Abstract
We determined cytologic features of histologically confirmed subcentimeter canine mammary tumors (CMTs) to determine reasonable criteria for an accurate cytologic diagnosis. Fifty-three CMTs from 28 bitches were included. All cytologic samples were collected by ultrasound-guided fine-needle aspiration biopsy, stained with May-Grünwald/Giemsa, and retrospectively evaluated using a scoring system established for 18 cytologic features. Mean nuclear area (MNA) was also measured for each sample by a computer-assisted program. Based on the histologic diagnosis, CMTs were divided into 2 groups: malignant tumors (25) and benign lesions (15). Data were statistically analyzed using Fisher and Mann–Whitney tests. Chromatin pattern (
Canine mammary tumors (CMTs) are the most common neoplasm in sexually intact female dogs. 17 CMTs may be cytologically well-differentiated, showing little cellular pleomorphism, and moderate criteria of malignancy. 2 Cytologic specificity and positive predictive value for malignancy in dogs have been reported to be 55–100% and 93–100%, respectively. However, sensitivity and negative predictive value have been reported to be lower, at 21–96% and 56–93%, respectively. This suggests that cytologic evaluation tends to underdiagnose mammary gland malignancies.1,7,15,16
Furthermore, specific surgical recommendations for subcentimeter CMTs have not yet been well established. Even if 50% or more CMTs are reported to be benign, single or regional mastectomies are chosen to achieve complete surgical margins and to reduce risks of de novo tumor development in mammary tissue. 3 Cytologic diagnosis for small canine mammary nodules instead of incisional or excisional biopsy could be pivotal in refining surgical “dose” and reducing costs for owners. We determined clinically significant cytologic features of histologically confirmed subcentimeter CMTs to determine reasonable criteria for an accurate diagnosis.
Cytologic specimens from subcentimeter canine mammary nodules collected by ultrasound-guided fine-needle aspiration biopsy (FNAB) from client-owned dogs with owners’ consent, from January 2012 to May 2013, were enrolled. Breed, age, spay status, number of tumors per dog, tumor localization, and clinical tumor features were reviewed. Pregnant, lactating, or dogs treated previously with hormonal therapy were excluded. Dogs with tumors >1 cm diameter and dogs with local recurrence or distant metastasis were also excluded.
All slides were stained with May-Grünwald/Giemsa stain and retrospectively evaluated in a double-blinded manner by 2 authors, designated A and B. A third blinded cytopathologist, designated C, examined slides if the 2 cytopathologists were not in agreement for any features. All cytologic smears were evaluated using a scoring system established for 18 cytologic features (Table 1, Fig. 1). The presence or absence of the following cytologic features was specifically recorded: lymphocyte infiltration, necrosis, multiple nuclei, and nuclear molding. The degrees of the presence of extracellular matrix, anisokaryosis, and cellular cohesion, and the presence of spindle cells, macrophage and neutrophil infiltration, and nuclear inclusion were chosen subjectively by the cytopathologists. Cellularity was considered low if <200 cells were counted in 5 low-power fields (10× objective). Cellular groups of at least 8–10 cells were considered as clusters. Clusters were evaluated for dimensional disposition of cells in 2 categories: 1-dimensional if cells were grouped in a single layer, and 3-dimensional if cells overlapped. Angular, pleomorphic, and macronucleoli were considered abnormal nucleoli. Ropy or cordlike chromatin was considered a reticular chromatin pattern. Finely dispersed nuclear chromatin was considered a granular chromatin pattern. The chromatin pattern category was chosen if >50% of cells per 10 high-power fields showed this specific chromatin feature. Necrosis, extracellular matrix, cellularity, cellular cohesion, and clusters were evaluated with 10× and 20× objectives. Mitoses, presence of spindle cells, macrophages, lymphocytes, neutrophils, anisokaryosis, multiple nuclei, and damaged cells were evaluated with a 40× objective. Nuclear edges, nuclear molding, nuclear inclusions, nucleoli, and chromatin pattern were evaluated with a 100× oil immersion objective. For each cytologic feature, a score from 0 to 1 or 0 to 2 was chosen. Cytologic samples with <100 total cells per slide were classified as inadequate.
