Abstract
Little is known about the potential consequences of pancreatic tissue sampling in dogs. The goal of the present study was to evaluate changes in serum trypsin–like immunoreactivity and canine-specific pancreatic lipase after pancreatic fine-needle aspiration and surgical biopsy in 27 clinically healthy dogs. Presurgical, ultrasound-guided aspiration of the pancreas was performed with the dogs under sedation. Subsequently, all the dogs underwent intraoperative pancreatic fine-needle aspiration and clamshell biopsy. After euthanasia, pancreata were sectioned for histopathologic evaluation. Serum pancreatic enzyme levels were measured at 3 time points: baseline, after ultrasound-guided aspiration, and after intraoperative aspiration and biopsy. No significant differences were detected among mean serum pancreatic lipase values at any point (P > 0.05). Serum trypsin–like immunoreactivity did not change from baseline (18.2 ± 2.1 μg/dl; mean ± standard error) after ultrasound-guided aspiration (13.6 ± 2.2 μg/dl) but increased significantly after intraoperative sampling (44.8 ± 1.9 μg/dl; P < 0.0005). After surgical biopsy, the 20 dogs that had both ultrasound-guided and intraoperative sampling had a higher mean (SE) serum trypsin–like immunoreactivity (44.8 ± 1.9 μg/dl) than the 7 dogs that had only intraoperative samples taken (36.4 ± 4.1 μg/dl; P < 0.05). All 27 pancreata were grossly normal before intraoperative sampling. Pancreatic sampling was associated with increased serum trypsin–like immunoreactivity and mild, peracute necrosis, inflammation, hemorrhage, and fibrin deposition. Tissue damage from sampling was not sufficient to cause an elevation in canine-specific pancreatic lipase in the time frame evaluated. Further studies are needed to determine longer-term effects of pancreatic sampling on enzyme levels and clinical outcome.
Introduction
The diagnosis of pancreatic disease is challenging because of the nonspecific nature of presenting clinical signs and the lack of highly sensitive and specific diagnostic tests. A variety of diagnostic methods have been used to attempt detection of canine pancreatitis, including serum amylase, lipase, trypsin-like immunoreactivity (TLI), radiography, and ultrasonography, with sensitivities of 62%, 54–73%, 33–36%, 24%, and 68%, respectively (Mansfield CS, Jones BR: 2000, Trypsinogen activation peptide in the diagnosis of canine pancreatitis. J Vet Intern Med 14:346. Abstract; Steiner JM, Gumminger SR, Rutz GM, Williams DA: 2001, Serum canine pancreatic lipase immunoreactivity (cPLI) concentrations in dogs with exocrine pancreatic insufficiency (EPI). J Vet Intern Med 15:274. Abstract). 4,8 More recently, a canine-specific pancreatic lipase immunoreactivity (cPLI) assay has undergone analytic validation. 9 There is some evidence that the cPLI has a high sensitivity for the detection of pancreatitis (82–93%), but specificity is preliminarily reported to be lower, at 78% (Steiner JM, et al.: 2001, Serum canine pancreatic lipase immunoreactivity; McCord K, Davis J, Leyva F, et al.: 2009, A multi-institutional study evaluating diagnostic utility of Spec cPL™ in the diagnosis of acute pancreatitis in dogs. J Vet Intern Med 23:734. Abstract). Furthermore, histopathologic lesions of pancreatitis in dogs can be focal and randomly distributed, making even the reference standard, a biopsy diagnosis, problematic. 5,7
Additional, minimally invasive tools for the evaluation of pancreatic disease are still needed. Fine-needle aspiration (FNA) of the pancreas and laparoscopic biopsy may prove to be useful, but little is currently known about the utility, safety, or complications of these techniques. The use of pancreatic FNA as a diagnostic tool has been reported in dogs only a few times in the veterinary literature for the detection of pancreatic pseudocysts and exocrine pancreatic neoplasia. 1,10 No clinical complications were noted in 1 retrospective study of cats and dogs undergoing laparoscopic pancreatic biopsies. 12 The single study to date that reported the effect of biopsy sampling on pancreatic enzyme levels documented a transient increase in the TLI in 1 of 3 dogs, although cPLI was not evaluated. 3 Therefore, additional information is needed regarding the utility and consequences of pancreatic tissue sampling in dogs.
