Abstract
Nephroblastomas are uncommon embryonal tumors in dogs. We report herein a blastema-predominant nephroblastoma with gingival metastasis in an 8-y-old Miniature Pinscher dog. Histologically, the mass was composed mainly of blastemal elements with minor epithelial and mesenchymal differentiation. Metastatic masses in the gingiva had histologic and immunohistochemical features similar to those of the primary renal nephroblastoma. Neoplastic cells were extensively positive for both vimentin and PAX8, and scattered positive for cytokeratin. Using the clinical staging of human Wilms tumor, we staged our case as stage IV with <4 mo of survival time. We summarized previous studies of canine renal and spinal nephroblastomas, and analyzed the correlations among clinical staging, histologic classification, and mean survival time of dogs with renal nephroblastomas. Clinical staging was significantly correlated with survival time, as shown in humans. In dogs, however, additional factors can potentially influence the outcome of treatment and disease development.
Nephroblastomas, also known as Wilms tumors or embryonal nephromas, originate from rests of metanephric blastema and represent defective nephrogenesis.1,6,23 The component subtypes of such tumors, whether epithelial, stromal, or blastemal, reflect incomplete conversion of the primitive blastemal cells during renal organogenesis. The blastemal element consists of anastomosing serpiginous aggregates or sheets of small hyperchromatic blastemal cells. 6 The epithelial element is usually composed of small tubules or cysts lined by primitive columnar or cuboidal cells; the mesenchymal element consists of spindle cell stroma, which may differentiate to a variety of soft tissues, such as myxoid and fibroblastic spindle cell stroma, skeletal or smooth muscle, cartilage, bone, and fat. 6 Renal nephroblastomas are not common in dogs or most other animal species, except for chickens and pigs.6,23 Usually developing in young animals and sometimes in fetuses, the tumors may remain unnoticed before becoming fatal or be identified as an incidental finding during surgery or autopsy. In dogs, metastasis occurred in 50% of reported nephroblastoma cases, whereas metastasis of spinal nephroblastomas is rare (Table 1). Herein, we report a case of a blastema-predominant canine nephroblastoma with gingival metastases.