Scoring system used in the current study for canine mammary tumor cytologic feature evaluation.
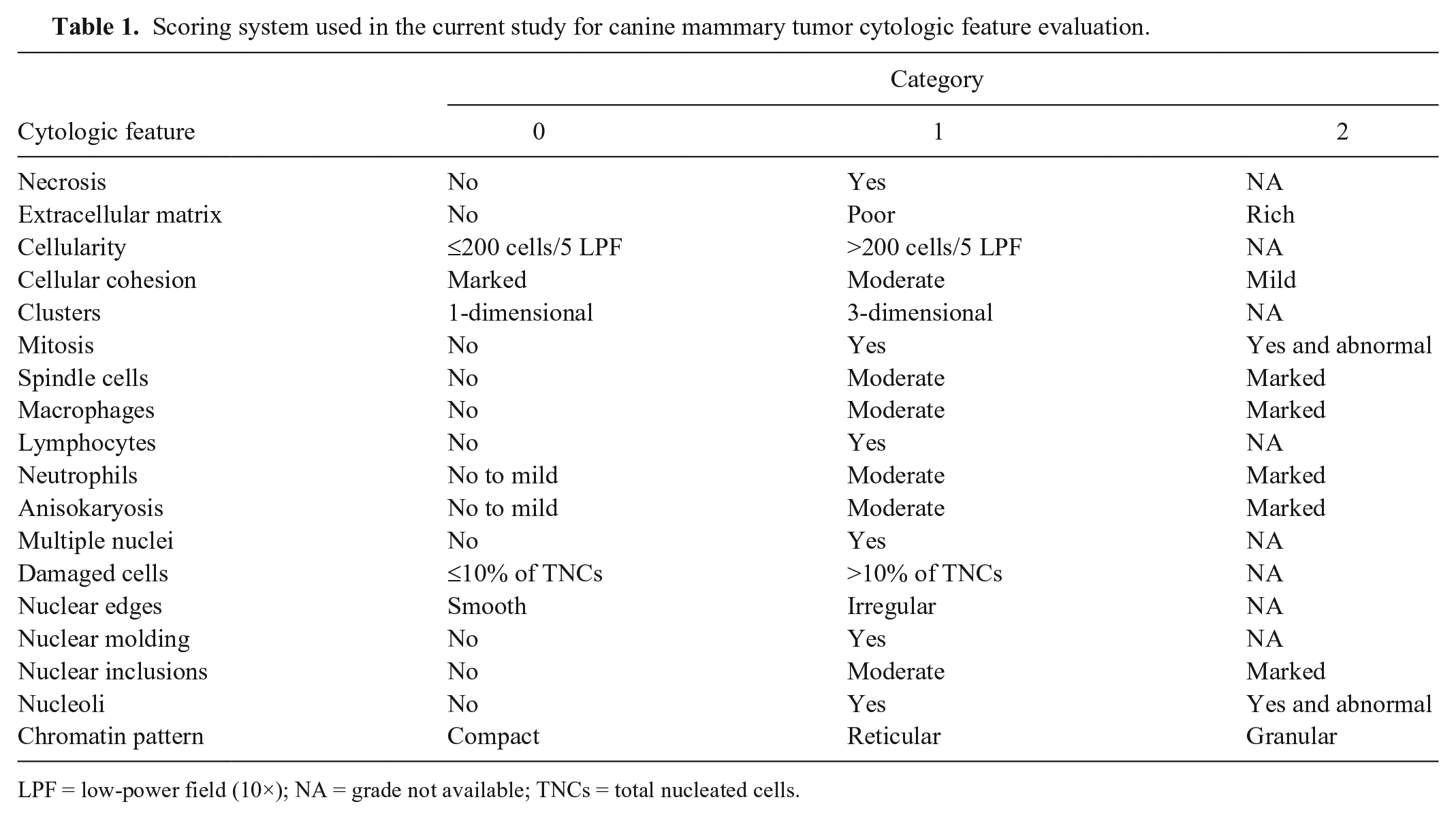
LPF = low-power field (10×); NA = grade not available; TNCs = total nucleated cells.
