Abstract
Gamna–Gandy (GG) bodies are non-infectious, hyphal-like structures associated with siderotic nodules in lymphoid tissue; GG bodies are very rarely reported in veterinary cytologic samples. Cytologically, GG bodies can be misidentified as hyphae or plant material. Seven canine lymphoid tissue aspiration cases that contained GG bodies were investigated for morphologic variability and staining characteristics. Available archived cytology slides containing GG bodies were stained with reagents known to show positive results (Prussian blue, Alizarin red S, Von Kossa) and negative results (Gomori methenamine silver) in histologic samples. Calcofluor white staining was also performed. GG bodies in Wright–Giemsa-stained cytology samples displayed considerable variability but were generally 2–5 µm diameter, 10–35 µm long, refractile, clear, pale-tan or pale-yellow, wavy or straight, tubular structures. Six cases allowed for cytochemical staining; staining properties were similar to histology samples. The bodies did not stain with calcofluor white; this stain may be helpful in distinguishing GG bodes from fungal hyphae.
Gamna–Gandy (GG) bodies are named after Charles Gandy and Carlos Gamna, who described them in the early 1900s. 10 The original description of the lesion was associated with pale nodules in the spleen comprised histologically of granulomatous inflammation with multinucleate giant cells, hemosiderin, and “curious, straw-colored, waxy, … spheroid, semilunar, or bamboo-shaped [formations which resembled] mycelian structures.” 17 The term “siderotic nodule” is currently used to describe the larger granulomatous lesions that result from organizing hemorrhage caused by trauma, hypertension, neoplastic processes, or age-related changes.3,6,7,12 In our report, GG bodies refer to the bamboo-shaped structures found within siderotic nodules, which are encrustations of calcium and iron that form around connective tissue fibers; GG bodies can display considerable morphologic overlap with fungal hyphae.2,6,10
Siderotic nodules may be seen incidentally and aspirated for cytologic evaluation during exploratory laparotomy or by advanced imaging modalities including ultrasound, nuclear magnetic resonance, and computed tomography. The correct identification of GG bodies in pathology samples is crucial to prevent misinterpretation of these non-pathologic structures as fungal organisms or plant material.2,10 In histologic sections, GG bodies demonstrate positive reactions with Prussian blue (PB), Alizarin red S (ARS), and Von Kossa (VK) stains because of their mineral content.8,10,12,16 They are also known to produce negative reactions with silver stains commonly used for detection of fungi, such as Gomori or Grocott methenamine silver (GMS).12,16 Calcofluor white (CFW) stain is a fluorescent dye that binds to components found in fungal walls, insect exoskeletons, and plant-based cellulose. 4 The CFW staining characteristics of GG bodies in histologic or cytologic samples have not been investigated, although this stain may be useful to differentiate fungal and plant material from GG bodies.
A single case study has been reported that describes GG bodies in veterinary cytology. 16 We illustrate a series of canine GG body cytology cases from fine-needle aspirates of lymphoid tissues after evaluation of the morphologic variability in GG body structure and the staining properties of GG bodies with PB, ARS, VK, GMS, and CFW on previously Wright–Giemsa (WG)-stained cytology slides.
Clinical history and archived fine-needle aspiration slides submitted for diagnostic cytologic evaluation in 7 cases with GG bodies were collected and reviewed. All cases were initially stained with a methanolic WG stain (5 cases from Colorado State University [CSU; Aerospray, Logan, UT]; 2 cases from University of Florida [EMD Millipore, Billerica, MA]) for cytologic evaluation. GG body size measurements were made using a calibrated digital imaging system (CellSens Entry 1.6, Olympus, Center Valley, PA). Additional cytochemical staining included PB (Polysciences, Warrington, PA), ARS (American MasterTech, Lodi. CA), VK and GMS (Rowley Biochemical, Danvers, MA), and CFW (Sigma-Aldrich, St Louis, MO). Where possible, canine cytologic samples were used for control material for cytochemical staining (Table 1). To contrast the staining pattern between GG bodies and fungal hyphae, WG-stained slides from a canine lymph node with disseminated aspergillosis (the GMS-positive control source) were also stained with PB, VK, and ARS. All cytochemical stains were performed at the CSU Diagnostic Laboratory (Fort Collins, CO) using protocols for previously WG-stained slides, which were based on published and established protocols for canine histologic samples.9,14 Our routine PB and CFW protocols for cytologic samples do not call for de-staining of WG-stained slides. Our routine protocols for GMS, VK, and ARS staining on WG-stained slides includes an initial de-staining step using 1% acid alcohol. Cytochemical stain reactions were interpreted on a scale of negative or slightly, moderately, or strongly positive.
