Abstract
Canine end-stage renal disease (ESRD) is defined as the almost complete failure of renal function or irreversible destruction and is characterized by extensive glomerular sclerosis, tubular atrophy, interstitial inflammation, and fibrosis. Renal fibrosis is a common pathway leading to kidney failure. Infiltrating immunocytes in the end-stage kidney and several related factors are involved in renal fibrogenesis. A total of 18 renal tissue samples were obtained from canine patients with ESRD using biopsy and necropsy procedures. The extent of renal fibrosis was histopathologically examined by Masson trichrome staining. T-cell and B-cell localization and macrophage lineages were determined by immunohistochemical staining. Additionally, interleukin-1 (IL-1), IL-2, and IL-6 levels in the canine ESRD kidney were immunohistochemically evaluated and compared with expression patterns in the normal kidney. Significant fibrosis and infiltrating immunocytes consistent with lymphocytes were observed. Although the B-cell count was increased in the end-stage kidney, immunostaining patterns disclosed a marked increase in the number of CD3+ cells. Furthermore, the remarkable increase in IL-1 and IL-6 levels suggests that T cells in the kidneys of dogs with ESRD spontaneously express these cytokines. In this study, the correlation between the degree of renal fibrosis and cytokines in canine ESRD was examined. The present study shows that T lymphocytes and IL-6 play important roles in renal fibrosis.
Introduction
End-stage renal disease (ESRD) represents the late stage of kidney disease, whereby the residual renal function is less than 5%.3,11 Fibrosis is an important response in renal disease as a final common pathway that leads to chronic progressive renal failure. Inflammatory and postinflammatory phases are well characterized and involve several cytokines, such as epidermal growth factor (EGF), fibroblast growth factor (FGF), and transforming growth factor-β (TGF-β). 20 At all phases of the ESRD kidney, infiltrating mononuclear inflammatory cells are identified in the tubulointerstitial space. In humans, the contribution of B cells to renal interstitial inflammation and its importance were analyzed in an earlier study. 9 These infiltrating cells possibly stimulate the phenotypic conversion of a heterogeneous group of resident fibroblasts and tubuloepithelial cells into activated fibroblasts with EGF, FGF, and TGF-β in the inflammatory phase. 20 Histopathologically, severe fibrosis and infiltrating mononuclear cells are observed in the canine ESRD kidney, similar to human renal diseases. 2
Cytokines secreted by immunocytes may be critical in regulating the synthesis of extracellular matrix (ECM) components by fibroblasts. The canine ESRD kidney contains a large number of infiltrating immunocytes. With retrospective materials of canine ESRD kidney, the infiltrating immunocytes and the expression of cytokines known to mainly act in the proinflammatory phase are helpful in understanding fibrosis. In this study, the infiltrating immunocytes and cytokines secreted in canine ESRD disease were identified. With the aid of immunohistochemistry (IHC) analyses, the distribution of mononuclear immunocytes and quantities of specific cytokines mainly acting in the proinflammatory phase were evaluated. The correlation between fibrosis and infiltrating immunocytes in canine ESRD and the roles of immunocytes are further investigated.
Materials and methods
Study population
This study used archival renal tissues from necropsy or biopsy from canine patients with ESRD (n = 18) between 2003 and 2006. Renal tissues were collected in the Konkuk University Veterinary Medical Diagnostic Laboratory (Small Tumor Diagnostic Center, Seoul, Korea). Diagnosis was based on light microscopy with hematoxylin and eosin staining. The following criteria were applicable for ESRD diagnosis: interstitial fibrosis with tubular atrophy and biochemical/clinical evidence of uremia. Most of the cases (14/18) had a history of azotemia (blood urea nitrogen = 180.7 ± 71.1 mg/dl, serum creatinine = 8.5 ± 4.7 mg/dl), and the 4 cases lacking any clinical information showed severe renal calcification. Among the canine patients, 13 were female and 5 were male. The mean patient age was 9.5 years (range: 5–16 years). Kidney tissues obtained from dogs that died without renal disease were used as the negative control (n = 3).
Preparation of tissues for IHC
Renal specimens were fixed in 10% neutral buffered formalin and subsequently embedded in paraffin. Serial 4-μm sections were acquired from each paraffin block. For immunohistochemical analysis, a set of monoclonal and polyclonal antibodies was used. Primary antibodies included polyclonal rabbit antihuman CD3, a antimouse CD79α, a antimouse CD20cy, a antimouse CD68, a and 2 types of antimouse CD4. b,c Antimouse interleukin-1 (IL-1), b antimouse IL-2, b and polyclonal antigoat IL-6 b were also applied for evaluation of cytokines in canine ESRD. The antibodies were diluted 1:50 (CD4), 1:70 (IL-1 and IL-2), 1:100 (CD3, CD79α, CD20cy, and CD68), or 1:150 (IL-6). Two-step Envision system-AP d and Envision system-HRP e were applied for detection.
The IHC analysis was essentially performed as previously described. 23 Briefly, slides were deparaffinized in xylene and hydrated in graded ethanol. Tissue sections were treated with 3% hydrogen peroxide (H2O2) solution for 20 min at room temperature, followed by phosphate buffered saline (PBS) washing 3 times. The antigens were retrieved by boiling the sections in tris-ethylenediamine tetra-acetic acid buffer (pH 9) for 10 min in the microwave oven (high power). After cooling, the slides were washed 3 times in PBS. Subsequently, sections were incubated with primary antibodies in the refrigerator (4°C) overnight. For secondary polymer, Envision system-AP was used for CD3, CD4, IL-1, IL-2, and IL-6. Envision system-HRP was used for detecting CD20cy, CD68, and CD79α. The secondary polymer was applied to each slide for 40 min at room temperature, and the slides were washed in PBS 4 times. Next, the slides were incubated with substrates for Envision system-AP and Envision system-HRP until the desired staining intensity developed. The color reaction was stopped by washing in distilled water twice and counter-stained with Harris hematoxylin.
