Abstract
The current study sought to compare the effectiveness of 2 virus isolation methods for the recovery of contemporary
Introduction
The emergence of the influenza A (H1N1)pdm09 virus and the subsequent detection of numerous genomic variant strains of
Pigs are often called “mixing vessels” for FLUAV strains because the receptors present in the porcine respiratory tract allow pigs to become infected with swine-, human-, and avian-origin FLUAV strains. 21 The pig’s susceptibility to FLUAV strains originating from multiple host species facilitates interspecies virus transmission and genomic reassortment in pigs thus making swine a source of novel FLUAV strains.26,31 Consequently, comprehensive surveillance programs targeted at the FLUAV strains circulating in swine populations could aid in the detection of emerging viruses that may threaten animal and human health. The effectiveness of these virus surveillance programs is reliant upon the effectiveness of virus isolation protocols to detect and characterize all FLUAV strains in the swine population.
Nasal swabs are routinely used for the antemortem detection of FLUAV strains in swine. 33 Isolation of such strains from nasal swabs collected from pigs has been performed using a variety of substrates,10,12,14,39 with embryonated chicken eggs (ECEs) and Madin–Darby canine kidney (MDCK) cells being most common.8,25,45 The preferential growth of FLUAV strains in various culture substrates has been repeatedly documented and is strain dependent.6,29,36,45 Embryonated chicken eggs have historically been the gold standard for isolating FLUAV strains.7,33,51 More recently, MDCK cells have been shown to be sensitive to a wide range of FLUAV strains, especially those circulating in mammals, and thus, are now routinely used for the isolation and propagation of mammalian-origin FLUAV strains.10,13
The continuous genetic drift and frequent genetic reassortment occurring among swine-origin FLUAV strains since the emergence of the triple-reassortant H3N2 virus in 1998 23,34,52 requires virus isolation protocols to be periodically validated with contemporary viral strains. The purpose of the present study was to compare the effectiveness of MDCK cells adapted to serum-free culture and ECEs for the recovery of contemporary FLUAV strains circulating in swine at agricultural exhibitions.
Materials and methods
Study sites for the investigation included 12 agricultural exhibitions in Ohio during July, August, and September of 2009. Nasal swabs were collected from 221 clinically healthy, market-weight pigs according to the World Organization for Animal Health and the Food and Agriculture Organization of the United Nations joint network of expertise on animal influenza guidelines (OFFLU: 2009, Collection of specimens for detection of influenza from swine. Available at http://www.offlu.net/index.php?id=184. Accessed on November 12, 2012). Samples were collected on the last or next to last day of the swine exhibition period at each fair. Protocol number 2009A0134 for animal use was approved by the Institutional Animal Care and Use Committee of The Ohio State University. A single nasal swab collected from each pig was placed in an individual vial containing 1.8 ml of brain heart infusion (BHI) broth supplemented with penicillin (10,000 U/ml) and streptomycin (10 mg/ml). 40 The vials were kept in a cooler with cool packs during sample collection, transported directly to the laboratory on dry ice, and then stored at −70°C until testing was initiated.
At the time of testing, vials were quickly thawed in a 37°C water bath. Amphotericin B (20 μg/ml), gentamicin sulfate (1,000 μg/ml), and kanamycin sulfate (325 μg/ml) were added to each vial as previously described to control bacterial and fungal contamination.4,43 Each vial was then vigorously agitated, after which, the vial was centrifuged at 1,200 ×
Virus isolation in MDCK cell culture
The methods used for FLUAV isolation in MDCK cell culture were adapted from previously described protocols44,49 to utilize a serum-free culture system. To summarize, MDCK cells a were seeded in tissue culture flasks and grown in minimum essential medium with Earle salts and L-glutamine b supplemented with 1.0 mM sodium pyruvate, c 0.1 mM nonessential amino acids, d and 10% fetal bovine serum (FBS). e Cells were maintained at 37°C in a 5% CO2 incubator. Once a productive cell line was established, MDCK cells were transitioned to serum-free medium (SFM) f by decreasing the concentration of FBS over 4 passages in a stepwise fashion according to the manufacturer’s guidelines. f Less than passage-25 MDCK cells fully adapted to SFM were used for all virus isolation attempts. The SFM-adapted MDCK cells were seeded into every other vertical column of a 24-well cell culture plate. One of the 4 wells in each empty column of wells was also seeded with SFM-adapted MDCK cells to serve as a negative control. This allowed for 3 samples to be tested in quadruplicate on one 24-well plate, with each sample having a negative control. Immediately prior to inoculation, the SFM from confluent cell monolayers in 24-well tissue culture plates was removed, and the monolayers were washed once with viral growth medium (VGM), which was removed immediately prior to inoculation. Viral growth medium was prepared by supplementing SFM with antimicrobials g (1,200 U/ml of penicillin, 1.2 mg/ml of streptomycin, and 3 µg/ml of amphotericin B) and tolylsulfonyl phenylalanyl chloromethyl ketone–treated trypsin (0.5 μg/ml).