Summary of canine renal and spinal nephroblastomas in the veterinary literature.
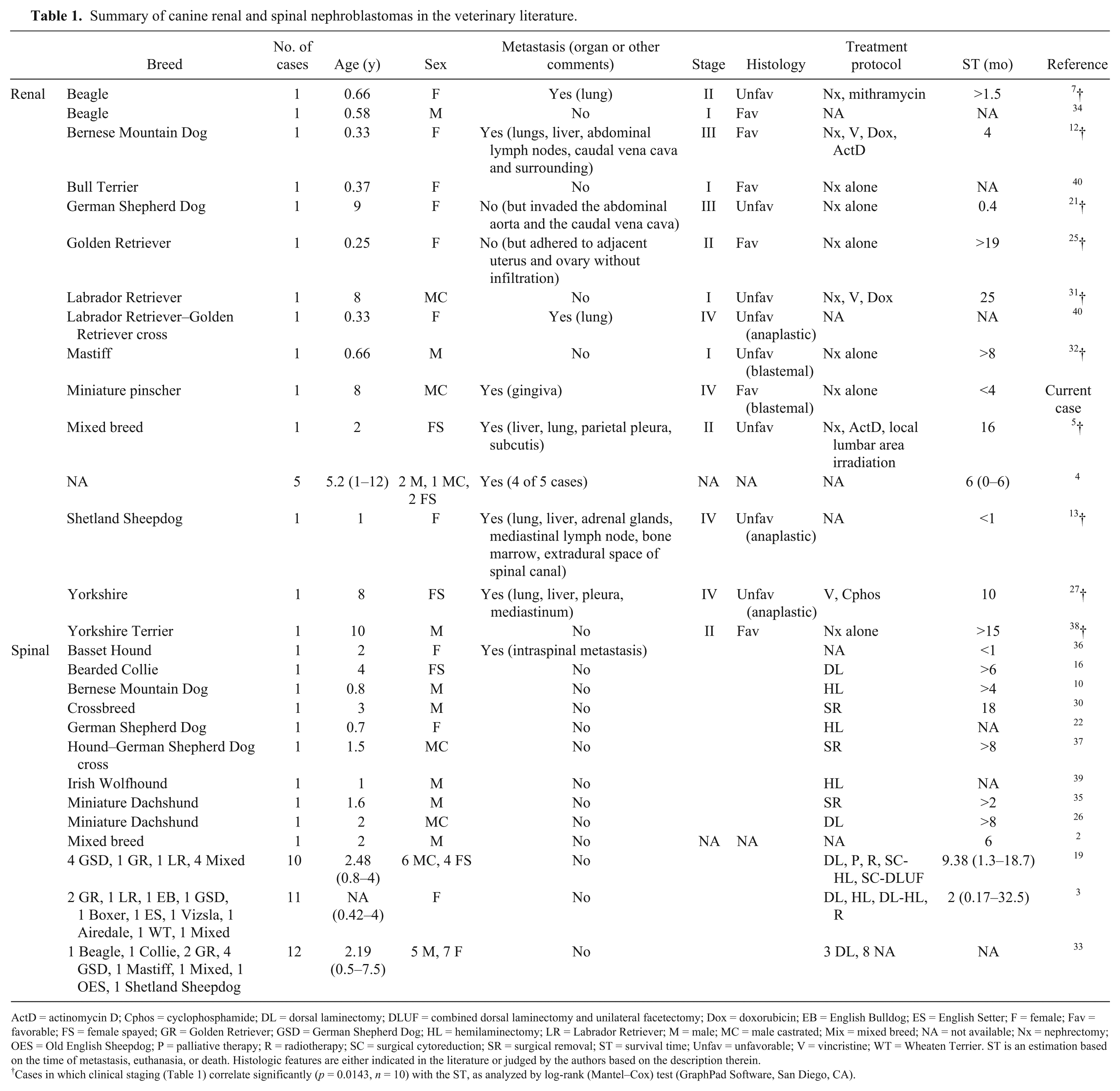
ActD = actinomycin D; Cphos = cyclophosphamide; DL = dorsal laminectomy; DLUF = combined dorsal laminectomy and unilateral facetectomy; Dox = doxorubicin; EB = English Bulldog; ES = English Setter; F = female; Fav = favorable; FS = female spayed; GR = Golden Retriever; GSD = German Shepherd Dog; HL = hemilaminectomy; LR = Labrador Retriever; M = male; MC = male castrated; Mix = mixed breed; NA = not available; Nx = nephrectomy; OES = Old English Sheepdog; P = palliative therapy; R = radiotherapy; SC = surgical cytoreduction; SR = surgical removal; ST = survival time; Unfav = unfavorable; V = vincristine; WT = Wheaten Terrier. ST is an estimation based on the time of metastasis, euthanasia, or death. Histologic features are either indicated in the literature or judged by the authors based on the description therein.
Cases in which clinical staging (Table 1) correlate significantly (p = 0.0143, n = 10) with the ST, as analyzed by log-rank (Mantel–Cox) test (GraphPad Software, San Diego, CA).
An 8-y-old castrated male Miniature Pinscher dog had hematuria and elevated renal indices for >1 mo. Ultrasonography and radiography revealed a mass caudal to the stomach on the left side, extending into and partially filling the peritoneal cavity, with differentials of renal cell carcinoma and nephroblastoma. Swelling of the gingiva was noted shortly afterwards. Both the abdominal and gingival masses were excised after a blood transfusion that was administered because of severe anemia.
Grossly, the 12 × 12 × 13 cm abdominal mass was a partially encapsulated irregular solid mass with nodular protrusions (Fig. 1). The mass was firm and white, with dark-red mottling and irregular depressions. The 0.5 × 0.7 × 1 cm gingival specimen was firm, irregular in shape, and beige-to-tan with dark-red spots.