Masson trichrome staining
To estimate the degrees of fibrosis, Masson trichrome staining was performed. The slides acquired by the same method with preparation for IHC were deparaffinized and rehydrated in the distilled water. The sections were first treated in 56°C Boulin fixative solution for 1 hr. After cooling at room temperature for 10 min, they were washed in running water and rinsed in distilled water. Ten minutes of Weigert iron hematoxylin staining, 17 min of Biebrich scarlet-acid fuchsin staining, and differentiation in phosphomolybdic-phosphotungstic acid solution for 10 min followed. Finally, collagen and area of fibrosis were stained with aniline blue solution for 10 min and differentiated in 1% acetic water for 1 min. The slides were dehydrated and cleared through graded ethyl alcohol.
Digital image analysis and statistical analysis
Digital images were acquired using microscope f and digital image transfer software. g Morphometric analysis was performed with image analysis software h on 40X magnification and 200X magnification. Three random fields of renal cortex were selected in each slide. The area stained blue with Masson trichrome stain was used for analyzing renal fibrosis on 40X magnification. Immunocytes and cytokines were assessed with the area stained positive (AP: stained red area, HRP: stained brown area) with the software h on 200X magnification. All positive signs utilized a point counting, and the positive area (positive-sign pixels) was divided by total area (total pixels of the picture) to gain the positive area rate.
For the comparison of means, nonparametric Mann-Whitney U-test was used. P < 0.05 was considered to be statistically significant. SigmaPlot 8.0 was used for preparation of the graphs.
Results