For each sample, 150 μl of BHI broth supernatant was inoculated into each of 4 wells of monolayered MDCK cells prepared as described above. Inoculum was allowed to adsorb for 60 min at 37°C before being removed and replaced with 0.5 ml of VGM. Plates were incubated at 37°C in 5% CO2 for 72 hr. Wells were observed daily for cytopathic effects (CPE) by inverted light microscopy, and the results were recorded. After 72 hr, supernatant from each well was tested for hemagglutination activity using 0.5% turkey red blood cells in V-bottom plates. 18 Hemagglutinating agents were screened for FLUAV strains using a commercially available diagnostic kit to identify FLUAV antigen. h If CPE and hemagglutination were not present, media from the 4 wells representing 1 original sample was aseptically harvested and pooled. Pooled first-pass media was stored at −70°C until a second passage was performed. Second passage was carried out by inoculating the pooled first-passage media in quadruplicate onto monolayers of MDCK cells as described above. If no hemagglutinating activity was detected after 2 passages, the media was harvested and pooled in the same manner used for the first-pass media, and subsequently tested with a FLUAV M gene–targeted real-time RT-PCR (described below) to verify the negative result.
Virus isolation in embryonated chicken eggs
All samples were tested with standard methods previously described.2,49 Briefly, 150 μl of clarified viral transport media was inoculated, in quadruplicate, via the allantoic sac into 10 day-old, specific pathogen–free embryonated chicken eggs. Eggs were incubated at 37°C with 45% relative humidity and candled daily with dead eggs removed and placed in refrigeration at 4°C. Embryos alive after 60 hr were chilled at 4°C overnight. Chorioallantoic fluids from all eggs were tested for hemagglutinating agents and FLUAV strains as described above. If hemagglutination was not detected, the chorioallantoic fluids from the 4 eggs representing 1 original sample were aseptically harvested, pooled, and stored at −70°C until a second passage was performed. Pooled first-passage chorioallantoic fluids were inoculated into 4 eggs to perform a second passage. If no hemagglutinating activity was detected after 2 passages, chorioallantoic fluids were harvested and pooled in the same manner as the first passage and then tested for the FLUAV M gene with real-time RT-PCR.
Real-time reverse transcription polymerase chain reaction
All original clarified samples and samples found negative after 2 passages in either virus isolation method were tested using a modified version of previously described FLUAV M gene–targeted real-time RT-PCR procedures.41,42 Briefly, RNA was purified using an RNA extraction kit i according to the manufacturer’s protocol. Following extraction, the RNA was subjected to real-time RT-PCR using a one-step quantitative (q)RT-PCR kit j in a 20-μl reaction mixture containing 10 μl of 2× qRT-PCR master mix, 8 pmol of each primer, 2.4 μM probe, 0.3 μl of diluted reference dye (5-carboxy-X-rhodamine, 1:500), 0.2 μl of 100 mM of dithiothreitol, 1 μl of RT/RNase block enzyme mixture, and 5 μl of extracted RNA. The reactions were performed on a qPCR system k under the following thermocycling conditions: stage 1, 50°C for 10 min; stage 2, 95°C for 3 min; and stage 3, 40 cycles of 95°C for 1 sec and 60°C for 20 sec. Threshold cycle (Ct) values were calculated for each sample automatically by the qPCR system’s software using the background-based method. Samples with a Ct of ≤40 were considered positive.