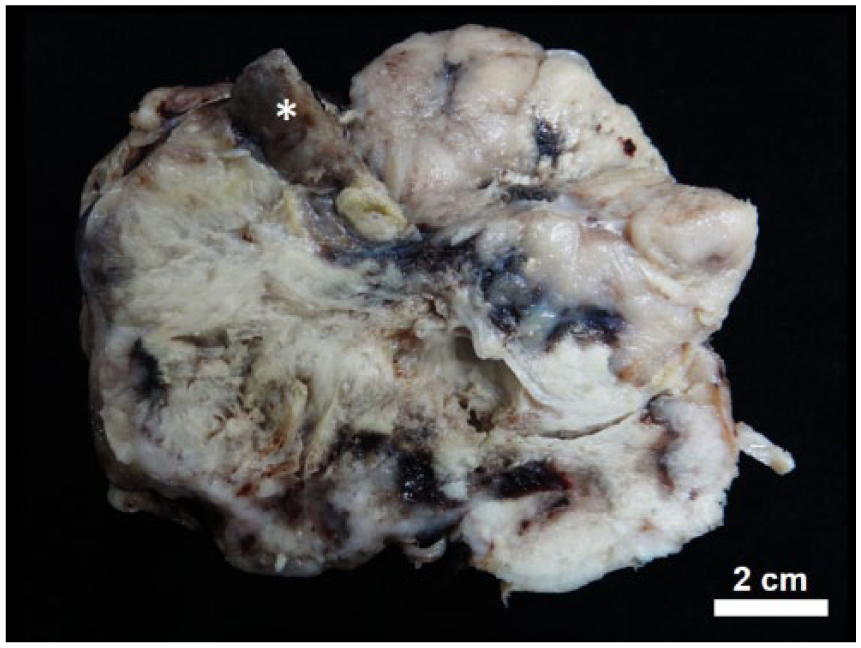
A 12 × 12 × 13 cm white raised renal nephroblastoma with random dark-red mottling and irregular areas of depression. The remaining renal parenchyma is brown (*). Bar = 2 cm.
Tissues were fixed in 10% neutral-buffered formalin, processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin. Microscopic examination revealed a partially encapsulated, poorly demarcated, and lobulated invasive neoplasm replacing and compressing the renal parenchyma (Fig. 1). The neoplasm was composed of a disorganized mixture of 3 distinct elements, mainly blastemal elements with minor epithelial or mesenchymal differentiation. The blastemal element was composed of polygonal neoplastic blastemal cells arranged in sheets, nests, and ribbons (Fig. 2A). These cells had scant indistinctly bordered eosinophilic cytoplasm, and contained a round-to-oval vacuolar nucleus with indistinct nucleoli (Fig. 2B). The epithelial element was composed of cuboidal-to-columnar cells arranged in tubules with occasional luminal tufts, forming “primitive glomeruli” (Fig. 2C). Separating and surrounding the blastemal and epithelial elements was the mesenchymal component of spindle cells loosely arranged in poorly delineated streams. The mitotic rates of the neoplastic blastema, epithelial, and mesenchymal cells were 7–8, <1, and <1 per 400× field, respectively. There were multiple areas of hemorrhagic necrosis, corresponding to the dark-red foci and depressions seen grossly. Based on these histologic features, blastema-predominant renal nephroblastoma was diagnosed. The gingival masses shared histologic features (Fig. 2D) with primary renal nephroblastoma, suggestive of a distant metastasis, which was supported by immunohistochemistry (IHC). Metastasis most likely occurred hematogenously, which is suggested by emboli of neoplastic cells in the renal vasculature (Fig. 2E) and severely dilated vascular lumens in the superficial lamina propria of the gingiva (Fig. 2F).