Degree of fibrosis analyzed with Masson trichrome staining
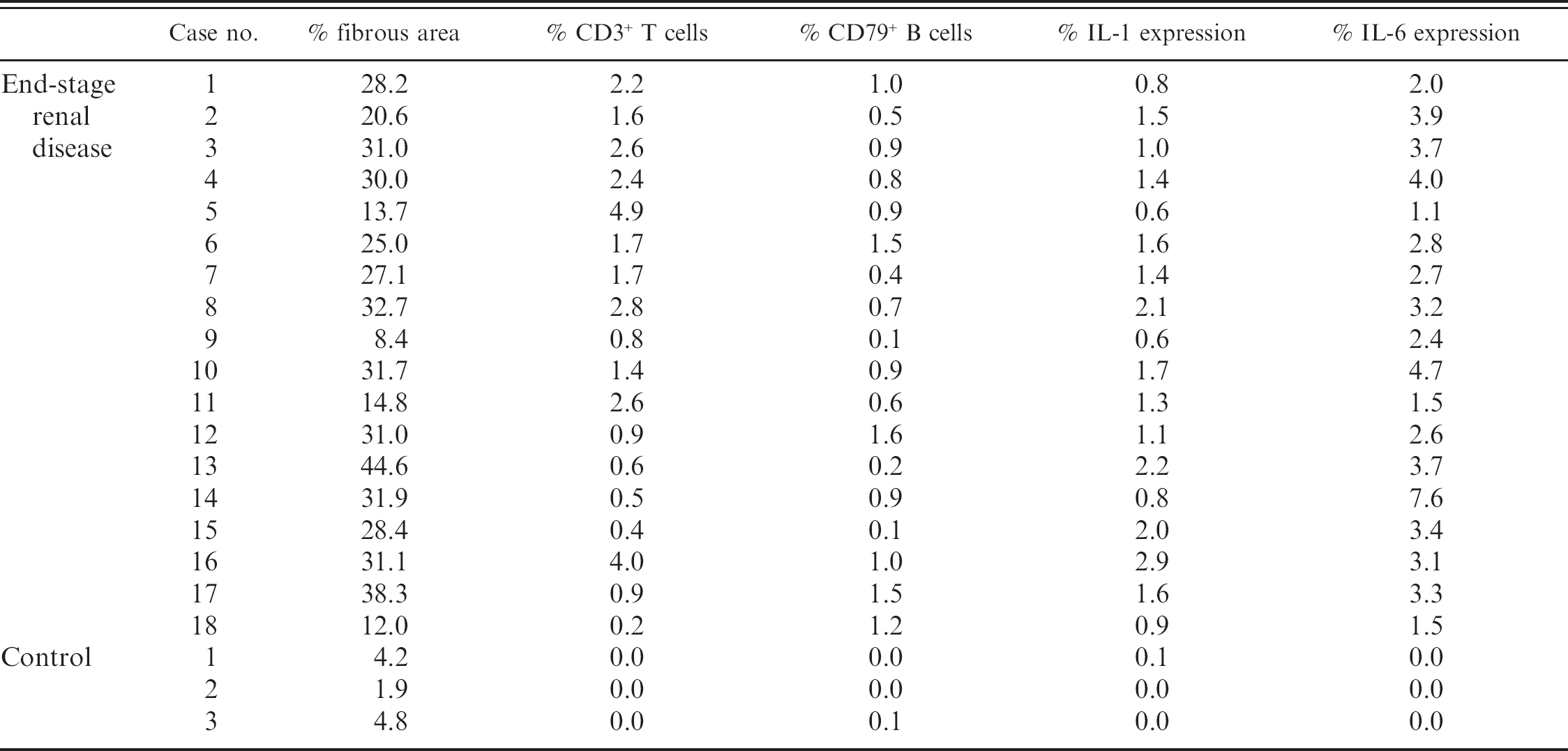
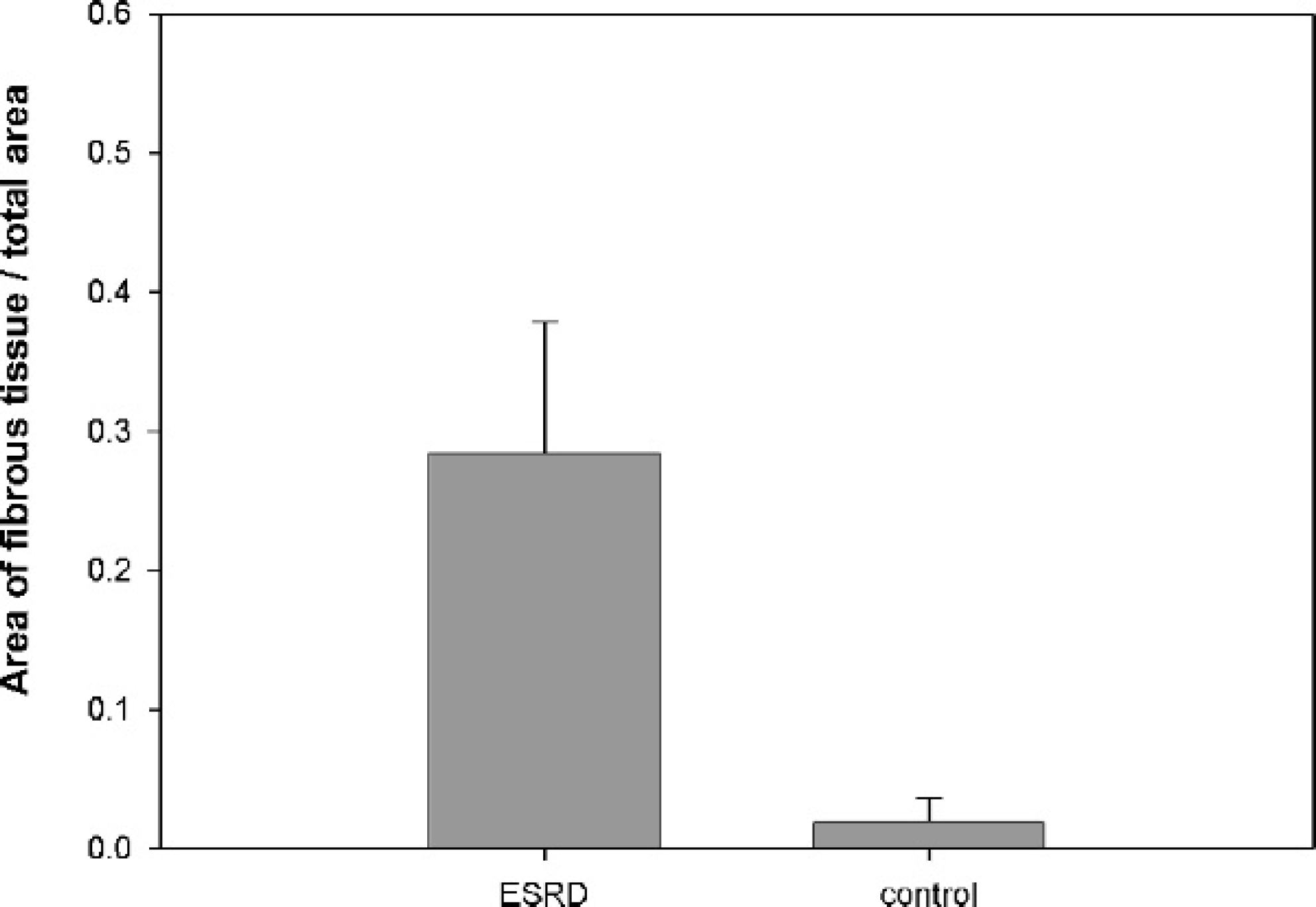
Increased fibrosis detected in the canine ESRD kidney was quantitatively estimated (Table 1). The area of fibrosis included proximal tubules destroyed, deposition of ECM, and spaces of infiltrating immunocytes. The fibrous tissue areas in ESRD and control kidneys are shown in Figure 1 (P < 0.01). The ESRD kidney contained a higher fibrous tissue area than the control kidney (Figs. 2A, 4A, 5A). Fibrosis was particularly severe in the renal cortex, which had large numbers of infiltrating immunocytes.
Histopathology and immunostaining of immunocytes
In the canine kidney with ESRD, severe infiltration of mononuclear immunocytes in the cortex was evident. In contrast, mononuclear immunocytes were rarely observed in control kidneys.
Expression patterns of CD3 and CD79α were used to determine the distributions of T cells and B cells in the ESRD kidney, respectively. The regions of infiltrating lymphocytes and severe fibrosis coincided in serial sections (Fig. 2A, 2B, 2C). Figure 3 displays a quantitative comparison of the positive areas for CD3 and CD79α between ESRD and control kidneys (P < 0.01). In the control kidney, CD3+ and CD79α+ cells were rarely observed. In contrast, ratios of the areas staining positive for CD3 and CD79α were each scored 1.8% and 0.8% in ESRD kidneys, respectively (Table 1; Fig. 2B, 2C). B-cell+ area was increased in ESRD kidneys. Moreover, IHC revealed an enormous increase in the number of CD3+ T lymphocytes. CD3+ T cells formed a significant proportion of immunocytes infiltrating the ESRD kidneys. In serial sections, the T-cell area was twice as high as the B-cell area (Fig. 3, P < 0.01).