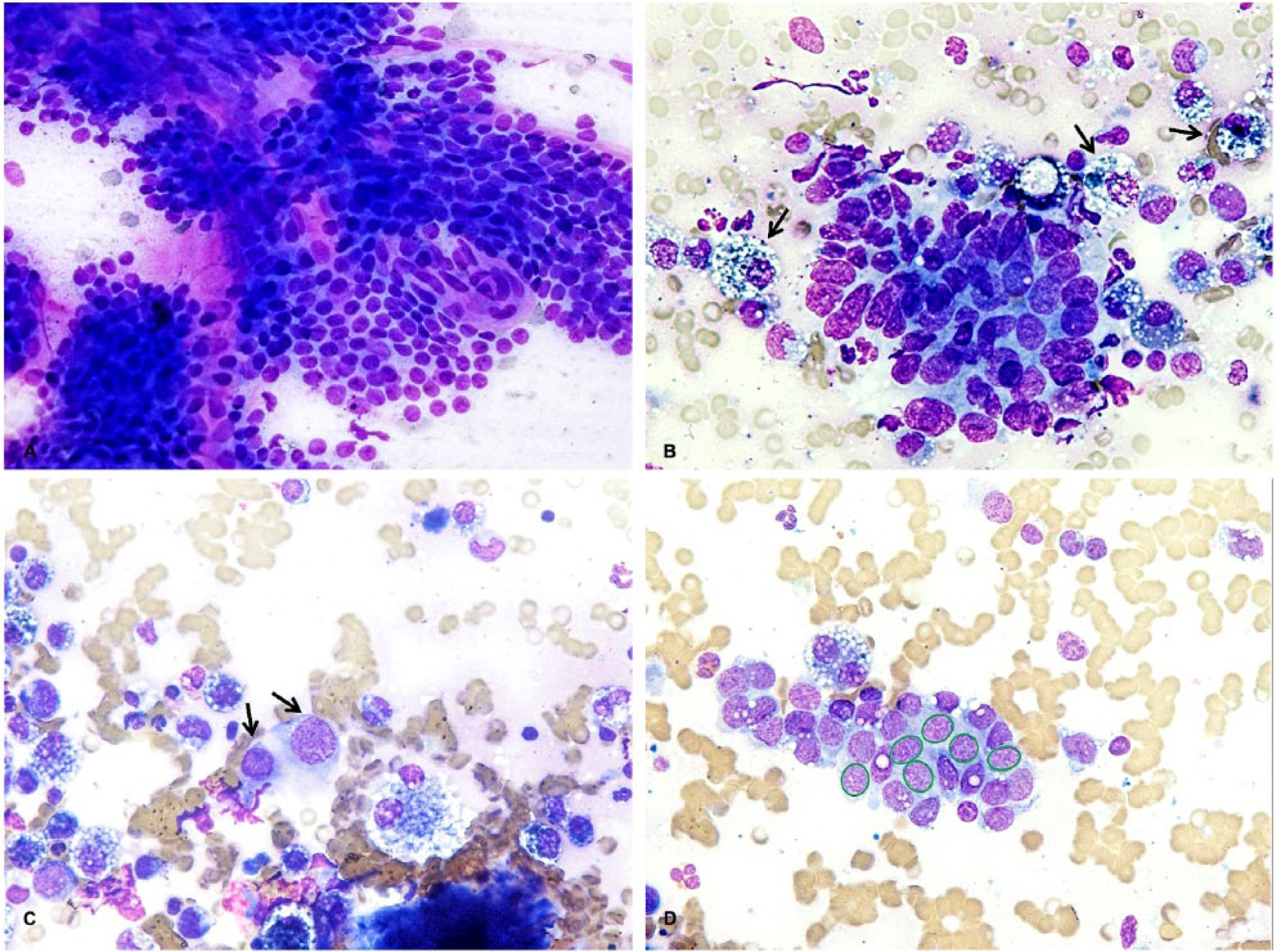
Evaluation of cytologic features of fine-needle aspiration biopsy for diagnosis of canine mammary tumors. May-Grünwald/Giemsa stain.