Subtyping
The genomic hemagglutinin and neuraminidase subtypes of the recovered FLUAV isolates were determined by real-time RT-PCR with primers and probes developed for swine-origin FLUAV strains. 37 RNA was extracted as described above and subjected to real-time RT-PCR using a one-step multiplex RT-PCR kit l in a 25-μl reaction mixture containing 12.5 μl of 2× multiplex RT-PCR master mix, 10 pmol of each primer, 0.2 μM each probe (except N2 probe, which was increased to 0.4 μM), 0.25 μl of RT mixture, and 8 μl of extracted RNA. Hemagglutinin H1 and H3 probes were labeled with hexachlorofluorescein and 5-carboxyfluorescein, respectively, and run as a duplex reaction. The neuraminidase N1 and N2 probes labeled with hexachlorofluorescein and 5-carboxyfluorescein, respectively, were run as a separate duplex reaction. The reactions were performed on a qPCR system k under the following thermocycling conditions: stage 1, 50°C for 20 min; stage 2, 95°C for 5 min; and stage 3, 40 cycles of 95°C for 15 sec and 54°C for 30 sec. Threshold cycle values were calculated for each sample automatically by the qPCR system’s software using the adaptive algorithm. Samples with a Ct of ≤40 were considered positive.
Statistical analysis
The results from virus isolation with MDCK cell culture and ECEs were cross-tabulated and analyzed using the McNemar chi-square statistic with a standard statistical program. 1 Because the nasal swabs were collected from groups of commingled pigs, the underlying assumption of independent samples was not valid, thus the clustering effect was adjusted for using the Eliasziw–Donner procedure. 11 Samples from the fairs where FLUAV strains were not detected by any method were excluded from the statistical analysis because these results did not contribute to the number of discordant pairs and severely skewed the within-cluster correlation.
Detection of α-2,3– and α-2,6–linked sialic acid residues by flow cytometry
A previously described protocol using flow cytometry
15
was modified to quantify the abundance of α-2,3 sialic acid (SA 2,3) and α-2,6 sialic acid (SA 2,6) linkages to galactose expressed by the SFM-adapted MDCK cells. Briefly, freshly harvested passage-20 SFM-adapted MDCK cells (1 × 106 cells/sample) were washed with 5% FBS in phosphate buffered saline with sodium azide and incubated in the dark at 4°C for 30 min in the presence of biotinylated