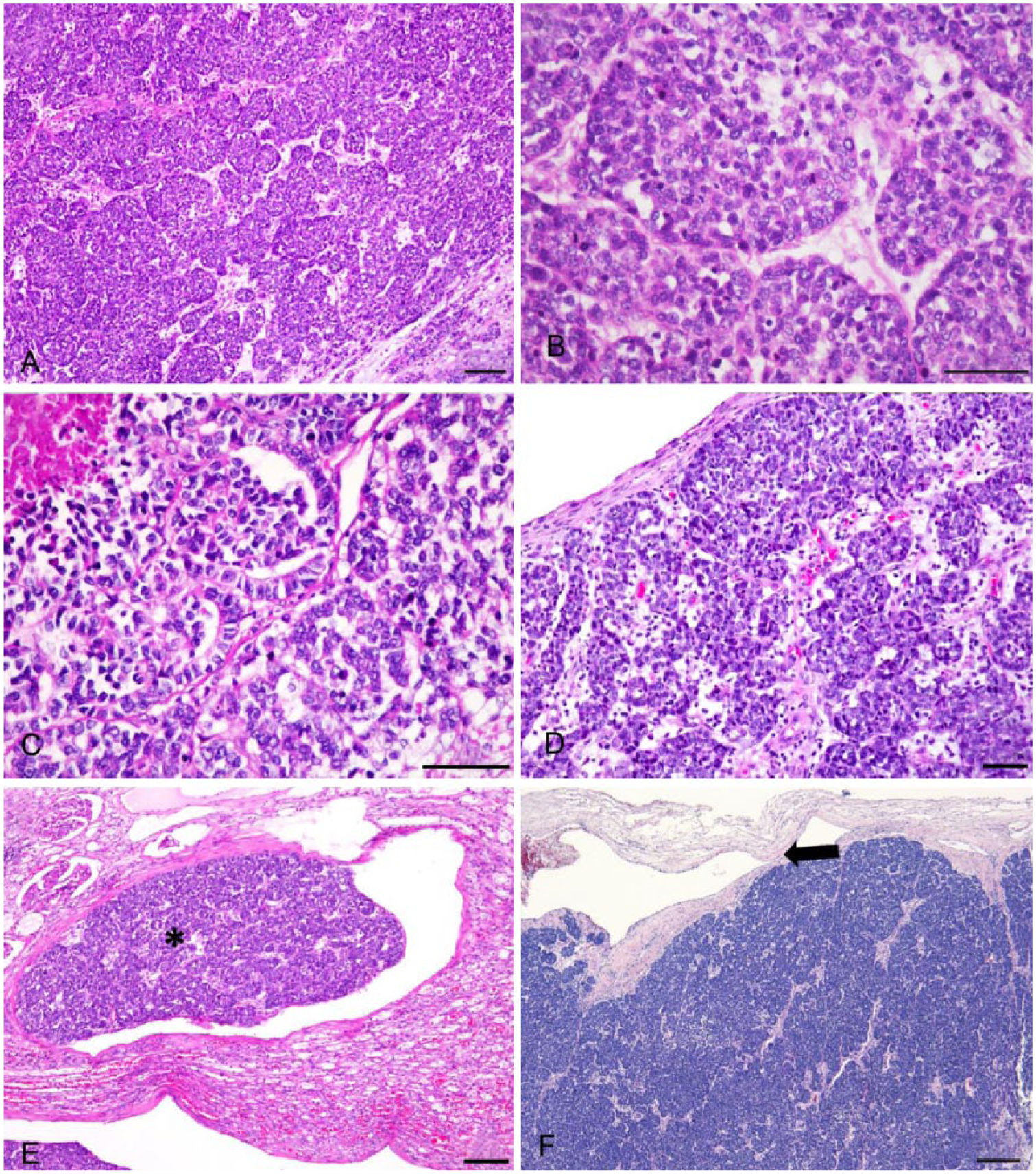
Microscopic findings of the renal and gingival nephroblastoma.