Mean nuclear area (MNA) for each sample was also measured by a computer-assisted program (NIS-Elements D v.3.1, Nikon, Amstelveen, The Netherlands), as reported previously. 14 Briefly, fields of highest cellularity were selected by one cytologist and examined under 40× magnification. Digital images of these fields were captured as .jpg files for evaluation. From the cells included in these fields, at least 100 intact nuclei were analyzed for clinical relevance.
All excised tumors were routinely processed and embedded in paraffin, and 5-µm thick sections were stained with hematoxylin and eosin. Histologic diagnosis was given according to the World Health Organization classification. 9 Histopathology was performed by a single pathologist. According to the histologic diagnosis, tumors were divided into 2 groups: malignant tumors and benign lesions (benign tumors and non-neoplastic lesions).
The agreement between cytologist A and B was calculated using the Cohen unweighted κ coefficient (http://vassarstats.net). Concordance was described as poor (κ < 0.2), fair (κ = 0.21– 0.40), moderate (κ = 0.41–0.60), substantial (κ = 0.61–0.80), or excellent (κ = 0.81–1.0). A Fisher exact test was performed to assess the relationship between each cytologic feature and the histologic diagnosis and to evaluate inadequate samples compared with previously published data. A Kolmogorov–Smirnov test was used to assess if the MNA values showed Gaussian distribution. Then, an unpaired
Twenty-eight dogs were included in our study. Mean age at the time of surgery was 9 y, with a range of 4–14 y. Dogs by breed were: 3 Poodles, 2 Labrador Retrievers, 2 Dachshunds, 1 Boxer, 1 Yorkshire Terrier, 1 Golden Retriever, 1 German Shepherd Dog, 1 Brie Shepherd, 1 Toy Poodle, 1 Giant Schnauzer, 1 Lagotto Romagnolo, 1 Italian Mastiff, 1 Maltese, 1 Pitbull, and 1 French Bulldog. The other 9 dogs were mixed breed. Of the 28 dogs included in the study, 25 were intact females and 3 were neutered.