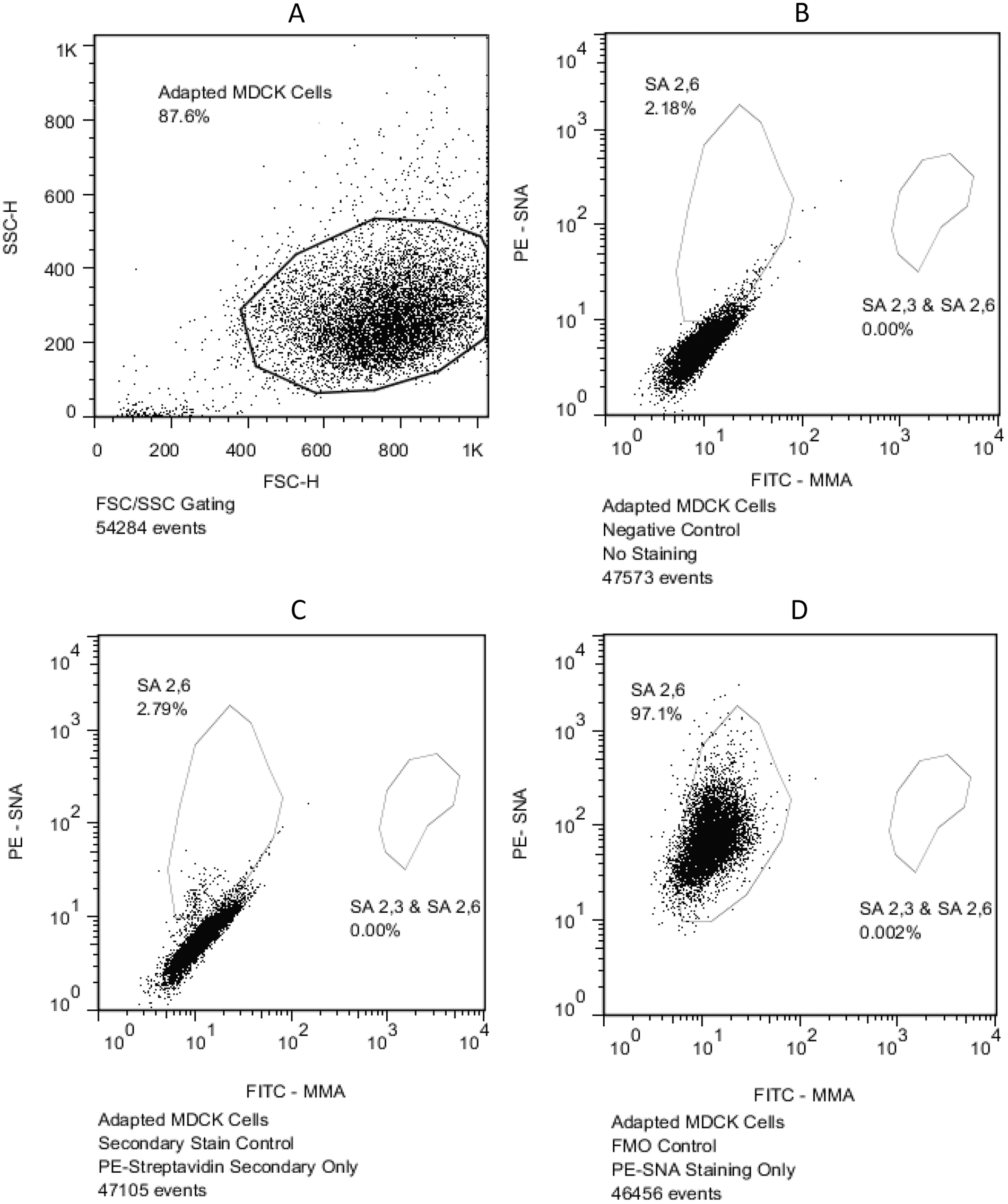
Side scatter (SSC) and forward scatter (FSC) analysis were used to isolate serum-free medium (SFM)-adapted Madin–Darby canine kidney (MDCK) cells (
Results
Of the 221 samples, FLUAV was detected in 40 samples (18.1%) with real-time RT-PCR, 40 samples (18.1%) with virus isolation with MDCK cells, and 13 samples (5.9%) with virus isolation with ECEs. The positive nasal swabs were collected from pigs from 3 of the 12 separate agricultural exhibitions. Virus isolation results with SFM-adapted MDCK cells and real-time RT-PCR analysis of original samples showed perfect agreement. All samples that were positive by ECEs were also positive in MDCK cells, but there were 27 specimens positive in MDCK cells and by real-time RT-PCR that failed to grow in ECEs (Table 1). The adjusted McNemar chi-square value comparing virus isolation in ECEs with virus isolation in MDCK cells was 5.95 (
Isolation of
Adjusted McNemar χ2 = 5.95 (
All CPE-positive cell culture wells were also positive for hemagglutinating activity, and all hemagglutinating agents were identified as FLUAV strains. No FLUAV strains were identified in CPE-negative MDCK monolayers or hemagglutination-negative cell culture fluids. Subtypes H1N2 and H3N2 were recovered from both MDCK cell culture and ECE virus isolation systems. For the 13 isolates recovered in ECEs, 8 (61.5%) were H1N2 and 5 (38.5%) were H3N2. Examining the 40 isolates recovered using SFM-adapted MDCK cells, 12 (30%) were H1N2 and 28 (70%) were H3N2. These hemagglutinin–neuraminidase subtype combinations were consistent with FLUAV strains contemporaneously circulating in U.S. pigs. 30
All FLUAV isolates recovered with MDCK cell culture were recovered during first passage. No additional strains were recovered from second passage in MDCK cell culture (Table 2) or detected by real-time RT-PCR on second-passage cell culture supernatant. In contrast, 8 FLUAV isolates were detected in first ECE passage and 5 additional FLUAV isolates were recovered from second passage in ECEs that were not detected in first passage. Real-time RT-PCR analyses confirmed all other second-passage chorioallantoic fluids as negative for FLUAV strains.