To identify the origin of the renal and gingival masses, IHC for cytokeratin (CK), vimentin, melan A, and paired box gene 8 (PAX8) was performed.14,18,28 The deparaffinized tissue sections were heated in retrieval solution (Trilogy, Cell Marque, Rocklin, CA), and incubated in 2.5% normal goat serum in Tris-buffered saline (TBS) solution for 30 min at room temperature. Tissue sections were then incubated with each of monoclonal antibody against CK (diluted 1:400; AE1/AE3, Dako, Glostrup, Denmark), vimentin (diluted 1:400; clone Vim 3B4, Dako), melan A (diluted 1:100; clone A103, Novocastra Laboratories, Newcastle upon Tyne, UK), and PAX8 (diluted 1:150; clone ZR1, Zeta, Sierra Madre, CA) at room temperature for 1 h. The sections were then treated with 3% hydrogen peroxide followed by peroxidase-conjugated secondary antibody (Real EnVision-HRP rabbit/mouse, Dako) at room temperature for 1 h. After exposure to 2% diaminobenzidine for 3 min, the slides were counterstained with hematoxylin for 15 s. Internal controls from the normal areas of stained tissues for the specific monoclonal antibodies were positive, indicative of credible results of the IHC staining. Neoplastic cells from both the kidney and gingiva showed similar positive patterns for CK, vimentin, and PAX8, but were diffusely negative for melan A. Some foci of the blastemal cells were positive for CK (Fig. 3A, 3B). Most areas showed positivity for vimentin (Fig. 3C, 3D) and PAX8 (Figs. 4, 5). These results are consistent with the gingival neoplasm being a metastasis of the renal nephroblastoma.
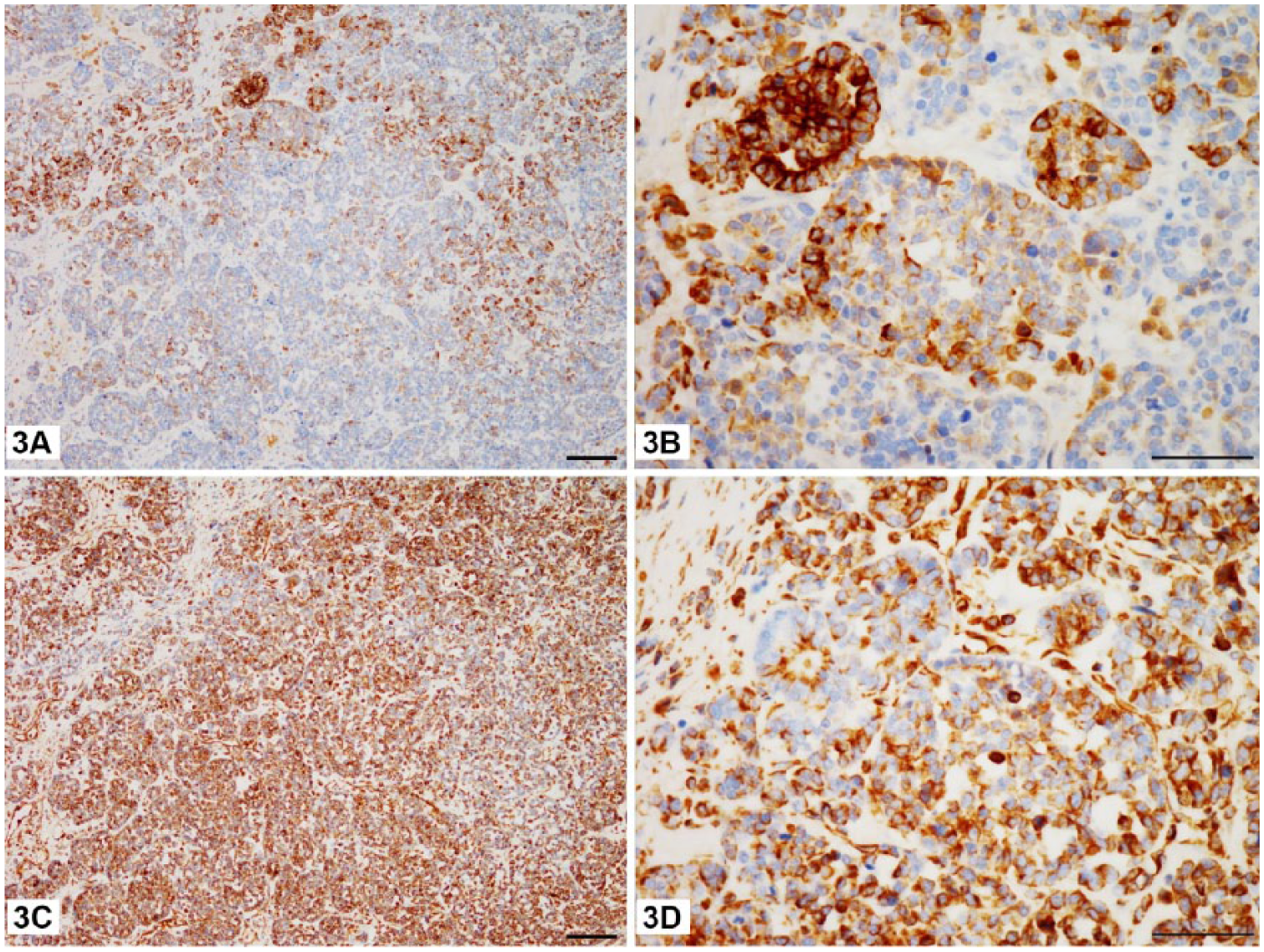
Expression of cytokeratin and vimentin in the renal mass.
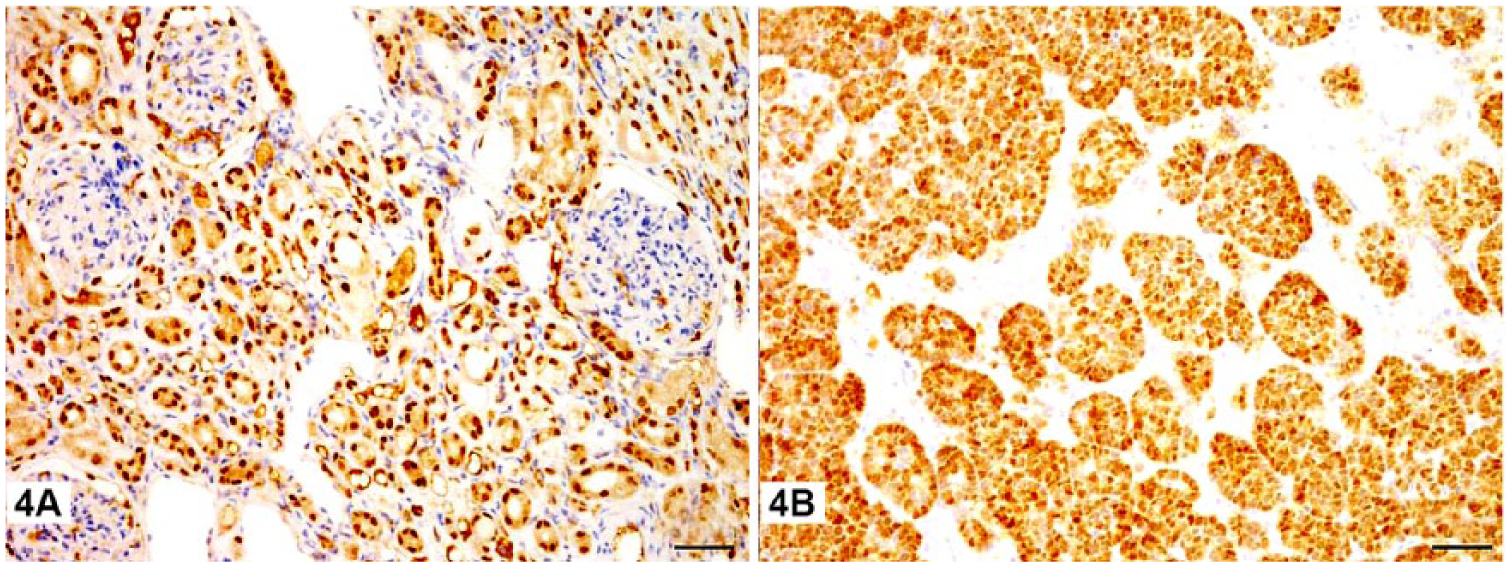
Positive nuclear labeling of PAX8 in the renal mass.