Fibrous area, areas of T cells and B cells, interleukin-1 (IL-1), and IL-6 in individual animals.
The IHC experiments with anti-CD4 and anti-CD20cy antibodies were consistently negative in the canine ESRD kidney (data not shown). Moreover, experiments with the CD68 antibody for staining mononuclear phagocyte lineage and macrophages revealed few or no positive signals (data not shown).
Immunostaining of cytokines
In the ESRD kidney, IL-1 expression was increased (Table 1) and was diffusely expressed in the fibrosis area (Fig. 4A, 4B). In comparison, IL-1+ signals were obscure or lower in the control kidney (Fig. 5B). In the ESRD and control kidneys, IL-1+ areas were quantitatively compared (Fig. 6, P < 0.05), revealing a 25-fold increase in the ESRD kidney. Moreover, the expression of IL-1 is increased in direct proportion to the rate of fibrous area (Fig. 7A).
Ratio of fibrous tissue area to total tissue area; canine end-stage renal disease (ESRD) kidney contains about 28% fibrous tissues in renal cortex. In contrast, control kidney includes 4% fibrous tissues in renal cortex (P < 0.01).
Quantitative comparison of the IL-6+ areas between ESRD and control kidneys was additionally performed (Fig. 6, P < 0.05). The IL-6+ signals were extremely low in the control kidney (Fig. 5C). High IL-6 expression was evident around the glomeruli and the region of infiltrating lymphocytes (Table 1; Fig. 4C). Notably, IL-6 expression in the ESRD kidney was about 150 times higher than that in the control kidney (Fig. 6, P < 0.05). In the current study, increased IL-6 expression in direct proportion to the rate of fibrous area was also confirmed (Fig. 7B).
There was no positive proof that the IL-2 is increased in the ESRD kidney. The IHC data obtained with anti-IL-2 was not distinctly positive in both ESRD and control kidneys (data not shown).
Discussion
Repair of damaged tissues is a fundamental biologic response that allows the replacement of cells after injury, a mechanism critically important for survival. 26 Fibrosis is often defined as a wound-healing process that has escalated out of control. Renal fibrosis, characterized by the excessive accumulation of type I collagen, is a major pathologic feature in renal disease triggered as a repair mechanism. 13 Renal fibrous tissue mainly comprising ECM is synthesized by resident fibroblasts, myofibroblasts, and epithelial-mesenchymal transition-derived fibroblasts. 7,16,20,21 The term “epithelial-mesenchymal transition” is used preferentially to describe the conversion of terminally differentiated epithelia into cells with a mesenchymal phenotype. 10 In renal fibrosis, it is theorized that tubular epithelial cells in areas of tubular basement membrane rupture may move into the interstitium and differentiate into fibroblasts producing ECM. 20,21 The tubular epithelial cells are stimulated by mononuclear cells secreting cytokines such as EGF, FGF, and TGF-β at the moment. 20
Kidney, end-stage renal disease (ESRD); canine.
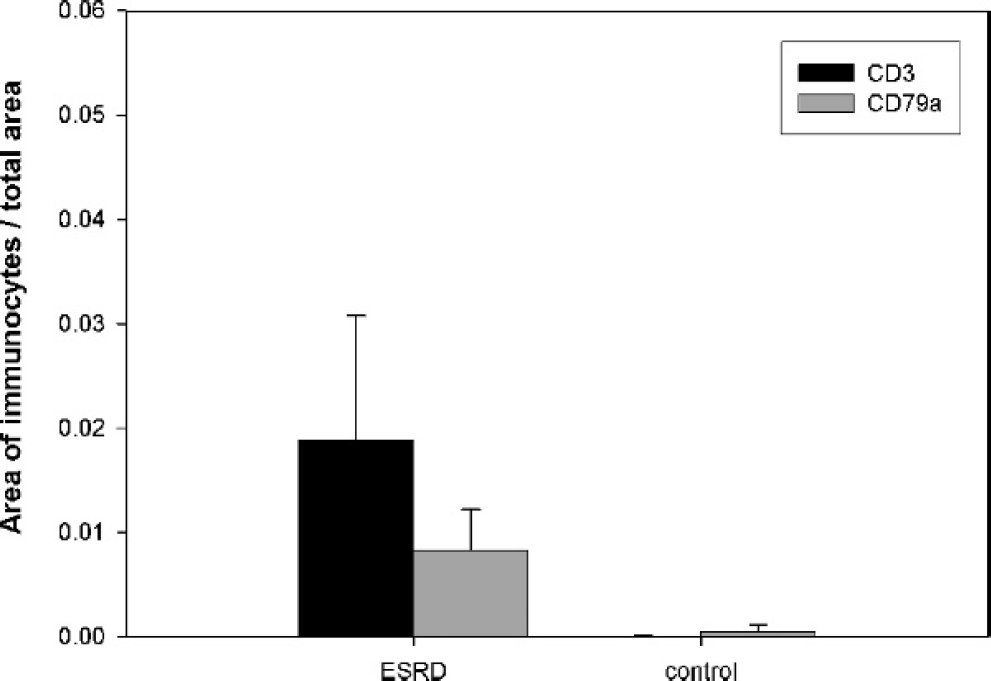
Area of immunocytes proportion in canine kidney; CD3 T cells are enormously increased. CD3+ T cells are counted approximately 2 times higher than CD79α B cells in canine endstage renal disease (ESRD) kidney (P < 0.01).