Staining protocols and control material used to identify Gamna–Gandy bodies in previously Wright–Giemsa stained cytologic samples. For indicated protocols, previously Wright–Giemsa-stained slides were de-stained in 1% acid alcohol. Control materials were canine cytologic preparations, unless otherwise noted.

Patient signalment, history, and concurrent clinical findings for each case are presented in Table 2. Ultrasonographic evaluation was performed in cases 3, 5, and 6; recognizable evidence of mineralization was not evident. The samples from spleen had a heterogeneous lymphoid population consisting predominantly of small lymphocytes. Extramedullary hematopoiesis was present in both splenic cases in either the erythroid line (case 3) or the megakaryocytic line (case 5). Incorporating clinical history, the changes present in the splenic aspirates were most consistent with benign age-related changes (siderotic nodules).
Signalment and relevant clinical history of 7 cases with Gamna–Gandy bodies present on cytologic samples.
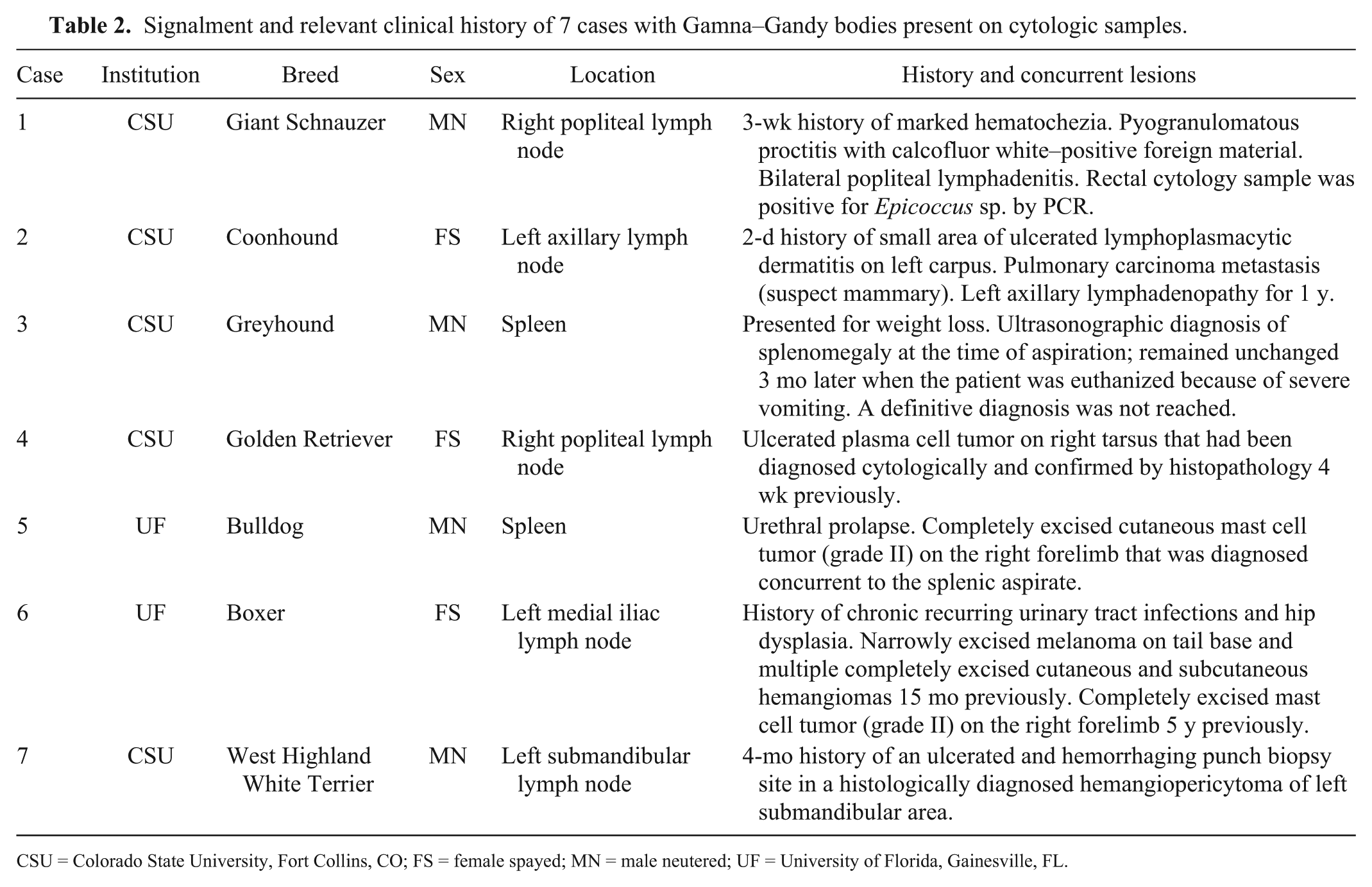
CSU = Colorado State University, Fort Collins, CO; FS = female spayed; MN = male neutered; UF = University of Florida, Gainesville, FL.
All cases demonstrated cytologic evidence of previous hemorrhage including extracellular hemosiderin and hemosiderophages. Hematoidin was found in 2 of 7 cases (cases 4 and 7). Active hemorrhage was present in all 5 cases from lymph nodes, as evidenced by erythrophagia. In contrast to the lymphoid population in the