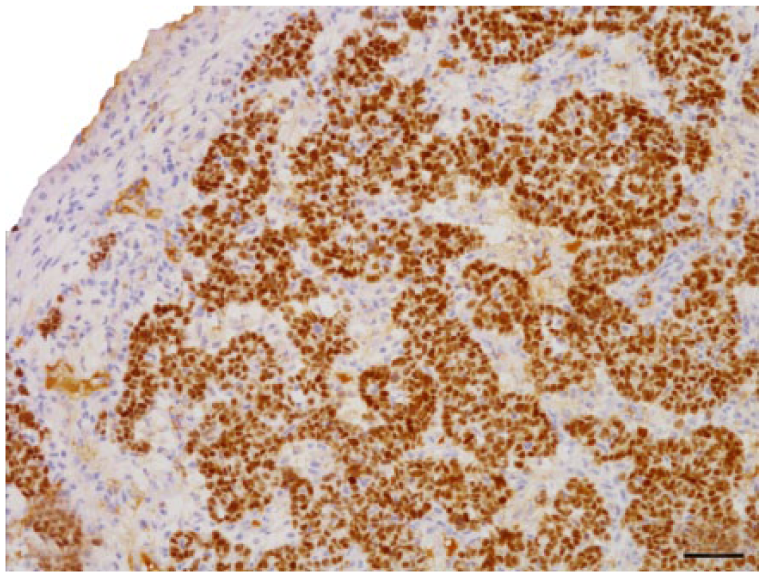
Neoplastic cells in the gingival mass are diffusely and intensely positive for PAX8. Non-keratinized squamous epithelium of the gingiva is visible in the upper left corner. Bar = 50 μm.
Three cell types, epithelial, mesenchymal, and blastemal, are present in a classic “triphasic” nephroblastoma. Renal nephroblastomas tend to occur in juvenile and middle-aged dogs (Table 1),4,6,21,27,31 generally without breed or sex predilection. One or both kidneys may be affected, and kidneys can be fused into one large abdominal mass. 6 Ectopic foci involving the spinal cord may also occur.3,6,19,37 Spinal nephroblastomas commonly occur in dogs from 3 mo to 12 y of age (Table 1) and are thought to originate from the metanephric blastema or from a persistent nephrogenic rest (NR) that is trapped in the dura during development.3,13,24
Half of the previous canine cases of renal nephroblastoma had widespread metastases, and metastatic sites included the contralateral kidney, lung, liver, adrenal, ovary, thymus, mesentery, lymph nodes, thyroid, spinal cord, and bone marrow (Table 1).4,5,12,13,21,27 To our knowledge, gingival metastasis from a primary renal nephroblastoma has not been reported previously. This gingival nephroblastoma was further confirmed by positive immunoreactivity with PAX8, indicative of renal origin (Fig. 5). PAX8-positive tumors may be of renal, Müllerian, thymic, and thyroid origin.18,28 In our case, the major blastemal element readily ruled out those non-renal origins. The similar expression pattern of CK, vimentin, and PAX8 between masses in both locations (Figs. 2–5) was supportive of its renal origin.