The total number of mammary nodules was 53, with 1–4 tumors per dog, and a median of 2 tumors per dog. All nodules had the following clinical features: size <1 cm, mobile, and covered with intact, non-inflamed skin. Of the 53 nodules, 21 involved the caudal abdominal mammary glands, 17 the inguinal glands, 7 the cranial abdominal glands, 7 the caudal thoracic glands, and 1 the cranial thoracic gland.
Thirteen cytologic samples were classified as inadequate by cytologic examination and were not included. All but 1 of these 13 samples were histologically diagnosed as benign lesions; the last was a complex carcinoma. Inadequate samples were associated with benign lesions (
Granular and reticular chromatin pattern (
Relationship between cytologic features and histologic diagnosis in canine mammary tumors and κ agreement between observers A and B for each cytological feature.
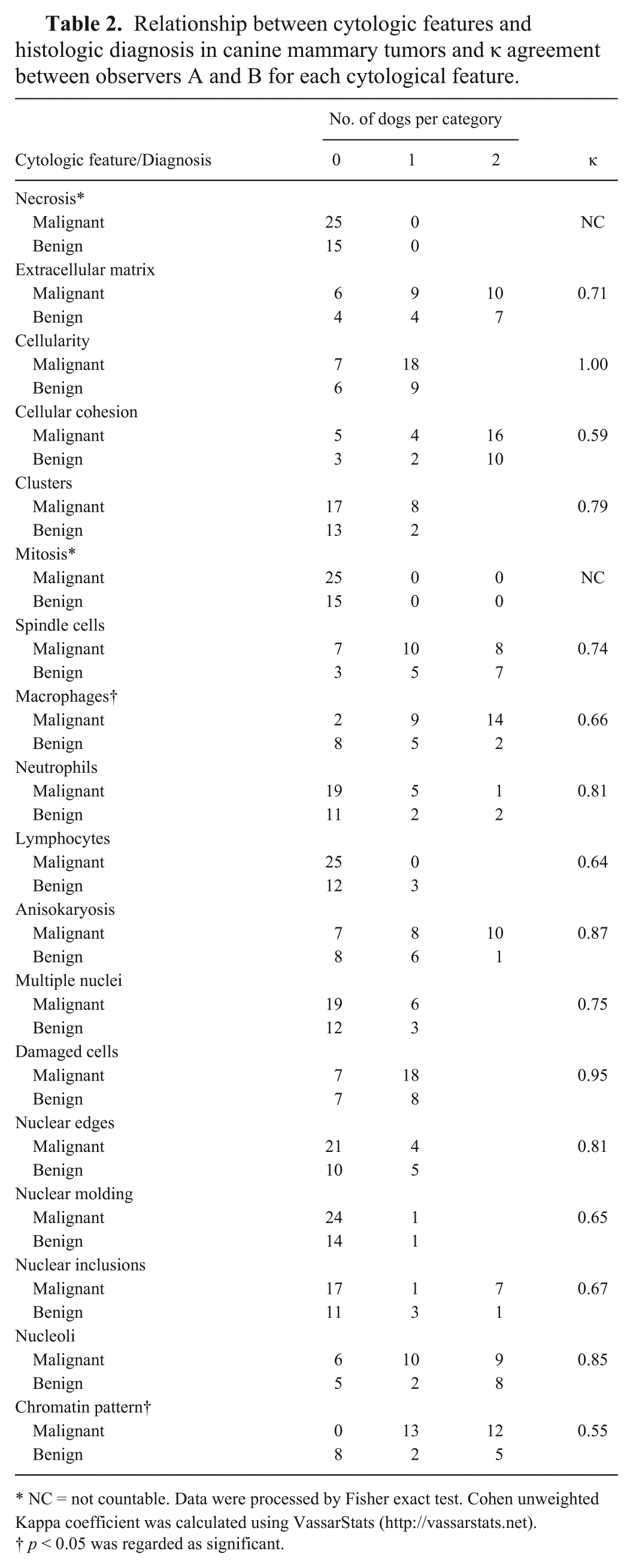
NC = not countable. Data were processed by Fisher exact test. Cohen unweighted Kappa coefficient was calculated using VassarStats (http://vassarstats.net).
MNA values showed a non-normal distribution. Median MNA was calculated as 82.2 µm2 (range: 50.5–93.5 µm2) and as 69.7 µm2 (range: 47.6–85.6 µm2) in malignant tumors and benign lesions, respectively. Using the nonparametric Mann–Whitney U test, greater MNA (
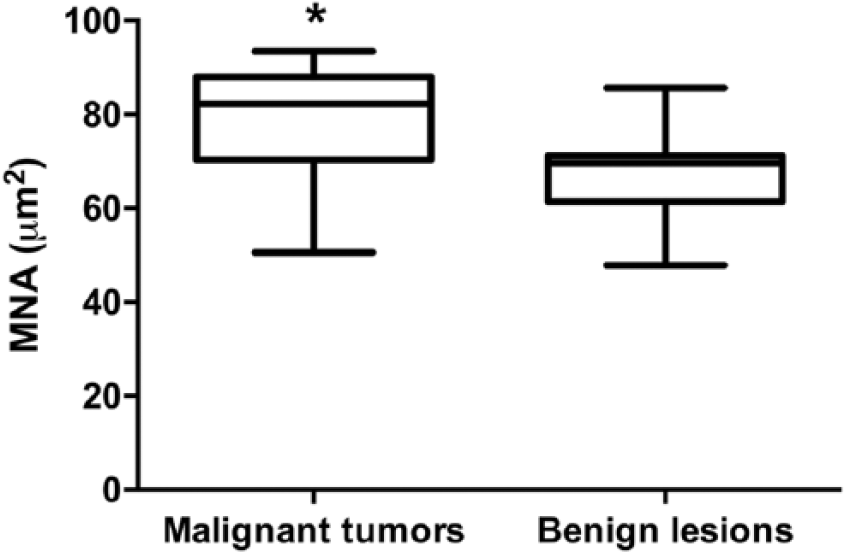
Mean nuclear area (MNA) in malignant tumors and benign lesions. Data were processed by Mann–Whitney U test + Tukey.
