This laboratory employs real-time polymerase chain reaction (real-time PCR) using Aspergillus-specific hybridization probes and nested primers targeting the conserved large ribosomal subunit (28S) gene sequences of Aspergillus spp. 9,10 This technique successfully amplified a fragment corresponding to the conserved large ribosomal subunit gene of Aspergillus spp.
Neither anaerobic nor aerobic bacteria were grown from cultures of the abdominal effusion, ileum, and mesenteric lymph nodes. Bacterial culture of lung samples yielded a moderate growth of mixed nonhemolytic coliforms and a sparse growth of nonhemolytic staphylococci; neither was considered significant. The negative bacterial culture results were unsurprising given the antibiotic therapy administered prior to referral. Bovine herpesvirus 1 (commonly referred to as Infectious bovine rhinotracheitis virus) antigen was not detected in lung using fluorescent antibody tests. Bovine viral diarrhea virus–specific RNA was not detected in spleen using reverse transcription PCR.
The primary cause of the depression and collapse of this animal was considered to be a combination of respiratory compromise due to severe embolic mycotic pneumonia and endotoxic shock associated with the ulcerative third-compartment gastritis. The etiological agent had morphology consistent with Aspergillus species, and this was corroborated by the results of the nested PCR. Pneumonic aspergillosis has previously been documented in alpacas 4,5 and a llama. 6
The presence of numerous fungal hyphae in the ulcerated portion of the third gastric compartment and in thrombosed hepatic and pulmonary vessels is strongly consistent with hematogenous spread of the fungus from the third gastric compartment. The pathogenesis of embolic mycotic pneumonia is well-recognized in horses, in which gastrointestinal mucosal compromise has been associated with pulmonary aspergillosis 8 and has been shown in a llama. 6 However, although necrosis of the stomach wall was seen in 1 case of an alpaca with disseminated aspergillosis, 5 to the authors' knowledge, no cases with histological evidence of fungal hyphae at the site of gastric ulceration have been reported. The present case therefore records the first histological evidence of embolic mycotic pneumonia in an alpaca with fungal invasion at the site of ulceration of the third gastric compartment and additional portal vascular invasion.
Third-compartment ulceration has been reported with a relatively high incidence in llamas and has been circumstantially linked to stress. 7 In the current study, it can be speculated that the pregnancy, or subsequent abortion 6 days prior to the death of this patient, may have led to stress, which may have predisposed the animal to ulceration or exacerbated existing ulcerative lesions. There was no history of corticosteroid or nonsteroidal anti-inflammatory drug administration in the current case. Unfortunately, the referring veterinary surgeon did not perform fungal culture on the aborted fetus, and the cause of the abortion was undetermined in spite of the diagnostic tests utilized.
It is notable that the alpaca in the current study did not exhibit signs of respiratory disease until it presented in a moribund state at the referral veterinary center. It has been suggested that in horses with disease causing gastrointestinal mucosal compromise and exhibiting clinical signs of respiratory disease, embolic mycotic pneumonia should be considered as a possible differential diagnosis. 8 The present case is notably different, as this alpaca did not present to the referring veterinary surgeon with respiratory signs. Similarly, in a previous equine case, 3 a horse developed rapidly debilitating, acute, pulmonary aspergillosis without exhibiting clinical signs characteristic of pulmonary infection.
The lack of fungal culture for definitive speciation of the fungus is a limitation of the present report. Fungal culture was not performed, as a systemic mycosis was not suspected at the time of necropsy. However, the use of real-time PCR targeting the large ribosomal subunit (28S) of Aspergillus species and the morphological appearance of the fungal hyphae provide confirmation of the histological diagnosis. Fungal culture remains the gold standard for identification and speciation of fungal infections. 1
The current report documents a case of pulmonary aspergillosis in an alpaca in which there is definitive histological evidence of fungal invasion through ulcers within the third gastric compartment, with real-time PCR confirmation of Aspergillus species as the implicated fungal agent. As is the case with horses, inalpacas embolic mycotic pneumonia should be considered as a possible differential diagnosis in disease states involving mucosal compromise of the gastrointestinal tract and clinical signs of respiratory disease. In addition, this case illustrates that alpacas with mycotic pneumonia may present without attendant clinical signs of respiratory disease until they reach an advanced stage of infection.
Acknowledgements. The authors would like to thank Dr. Elizabeth M. Johnson and Dr. Christopher J. Linton, both of the Mycology Reference Laboratory, Bristol (United Kingdom), for performing the Aspergillus real-time polymerase chain reaction; and Dr. Timothy J. Scase of The University of Cambridge (United Kingdom) for his comments on the manuscript.
Footnotes
a.
Baytril 10% injection, Bayer pic, Berkshire, United Kingdom.
b.
Buscopan™ Compositum, Boehringer Ingelheim Limited, Berkshire, United Kingdom.
c.
Pentoject, Animalcare Limited, Yorkshire, United Kingdom.