A previously reported blastema-predominant canine nephroblastoma 32 had the blastemal element but lacked the glomeruloid structures shown in Figure 2C. In our case, blastemal cells expressed both vimentin and scattered CK markers, implicating an antigenic shift from vimentin to CK (Fig. 3) and resembling the mesenchymal–epithelial transition (blastemal to tubular or glomerular) that occurs in normal organogenesis. 29 In humans, the presence of NRs, which are foci of persistent nephrogenic cells resembling those of the developing kidney, represents failure of maturation of fetal tissue to normal renal parenchyma and has been considered a precursor of renal nephroblastoma.1,20 NRs can be categorized into perilobar (located at the periphery of the renal lobes with predominance of blastemal cells) and intralobar (located in the cortex or medulla of the renal lobe with predominance of stromal element), and may be single, multiple, or diffuse.1,8,11,20 Furthermore, multiple or diffuse NRs are also known as nephroblastomatosis.11,20 NRs can be classified by their histologic feature as 1) dormant or nascent, 2) maturing, sclerosing, and obsolescent, 3) hyperplastic, or 4) neoplastic.1,11,19 Dormant or nascent NRs are small and composed of blastema with extremely rare mitotic figures; maturing, sclerosing, and obsolescent NRs usually show differentiation into stromal and epithelial cells with hyalinization of stroma. 11 Hyperplastic NRs are usually macroscopic lesions with a nodular growth pattern and composed of blastemal, embryonic, or sclerosing regions. 11 Neoplastic NRs can be subclassified into adenomatous and nephroblastomatous types, and are thought to give rise to or be synonymous with nephroblastoma.1,11,20
Nephroblastomas are the most common primary renal cancer in children. Advances in clinical staging and treatment allow for a 2-y survival rate of ~90% in children.9,17 Histopathologic features (favorable or unfavorable) and clinical stages (I–V) are the 2 most significant prognostic factors in human renal nephroblastomas,15,17 and have been adopted for canine cases.12,21,25,31 Tubular and glomerular differentiation indicate a good prognosis, whereas anaplasia and sarcomatous stroma are associated with metastasis and poor prognosis.6,17,25 Anaplasia is present in ~5% of human renal nephroblastomas with focal or diffuse distribution in histology. To identify unfavorable histology (Table 1), 17 3 criteria must be met: 1) nuclei enlarged to at least 3 times the size of adjacent nuclei of the same cell type; 2) marked hyperchromasia of the enlarged nuclei; and 3) multipolar mitotic figures, in contrast to more regular “bipolar” mitotic figures. Our case did not match these features of anaplasia. However, the blastemal cells were primitive and mitotically active with aggressive growth, signifying a higher risk of malignancy. 17
In our case, hematogenous metastasis to the gingiva qualified the case as stage IV (advanced stage); however, the blastemal-predominant element did not suggest “unfavorable” histopathology. This discrepancy between clinical staging and histopathologic classification suggests that the current human staging method (Supplementary Table 1) is insufficient for canine cases. Parameters on some reported canine renal and spinal nephroblastomas are summarized in Table 1. The dog in our case had been found dead in a follow-up conducted 4 mo later, without autopsy, so the exact survival time is unknown. Even so, according to previously reported cases of canine renal nephroblastomas, clinical staging was significantly correlated with survival time (p = 0.0143, log-rank [Mantel–Cox] test; GraphPad Software, San Diego, CA; n = 10, those marked by † in Table 1), as shown in humans. However, there was no statistically significant correlation between histopathologic features (including both favorable and unfavorable as listed in Table 1) and survival time (p = 0.9002, log-rank [Mantel–Cox] test; GraphPad Software; n = 10, those marked by † in Table 1).
In veterinary medicine, factors other than those listed in Supplementary Table 1 may independently or interactively influence survival time. Some of these factors include the age of onset (pediatric patients tend to have poorer prognosis), tumor size and rate of growth, histologic subtypes, complete or incomplete resection of kidney or accompanied ureter or regional lymph nodes, therapy protocol, postoperative care, side effects of therapy and surgery, paraneoplastic syndromes, and concurrent neoplasia.4,5,7,12,17,21,25
Supplemental Material
DS1_JDVI_10.1177_1040638718762560 – Supplemental material for A blastema-predominant canine renal nephroblastoma with gingival metastasis: case report and literature review
Supplemental material, DS1_JDVI_10.1177_1040638718762560 for A blastema-predominant canine renal nephroblastoma with gingival metastasis: case report and literature review by Bo Chen, Wen-Ta Li and Fun-In Wang in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