In the present study, the fibrous tissue content was 3.7% in the normal parenchyma of the control kidney. In contrast, the fibrous tissue area in the renal cortex of the ESRD kidney was 26.7%, 7 times higher than that of the control kidney. Fibrosis was severe in the renal cortex, with large numbers of infiltrating immunocytes.
Infiltrating immunocytes were identified as T lymphocytes and B lymphocytes using IHC. CD3+ T cells formed a significant proportion of ESRD infiltrates. The T-cell population was about twice as large as the B-cell population (Fig. 3, P < 0.01). T helper (Th) cells play a significant role in T-cell immune responses by secreting various cytokines. A previous report about human renal disease showed that Th1 cells secrete interferon-γ, IL-2, and tumor necrosis factor-β, while Th2 cells generate IL-4, IL-5, IL-6, and IL-10. 22 In view of their cytokine secretion properties, the T-cell population in the ESRD kidney may mainly consist of Th cells. The current study used 2 different products of CD4 b,c as the Th-cell marker; however, both CD4 antibodies failed to detect canine Th cells in the infiltrates. This finding does not necessarily signify the absence of Th cells because canine tonsil tissue used as a positive control did not stain for CD4. It must be noted that both CD4 antibodies were designed for humans and have not been tested in dogs.
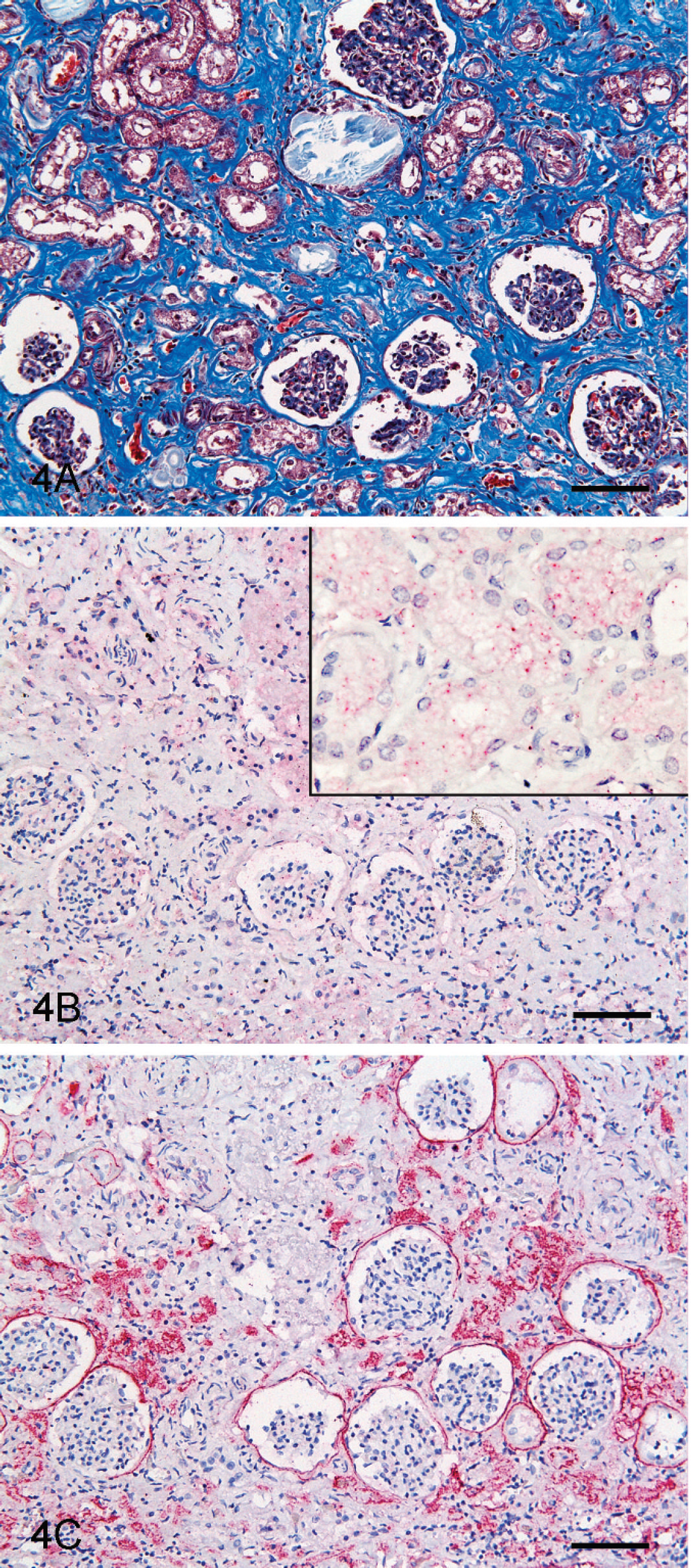
Kidney, end-stage renal disease (ESRD); canine.