Recovery of
Two-color staining using biotinylated (SNA) lectin and fluorescein isothiocyanate–conjugated MMA lectin demonstrated that more than 98% of SFM-adapted MDCK cells reacted with SNA, indicating dominant SA 2,6 residues in SFM-adapted MDCK cells (Fig. 2). Of those, less than 2.25% of the cells were double-positive, also reacting with the MMA lectin, indicating the presence of SA 2,3 residues. The SFM-adapted MDCK cells with only SA 2,3 residues were lacking, as demonstrated by the failure to detect cells reacting with only the MMA lectin and not the SNA lectin. The finding was further validated by demonstrating that the MMA lectin bound as expected to the positive controls (mouse alveolar epithelial type II cells, which express high levels of SA 2,3 20 ; Fig. 3).
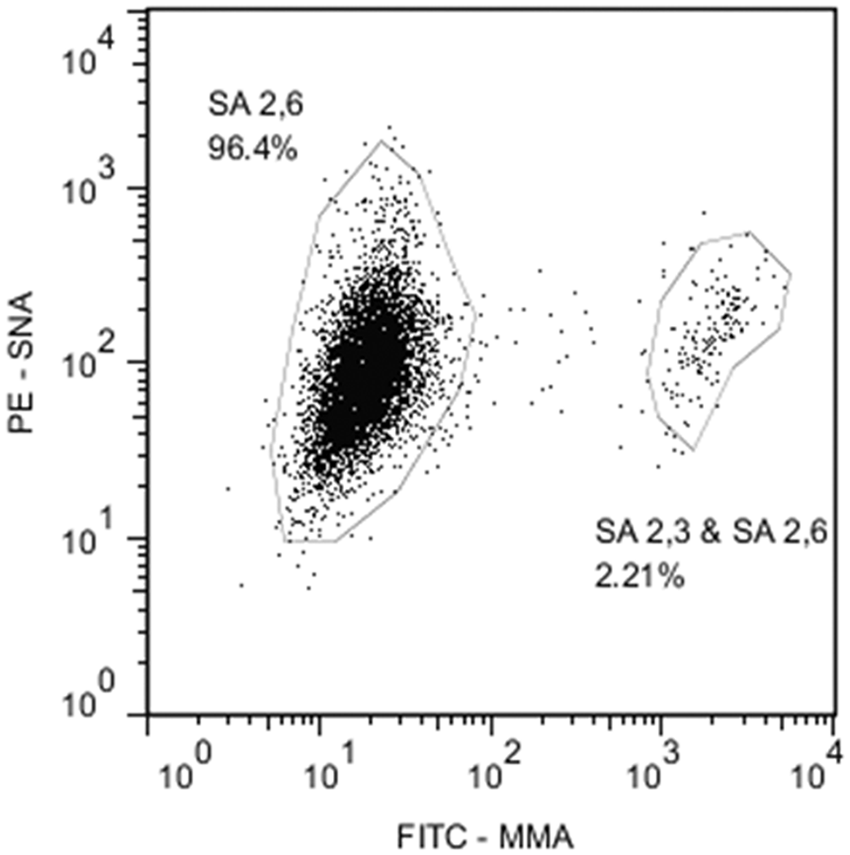
Flow cytometric analysis of serum-free medium (SFM)-adapted Madin–Darby canine kidney (MDCK) cells for the detection of α-2,3– and α-2,6–linked sialic acid (SA 2,3 and SA 2,6, respectively) residues. Greater than 98% of SFM-adapted MDCK cells expressed SA 2,6 residues, and of that population less than 2.25% of the cells also expressed SA 2,3 residues. Cells expressing only SA 2,3 residues were not detected. SNA =
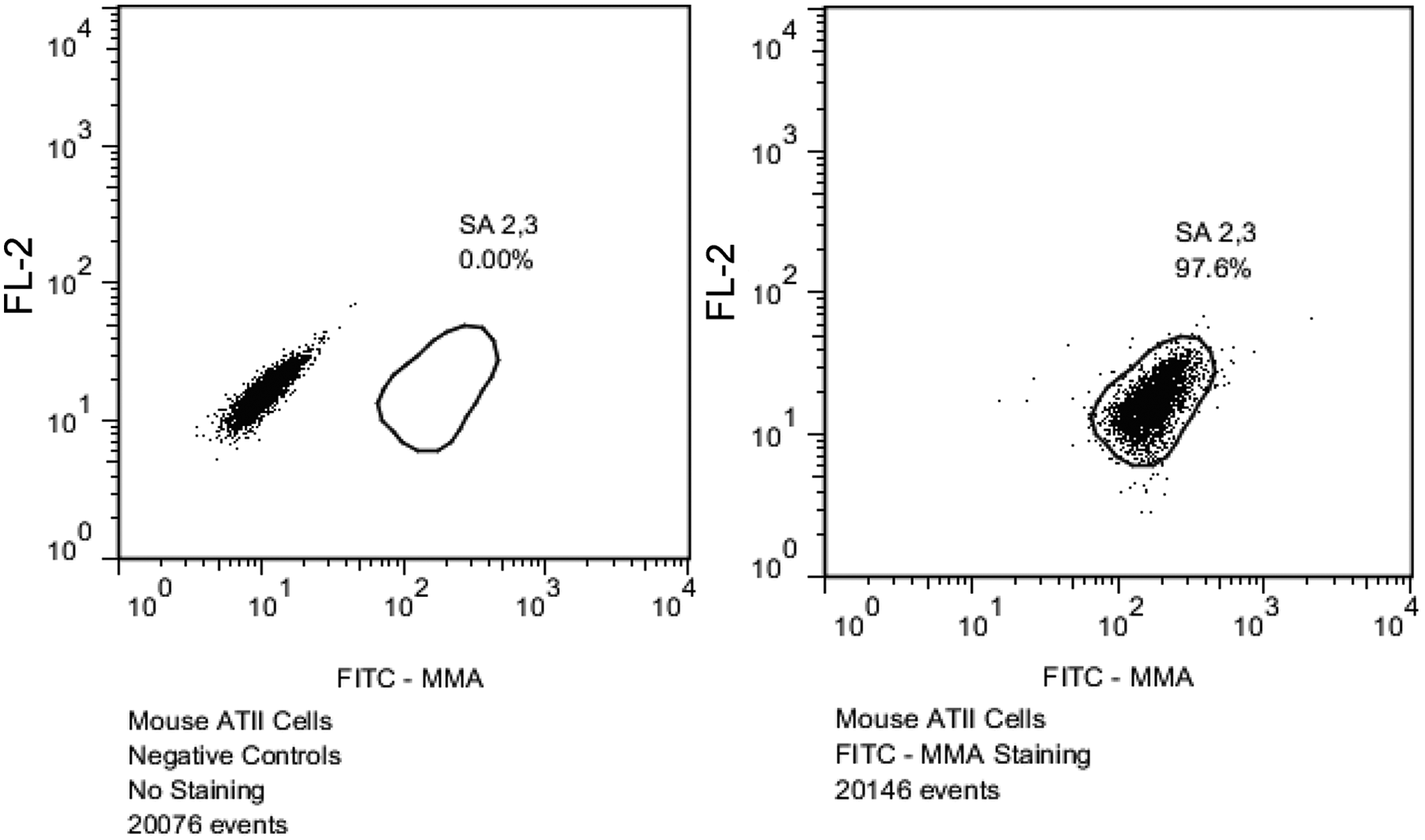
Flow cytometric analysis of mouse alveolar epithelial type II (ATII) cells used as positive controls for the detection of α-2,3–linked sialic acid (SA 2,3) residues. Greater than 98% of mouse ATII cells expressed SA 2,3 as expected. FITC = fluorescein isothiocyanate; MMA =
Discussion
Veterinary and public health officials are increasing surveillance of FLUAV strains in swine in response to numerous calls for earlier detection of emerging strains that may threaten animal and human health.16,17,35 Depending on the objective, virus isolation and/or real-time RT-PCR are currently the primary methods used to detect FLUAV strains among swine. The results of the current study show excellent agreement between real-time RT-PCR and virus isolation in SFM-adapted MDCK cell culture. However, caution must be exercised when interpreting the agreement between real-time RT-PCR and MDCK found in the study because the sampled pigs were in a noncommercial setting, and were likely exposed to FLUAV 5–7 days earlier upon entry into their respective exhibitions via a point-source or near point-source introduction of virus. Previous studies have shown pigs infected with FLUAV begin shedding virus 48 hr postexposure and can continue to shed for up to greater than 10 days.27,38,48 In the present study, nasal swabs were collected at the end of the exhibition, which likely coincided with peak viral shedding in the swine population. Consequently, real-time RT-PCR and virus isolation in SFM-adapted MDCK cell culture performed equally well in this setting. Under different circumstances it would not be surprising to find more real-time RT-PCR–positive samples than virus isolation–positive samples.