spleen samples, all lymph nodes showed mild-to-moderate degrees of lymphoid reactivity characterized by an increased percentage of plasma cells. Four of the 5 cases from lymph nodes had a neoplastic or ulcerated lesion in the area drained by the node. Case 2 had moderate mastocytosis, which raised concern for mast cell neoplasia that was ruled out with subsequent histologic evaluation.
Figure 1 illustrates the morphologic variability of GG bodies observed in canine lymphoid tissue cytology. The GG bodies were most commonly found associated with aggregates of stroma, although some were observed as free individual filaments in the background. Most GG bodies appeared clear, pale-tan, or pale-yellow, although they occasionally ranged in color from yellow to gold-brown or, in cases 2 and 7, pale blue. Most were refractile and, in all cases, showed no birefringence under polarized light. The GG bodies varied from wavy to serpentine to straight tubular structures, which often branched at variable angles or formed lattice-like aggregates. Most individualized GG bodies were 2–5 µm diameter; although the filaments ranged from 1 µm diameter (case 4) to 19 µm diameter (case 3). Larger aggregates of GG bodies were up to 32 µm diameter (case 3). GG bodies were most often 10–35 µm in length, although short 1-µm fragments (case 5) and larger filaments up to 150 µm long (case 7) were found. They often contained fracture lines resembling septa, spaced at irregular intervals between 8 and 25 µm along the length of the filaments.