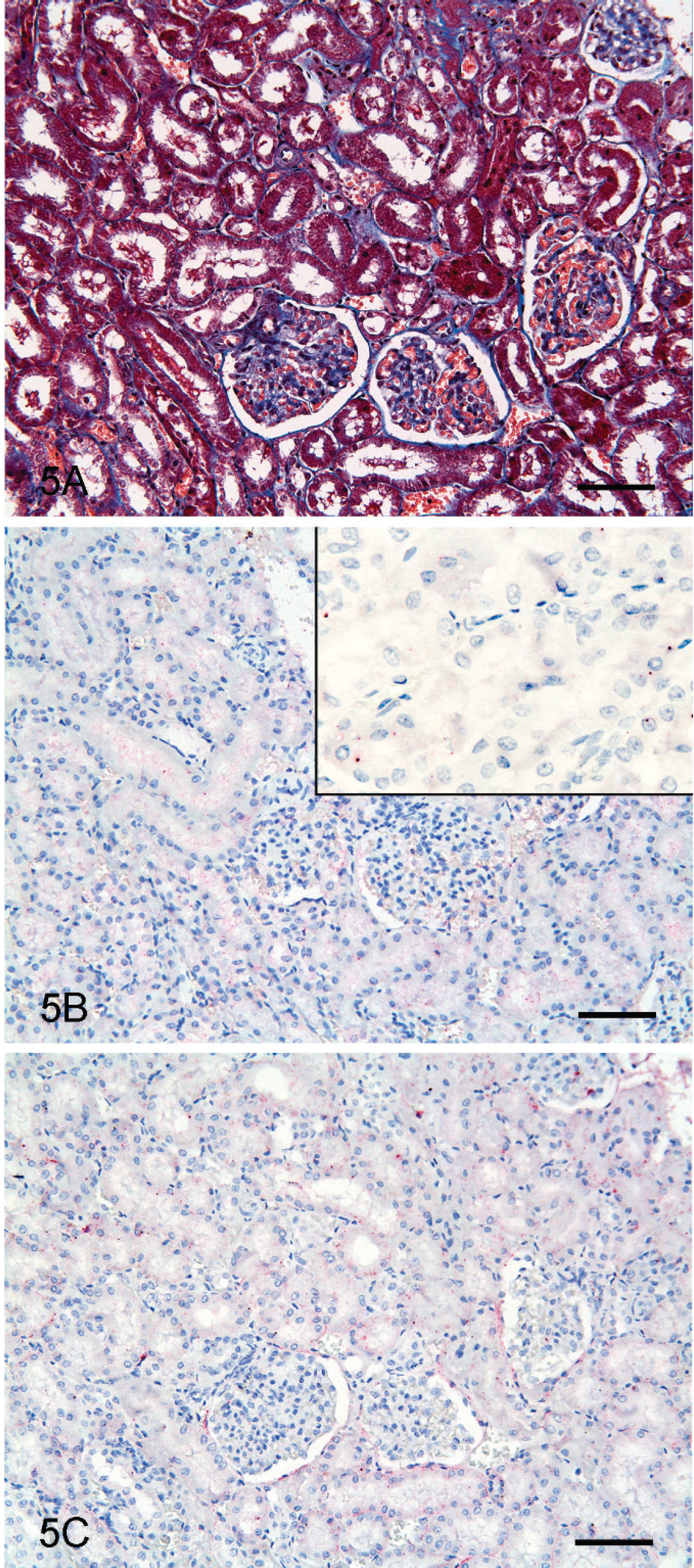
Kidney; canine.
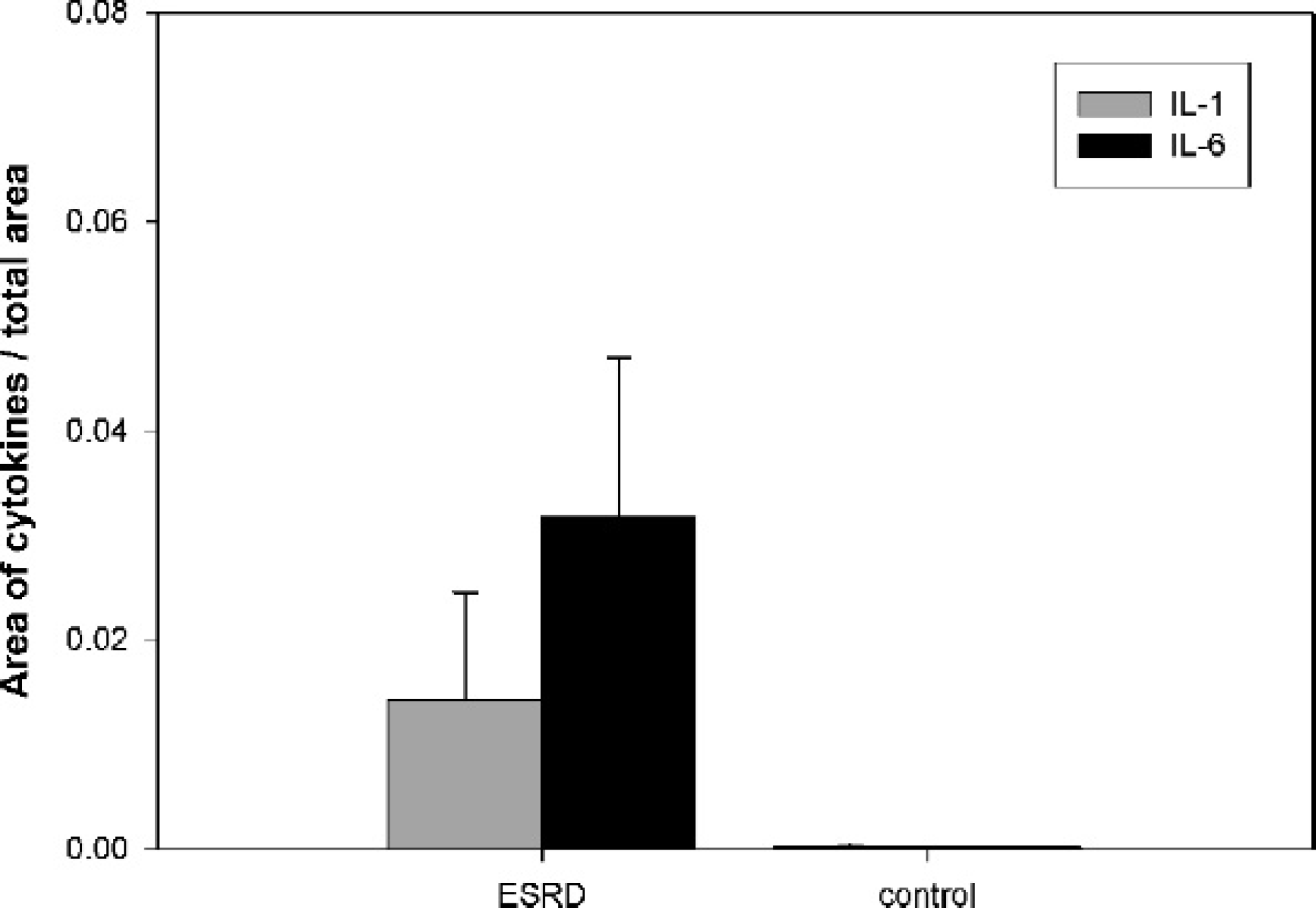
Area of cytokines in canine kidney. Both interleukin-1 (IL-1) and IL-6 are increased in canine end-stage renal disease (ESRD) kidney. IL-1 in ESRD kidney was expressed about 25-fold of increase, and expression of IL-6 in ESRD kidney was about 150 times higher than control kidney (P < 0.05).
In the ESRD kidney, CD79α+ B-cell counts were also observed. To describe further stages of B-cell maturation, the CD20cy marker was used. The CD20cy marker is expressed by B cells (from pre-B cell to the preplasma cell stage), 5 and CD20cy+ B cells form a prominent part of human interstitial nephritis. 9 However, CD20cy+ B cells were not detected in the infiltrates of the canine ESRD kidney in this study. The tissue of canine B-cell lymphoma was used as a positive control, and it was positive.
CD68+ mononuclear phagocyte lineage cells or macrophages were scarcely found in this study. Lung with pneumonia and many alveolar macrophages was used as a positive control in this study, and it was positive. Mononuclear phagocyte lineage cells and macrophages did not appear to be a significant component of the inflammation in the cases of ESRD in the current study. The current study showed that macrophages are not significantly involved in the inflammation of canine ESRD.
In renal fibrosis, fibroblasts are mainly stimulated by cytokines, such as EGF, FGF, connective tissue growth factor, and TGF-β in the inflammatory phase. Synthesis of ECM components by fibroblasts may be partly regulated by cytokines in the inflammatory phase. However, cytokines secreted in the induction phase as a trigger stage may play a critical role in regulating the synthesis of ECM components. The cellular and molecular mechanisms of excessive ECM production are enhanced expression of the collagen gene of fibroblasts. These enhanced expressions are related to abnormal activation of receptors and signaling pathways. 19,24 In addition to participating in B-cell maturation, 6 IL-1 is an activator of T cells and B cells, as well as NK cells. It is primarily released from stimulated macrophages and monocytes, and renal fibroblasts display increased IL-1 gene expression. 6,14 Consistently, IL-1 expression in the ESRD kidney was about 25 times higher than that of control kidney, and the expression of IL-1 is increased in direct proportion to the rate of fibrous area. Elevated IL-1 in canine ESRD kidneys may activate infiltrating T cells and B cells. Experiments with anti-CD68 disclosed few or faint mononuclear phagocytes secreting IL-1 in the ESRD kidney.