In order to amplify FLUAV strains to a detectable level during virus isolation procedures, the use of multiple passages in cell culture and ECE has been widely recommended.33,44,49 The results of the present study show that second passage failed to yield any additional virus isolates in the SFM-adapted MDCK cell culture system. While additional viruses were recovered with a second passage in the ECE system, virus isolation with MDCK cell culture still greatly outperformed virus isolation with ECEs. The additional time and costs associated with performing a second pass in SFM-adapted MDCK cell culture were not necessary under the circumstances of the current investigation. Moreover, even with good laboratory procedures, each additional handling of potentially infectious material significantly increases the likelihood of cross-contamination and thus spurious results. The need to perform multiple passages in either system would be warranted when an original sample or first-passage cell culture fluid is real-time RT-PCR positive but FLUAV was not detected after a single passage.
In the present study, the use of SFM-adapted MDCK cell culture was highly sensitive for the recovery of H1N2 and H3N2 FLUAV strains circulating among swine at the selected agricultural exhibitions. The use of SFM-adapted MDCK cells to cultivate human-origin FLUAV strains has been previously described 24 and is currently being investigated as a substrate to produce live attenuated FLUAV vaccines for human use. 28 Human-origin strains have been reported to grow to higher titers in MDCK cells adapted to SFM than in traditional MDCK culture systems.32,46 The use of SFM culture maintains physiological consistency and eliminates the problems associated with the lot-to-lot variability of FBS. At the time of writing, the cost of SFM and traditional culture media were comparable.
Binding to cell surface receptors with specific sialic acid groups is needed for FLUAV strains to enter a host cell 50 and thus a substrate with an appropriate receptor is required for successful virus isolation. Most avian-origin FLUAV strains preferentially bind to SA 2,3 whereas mammalian-origin strains, especially those infecting human beings, prefer SA 2,6. 3 Conventional MDCK cells express both SA 2,3 and SA 2,6 linkages in similar proportions.15,22,47 The SFM-adapted MDCK cells used in the present study clearly have higher SA 2,6 and lower SA 2,3 receptor levels than traditional MDCK cells. In contrast to the SFM-adapted MDCK cells, the allantoic cells of ECE are known to be rich in SA 2,3 but low in SA 2,6, 22 which likely contributes to the improved recovery of swine-origin FLUAV strains with SFM-adapted MDCK cells. Cells predominantly expressing SA 2,6 could improve the recovery of FLUAV strains threatening swine and human health while selecting against avian-origin strains. Given the lack of SFM-adapted MDCK cells expressing SA 2,3, the protocol described above may be of limited value to investigations of FLUAV strains at the avian–swine interface.
Presently, there are several different lineages of MDCK cells commercially available, 9 which makes poorly defined diagnostic protocols subject to unintentional variability. Additionally, the parental lines of MDCK cells, like those used in the present study, are a heterogeneous population of epithelial cells, which allows for preferential selection of a particular subpopulation of cells. 19 The long-term effects of maintaining MDCK cells in SFM culture have not been thoroughly investigated. Although, only low passage number cells (≤ passage 25) were used in the present study in an attempt to minimize the impact of selection pressure on the MDCK cells, the use of SFM may alter the virus susceptibility of MDCK cells. Traditional MDCK cell culture protocols using FBS 44,49 were not used in the present study due to difficulty maintaining consistently adhered monolayers of MDCK cells when FBS containing cell growth medium was removed and replaced with VGM without FBS following inoculation. By adapting to the protocols to utilize SFM, the MDCK cells were no longer subjected to the sudden removal of FBS, and the resulting monolayers remained adhered to the plate surface, thus aiding in the visual detection of CPE. Ultimately, further studies are needed to compare the use SFM-adapted MDCK cell culture to traditional MDCK cell culture with FBS for the isolation of FLUAV strains from swine.