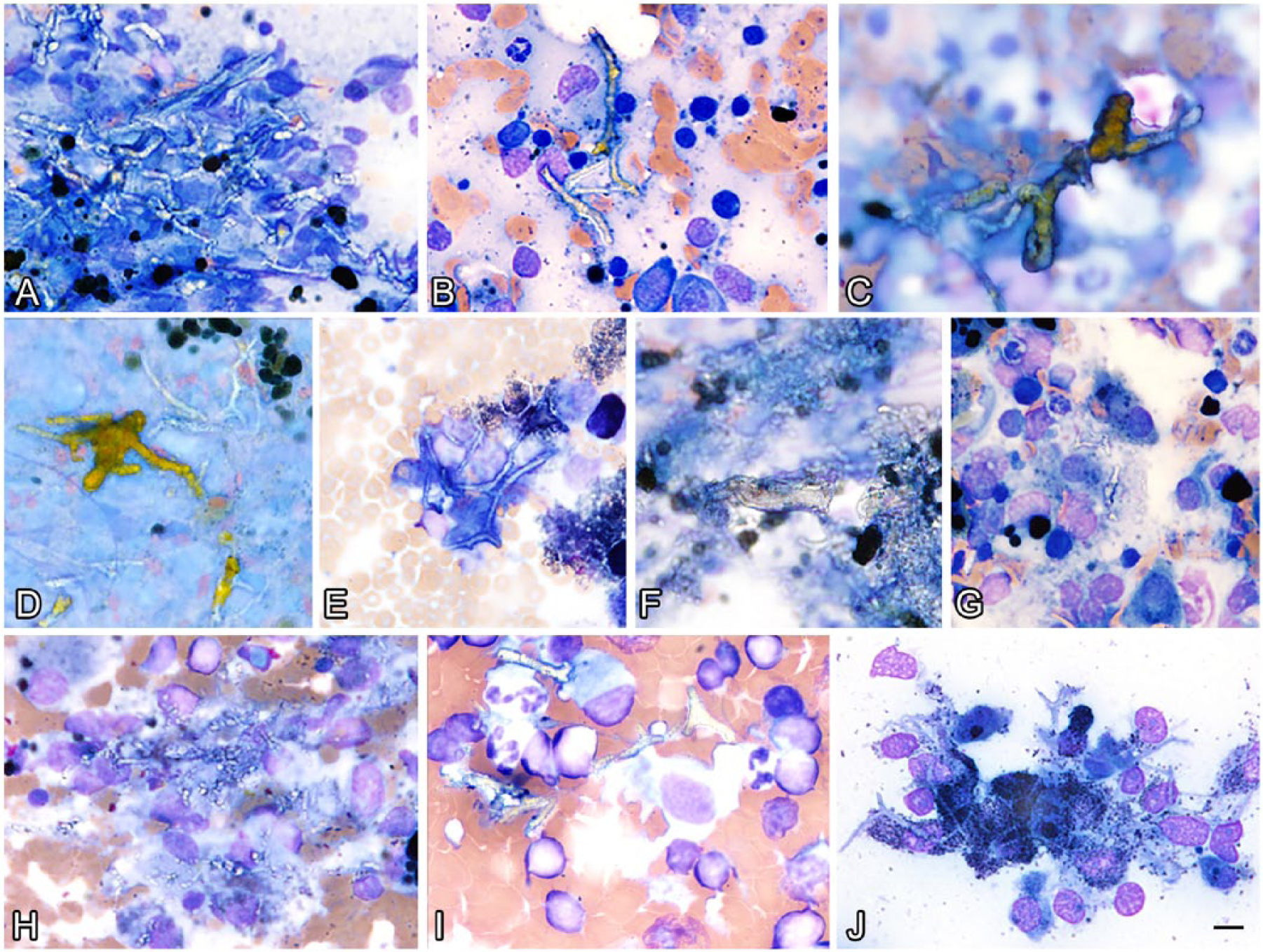
Representative images demonstrating the morphologic similarity and variability of Gamna–Gandy (GG) bodies in cytologic preparations from 7 canine cases.
Cytochemical stains were applied to cases with multiple available slides; there were insufficient slides from case 6 for any additional stains. One slide from case 1 was unstained and was used for VK staining. All stain controls were deemed appropriate. Table 3 details which cytochemical stains were applied on each case and the results of stain intensity of the GG bodies. In the 6 cases stained with ARS, low numbers of GG bodies did not stain positively. Figure 2 provides examples of typical GG body staining with each of the stain protocols.
Cytochemical staining patterns of Gamna–Gandy bodies in fine-needle aspirates from canine lymphoid tissue.

ND = not done; Slight = performed on an unstained slide.