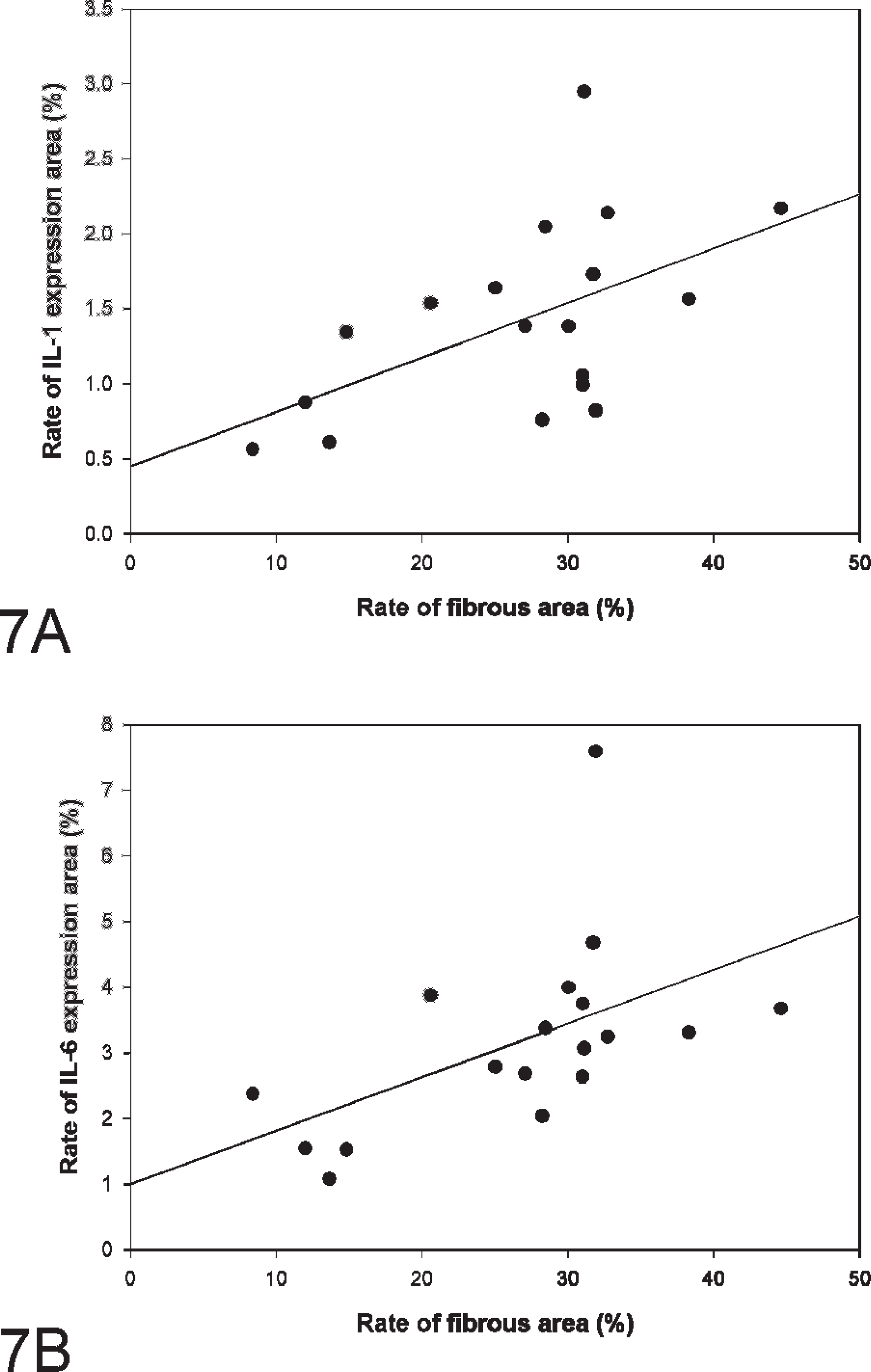
Correlation between the degree of renal fibrosis and cytokines in canine end-stage renal disease (ESRD).
IL-6 is a cytokine secreted by T cells, epithelial cells, and IL-1-activated fibroblasts. 1,17,22 It is usually not constitutively produced by normal cells, but is readily induced by a variety of cytokines, 18 lipopoly-saccharides, 15 or viral infections. 18 The results of the current study show that expression of IL-6 in the ESRD kidney is about 150 times higher than that of control kidney, and the expression of IL-6 is increased in direct proportion to the rate of fibrous area. Moreover, IL-6 expression localizes in the regions of infiltrating lymphocytes and around the glomeruli in the present study (Fig. 4C). This pattern of expression indicates that T lymphocytes in the infiltrate regions and IL-1-activated fibroblasts around the glomeruli secrete IL-6 as an immune response in the ESRD kidney. In addition to stimulating collagen synthesis in fibroblasts, IL-6 is generally involved in the induction of B-cell differentiation, proliferation, and differentiation of T cells. 4,6,8 In the canine ESRD kidney, IL-6 may also play an important role in the control of matrix synthesis by performing these functions.
Interleukin-2 is a lymphokine secreted by activated lymphocytes, which acts as a cofactor in the replication and differentiation of T cells, B cells, and NK cells during inflammation. 6,13 It appears to interact with IL-2 to stimulate the proliferation of T lymphocytes. 26 However, no IL-2 expression in the canine ESRD kidney was observed.
In conclusion, renal fibrosis of canine ESRD kidney is characterized in the present study. The fibrous tissue content was 3.7% in the renal cortex of the control kidney. In contrast, the histopathology of ESRD kidneys displayed 26.7% of fibrous area with variable numbers of infiltrating mononuclear cells. T cells forming a significant proportion of infiltrates appear to play a key role in the renal fibrosis of canine ESRD kidney, in view of the finding that the areas of fibrosis and the T cells coincide. B cells were also increased in the ESRD kidney and may be partly involved in renal fibrosis. Proinflammatory cytokines (including IL-1 and IL-6) that control fibrosis as a trigger of inflammation and renal fibrosis in dogs displayed elevated expression. The results of the current study provide histopathologic evidence that T lymphocytes and IL-6 are involved in controlling renal fibrosis of the canine ESRD kidney.
Acknowledgements
Thanks to Ms. R. H. Jang for excellent technical assistance. This paper was supported by Konkuk University in 2008. This report represents a thesis submitted by J.-Y. Yhee to fulfill the requirements for a PhD degree.
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
Santa Cruz Biotechnology, Santa Cruz, CA.
c.
BioGenex Laboratories Inc., San Ramon, CA.
d.
LSAB kit, Dako North America Inc., Carpinteria, CA.
e.
Dako REALTM Envision kit (K5007), Dako North America Inc., Carpinteria, CA.
f.
Olympus, BX41, Melville, NY.
g.
Leica Application suite 2.7, Heerburg, Switzerland.
h.
Image Pro Plus 4.1, Media Cybernetics Inc., Bethesda, MD.