Several studies on canine mammary cytology have been reported, but none has been focused on tumor size, to the authors’ knowledge.1,7 Diagnosis of malignant tumors seems to be reliable based on cytologic atypias; however, some well-differentiated carcinomas may be missed.15,16
The chromatin pattern was variable in benign lesions, and only a moderate agreement between operator A and B was found. However, when chromatin patterns were divided into 2 groups, normal (score 0) and altered (score 1 and 2), the agreement improved (κ = 0.89). Normal mammary gland cells have been reported to exhibit uniform dark nuclei and compact chromatin pattern. Dispersed chromatin was considered cytologic atypia, although taken alone, it was not consistent with diagnosis of a malignant tumor, unless other specific cytologic atypias were present.1,7 We never observed a normal pattern in malignancies; the presence of altered chromatin patterns may be useful to diagnose malignancy. In our opinion, cytologist experience could influence inter-operator agreement in evaluation of reticular versus granular chromatin. Despite this, compact chromatin may be assessed more easily and could be useful to exclude malignancy.
We found an association between malignant tumors and macrophage infiltration. During the last decade, tumor microenvironment has been the topic of numerous studies. Several authors have reported a strong association between tumor-related inflammation and cancer progression and invasiveness. 6 In particular, tumor-associated macrophages (TAMs) seem to be able to promote cancer progression by producing growth and pro-angiogenic factors and by reducing physiological immune-mediated response. 12 In dogs, TAMs have been associated with canine malignant mammary tumors. Of 50 canine mammary adenocarcinomas, a significantly increased TAM number was related to the ability of cancer to metastasize. 8 In another study of 59 CMTs, significantly increased TAM values were observed in malignant versus benign lesions. 10 Furthermore, higher TAM values were associated with lower overall survival times. In a 2015 study, TAMs were associated with higher vascular endothelial growth factor expression, suggesting that TAMs may influence angiogenesis in malignant mammary tumors in dogs. 11 Macrophage cytologic evaluation may be useful to identify malignant mammary tumors even if cytologically well-differentiated.
We found that malignant tumors have a statistically significant large MNA. During the last decade, several studies have focused on assessing cytologic MNA in dogs with cancer.13,14 In accordance with our findings, larger MNA was reported to be associated with malignant mammary tumors in previous studies. 13 Moreover, MNA was reported to be predictive of lymph node metastasis. 5 Although MNA is not easy to perform and is not yet cost effective, preoperative cytologic MNA assessment in small mammary nodules may be useful and may play a role in decision-making in CMTs with doubtful cytology.
In our study, 13 of 53 samples had <100 cells per slide and were considered non-diagnostic. This finding is in agreement with previous studies.15,16 However, in these previous studies, the authors did not report the incidence of inadequate samples compared to tumor size. Furthermore, ultrasonography was not used to perform FNAB. For these reasons, it is not easy to make a direct comparison between results reported by these studies. There are a number of reasons for non-representative samples in cytologic examination. First, sampling small nodules can be a challenge even if done by ultrasonography. Second, non-neoplastic lesions and benign tumors tend to exfoliate fewer cells than malignant tumors. Third, some benign or malignant tumors could be cystic, resulting in smears of scattered cells, making interpretation difficult. Moreover, inadequate samples were associated with benign lesions (12 of 13), and the aforementioned reasons may corroborate this finding.
There are some limitations in our study. First, there is no non–ultrasound-guided FNAB control group for better assessment of ultrasound-guided sampling impact on diagnostic accuracy. Second, the nuclear feature evaluation may be biased by May-Grünwald/Giemsa staining. Papanicolaou-type stain remains the traditional and preferred stain for the study of nuclear features; nevertheless, this stain is not used commonly in veterinary practice. 4 Finally, the low number of cases might influence statistical results.
Although histologic examination is the gold standard for the diagnosis of CMTs, cytologic observation of chromatin pattern, macrophage infiltration, and MNA may help cytopathologists make a definitive diagnosis. Further prospective studies are necessary to validate data reported in our study.
Footnotes
Acknowledgements
We thank Blaise Burke for his writing assistance and contribution to this paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