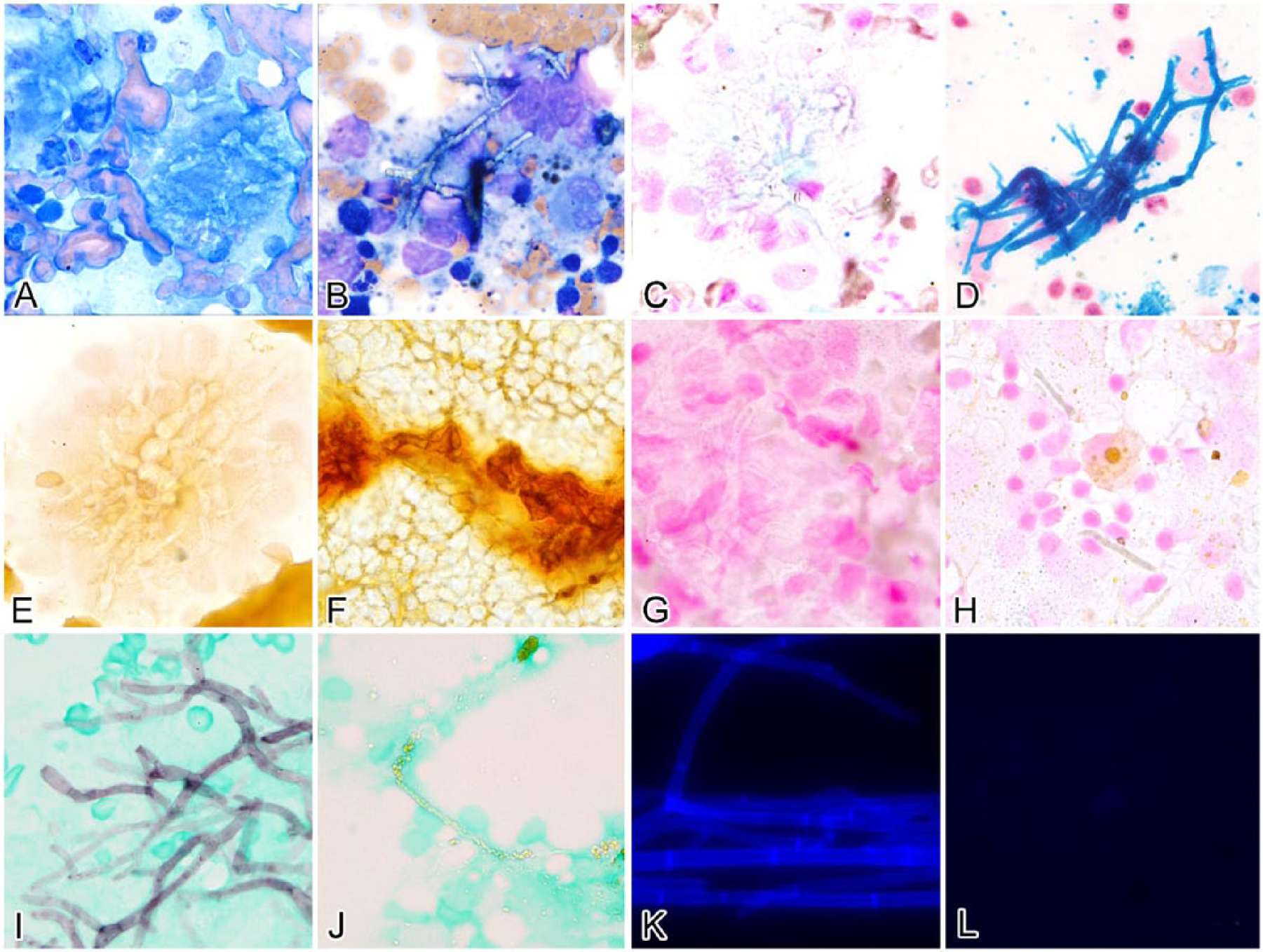
Comparison of staining patterns among cytologic samples with fungal hyphal elements
The PB reaction confirmed the presence of hemosiderin in macrophages in all 6 cases in which this stain was used. Fungal hyphae in the slides of lymph node aspergillosis were consistently negative staining with PB, ARS, and VK (Fig. 2C, 2E, and 2G, respectively) and strongly positive with GMS (Fig. 2I). Positive control cytologic smears of saprophytic fungal hyphae prepared from a fungal culture (the positive control for CFW staining) were strongly positive for CFW (Fig. 2K).