Under the circumstances of the present study, virus isolation with SFM-adapted MDCK cell culture was significantly superior to virus isolation with ECEs for the recovery of the contemporary FLUAV strains circulating in selected swine populations at the time of sampling. While the strains isolated in the study preferentially grew in MDCK cell culture over ECE, this finding may not hold true for all contemporary strains in the U.S. swine herd. Indeed, it has been shown that differences in growth of swine-origin FLUAV strains in ECE and cell culture can be associated with viral strain.6,29 A previous study compared ECE to MDCK cell culture for the isolation of FLUAV strains and found ECE superior to MDCK cell culture. 8 Although the current results contradict those previous findings, it is important to note that the previous study used one H1N1 egg adapted strain, not field strains from original samples. Additionally, all 3 previous studies6,8,29 utilized traditional MDCK cell culture methods rather than the SFM-adapted MDCK cell culture utilized in the present study. Nevertheless, the previous findings led to the suggestion that ECE and cell culture should be used in tandem for the detection of swine-origin FLUAV strains.6,8 However, the financial cost and labor required to conduct virus isolation in ECE is significantly higher than that of MDCK cell culture, which makes conducting blind passage in parallel virus isolation systems unfeasible for large-scale surveillance. The results of the current study, as well as one previous study, 5 show that MDCK cell culture provides better results for primary isolation of swine-origin FLUAV strains than ECE. However, the use of ECE and/or other cell lines should be considered when real-time RT-PCR–positive samples do not yield recoverable FLUAV in MDCK cell culture.
The methods chosen for the detection and/or characterization of FLUAV strains from swine will vary based on viral strains, animal population dynamics, laboratory resources, and goal of the project. The results of the present study support the use of MDCK cells adapted to serum-free culture as the primary isolation method for FLUAV surveillance among swine at agricultural exhibitions. Regardless of what methodology is used, the genetic drift and genomic reassortment of FLUAV strains requires continuous validation of any protocol with contemporary viruses to ensure highest sensitivity and accurate results.
Footnotes
Acknowledgements
The authors wish to acknowledge the support of the personnel at the fairs for participating in this study. Additionally, the authors acknowledge assistance of Dr. Jillian Yarnell.
a.
MDCK cell line canine (catalog no. 84121903), Sigma-Aldrich, St. Louis, MO.
b.
MEM/EBSS, HyClone Laboratories Inc., Logan, UT.
c.
Gibco MEM sodium pyruvate solution (100 mM), Invitrogen Corp., Carlsbad, CA.
d.
Gibco MEM non-essential amino acids solution (10 mM), Invitrogen Corp., Carlsbad, CA.
e.
Gibco fetal bovine serum (certified, heat inactivated), Invitrogen Corp., Carlsbad, CA.
f.
BioWhittaker UltraMDCK serum-free medium (document no. P-SFC 11/09), Lonza Walkersville Inc., Walkersville, MD.
g.
Gibco antibiotic-antimycotic (100×), Invitrogen Corp., Carlsbad, CA.
h.
Flu DETECT, Synbiotics Corp., Kansas City, MO.
i.
PrepEase RNA Spin kit, Affymetrix Inc., Cleveland, OH.
j.
Brilliant III Ultra-Fast QRT-PCR master mix, Agilent Technologies Inc., Santa Clara, CA.
k.
Mx3000P QPCR system, Agilent Technologies Inc., Santa Clara, CA.
l.
QuantiFast Multiplex RT-PCR kit, Qiagen Inc., Valencia, CA.
m.
Vector Laboratories Inc., Burlingame, CA.
n.
EY Laboratories Inc., San Mateo, CA.
o.
R&D Systems Inc., Minneapolis, MN.
p.
CytoFix, BD, San Jose, CA.
q.
FACS Calibur flow cytometer, BD, Franklin Lakes, NJ.
r.
FlowJo, Treestar Inc., Ashland, OR.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been funded in part by Minnesota Center of Excellence for Influenza Research and Surveillance (MCEIRS) with federal funds from the Centers of Excellence for Influenza Research and Surveillance (CEIRS), National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services, under contract no. HHSN266200700007C.