Case 1 is a clinically relevant example of the potential for adverse consequences associated with misdiagnosis of GG bodies as fungal hyphae based on WG-stained morphology alone. A clinical suspicion of fungal proctitis with hyphal-like structures in samples from both the rectal mass and peripheral lymph node could have led to a clinical diagnosis of fungal lymphadenitis and disseminated mycosis. Fortuitously, there were morphologic differences between the hyphae-like structures in both locations that warranted additional staining.
In human medicine, GG bodies have been reported in the spleen (associated with portal hypertension, sickle-cell anemia, and hemochromatosis), lymph node, and ovary, as well as cerebral lesions (hemorrhage and metastatic melanoma) and several types of tumors (cardiac myxoma, thymoma, thyroid neoplasm, and renal carcinoma).6,8,11,12 In veterinary species, GG bodies have been systematically described in the canine spleen and were most commonly found associated with hematomas, hemangiosarcoma, or aging. 3 Experimentally, alcohol injection induces GG body formation in the feline spleen. 5 In our case series, we found concurrent cytologic evidence of GG bodies associated with hemorrhage or increased iron in the aspirates of canine spleen and lymph node. Shared clinical findings included previous biopsy sampling of the location or neoplasia in the region. A common underlying process occurring in all of these cases was tissue disruption with erythrocyte breakdown and associated mineralization.
The cause of the significant morphologic variability demonstrated in the GG bodies in these cases is unknown. Most GG bodies tended to be 2–5 µm diameter wide and were consistent with previous reports. 16 However, we found considerable variability both between cases and between GG bodies within the same case. This variability may reflect the type of collagen involved in GG body formation; previous work has shown that GG bodies are associated with both type 1 and type 3 collagen. 12 Variability of the structures within a case may be useful as a morphologic clue to help distinguish GG bodies cytologically from fungal hyphae using WG staining alone.
Limited evaluation of the use of PB, ARS, VK, and GMS stain over previously WG-stained cytologic slides has been published, some of which include only a single case.13,16 When applied on previously WG-stained slides in these cases, the PB, ARS, and GMS staining patterns were similar to previously unstained histologic sections of these types of structures. The VK results are interesting in that the previously WG-stained slide was negative, whereas the slide that had not been stained with WG contained areas of positive reaction. Reports indicate that GG bodies can be variably positive for VK.8,16 It is uncertain if the GG bodies did not stain positive for VK because of previous WG staining or as a result of inherent variability in the staining of GG bodies themselves; further investigation of the ability to use VK stain over previously WG-stained slides is warranted.
Traditionally, GMS has been recommended to rule out fungi in histologic sections containing GG bodies. Indeed, the 2 GG body cases we tested were GMS negative. However, GMS is not always definitive for fungal structures, given that collagen fibers can stain positive with silver stains. 1 GG bodies can form around collagen fibers, which raises the concern for false-positive staining of GG bodies with GMS. Calcofluor white is a fluorescent dye that displays blue-green fluorescence when bound to β 1-3– or β 1-4–linked polysaccharides and excited with ultraviolet light. These linkages are present in fungal walls, chitin, and cellulose. 4 CFW is commonly used in human medicine. 15 It is a rapid stain procedure (as little as 10 min) that can be applied over previously stained samples. 14 Reportedly, when CFW is used over Papanicolaou-stained human gynecologic samples, aberrant fluorescence of elastin and collagen can occur. 14 Therefore, although GG bodies would be expected to be negative with CFW, it was felt possible that previously WG-stained GG bodies could be positive with CFW. We document the use of CFW on samples containing GG bodies. Although only a low number of cases were evaluated, none of the GG bodies observed stained positive with CFW. In fine-needle aspirates of lymphoid organs, we found CFW to be a rapid staining technique that aided differentiation between positively staining fungal elements and negatively reacting GG bodies.
Ultimately, the most important diagnostic aspect of GG bodies is the recognition that they are not infectious agents. Cytochemical staining can and should be used to help distinguish and assign the correct clinical significance to these clinically insignificant structures.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded in part by the CSU Clinical Pathology Research and Development Funds.