The objectives of the current study were 3-fold. First, the authors aimed to evaluate changes in serum pancreatic enzyme levels (TLI and cPLI) after pancreatic FNA and biopsy in clinically healthy dogs. The second objective was to characterize the cytologic yield by using 3 sampling techniques in these dogs. The third objective was to compare cytologic and histopathologic findings. The following hypotheses were proposed: 1) serum pancreatic enzyme levels would not change in association with FNA but may be increased after surgical biopsy, 2) adequately cellular cytology samples could be obtained by using ultrasound-guided FNA, intraoperative FNA, and biopsy-obtained impression smears in healthy dogs, and 3) cytologic and histopathologic findings would correlate well in healthy dogs.
Materials and methods
Animals
Twenty-seven adult Beagle dogs were included in the study. The dogs were purchased by the University of Minnesota (Saint Paul, Minnesota) for use in a terminal surgery laboratory for veterinary students. The dogs were housed at the Veterinary Medical Center, and all in vivo components of the study were performed on a single day with University of Minnesota Institutional Animal Care and Use Committee approval.
Health evaluation and blood sampling
A baseline physical examination and routine screening tests (complete blood cell count [CBC] with blood film evaluation, serum biochemistry profile, and urinalysis) were performed on each dog at the start of the study just before sedation. Blood samples from each dog for serum TLI and cPLI a were obtained at baseline, after ultrasound-guided FNA, and after intraoperative FNA and biopsy. All dogs were fasted for at least 12 hr before sampling.
Ultrasound-guided FNA
Before ultrasound-guided FNA of the pancreas, the dogs were sedated intravenously (IV) with acepromazine b (0.02 mg/kg) and butorphanol c (0.3 mg/kg). The pancreas was visualized by a single radiology technician with a high-quality ultrasound machine. d In dogs in which the pancreas was visualized and within reach for aspiration, the abdomen was shaved and aseptically prepared by using chlorhexidine surgical scrub and alcohol. By using a 22-gauge 7.6-cm (3-inch) spinal needle, e a preattached 6-ml syringe, f and a needle guide, g aspirates were obtained from the right pancreatic limb by the same board-certified internist (PJA). One milliliter of air was drawn into the syringe before insertion, and the needle–syringe combination was moved vertically along one plane within the pancreas. At least 2 samples were obtained from each pancreas, 1 with and 1 without negative pressure applied with the syringe. Lastly, cellular material was expelled and gently spread onto clean glass slides. The samples were allowed to air-dry for subsequent routine cytologic staining with modified Wright–Giemsa stain. h The dogs were monitored for comfort and changes in rectal temperature, heart rate, and respiratory rate after the ultrasound-guided aspirates.
Intraoperative and necropsy sampling
The dogs were premedicated with atropine i (0.02–0.04 mg/kg IV) and, if needed, additional acepromazine (0.01–0.05 mg/kg IV). General anesthesia was induced with thiopental j (15 mg/kg) and maintained with oxygen and isoflurane inhalant anesthetic. k Intravenous crystalloid support was also provided with the dogs under anesthesia. Laparotomy was performed by supervised veterinary students by using routine surgical approach and technique. During surgery, pancreata were photographed and grossly inspected for evidence of damage or trauma related to ultrasound-guided aspiration. The FNA samples were then obtained from the right pancreatic limb by using the same size and gauge needles, syringes, and technique as described above. A clamshell biopsy was obtained by using a biopsy cup forceps l at the site immediately adjacent to the intraoperative FNA. Touch impressions for cytologic evaluation were made from the clamshell biopsy samples before they were placed in formalin m for histopathology. The combination of both intraoperative FNA and clamshell biopsy is hereafter defined as intraoperative sampling. Before or after obtaining pancreatic tissue samples, several other routine surgical procedures were performed on each dog, which involved intra-abdominal organs other than the pancreas as part of a teaching exercise, before humane euthanasia with IV pentobarbital overdose. Students were specifically instructed not to manipulate the pancreas during surgery. At postmortem, each pancreas was excised and sectioned at 2-cm intervals for complete histopathologic evaluation.
Sample evaluation
All cytology slides (ultrasound-guided aspirates, intraoperative aspirates, and touch impressions) were evaluated by a single clinical pathologist (LCS). Cytologic yield was determined and calculated as the number of cases with diagnostic quality samples divided by total number of cases. Histopathologic lesions in biopsy samples and entire pancreata were evaluated and scored by a single histopathologist (SJN), according to a previously published protocol. 7 In brief, this pancreatic histopathology scoring protocol describes 8 lesion types (neutrophilic inflammation, lymphocytic inflammation, pancreatic necrosis, pancreatic fat necrosis, edema, fibrosis, atrophy, and hyperplastic nodules). The presence or absence, severity (mild, moderate, severe), and frequency of these 8 lesions in each pancreas are described. 7 The cytologist and histopathologist were masked to all other data.
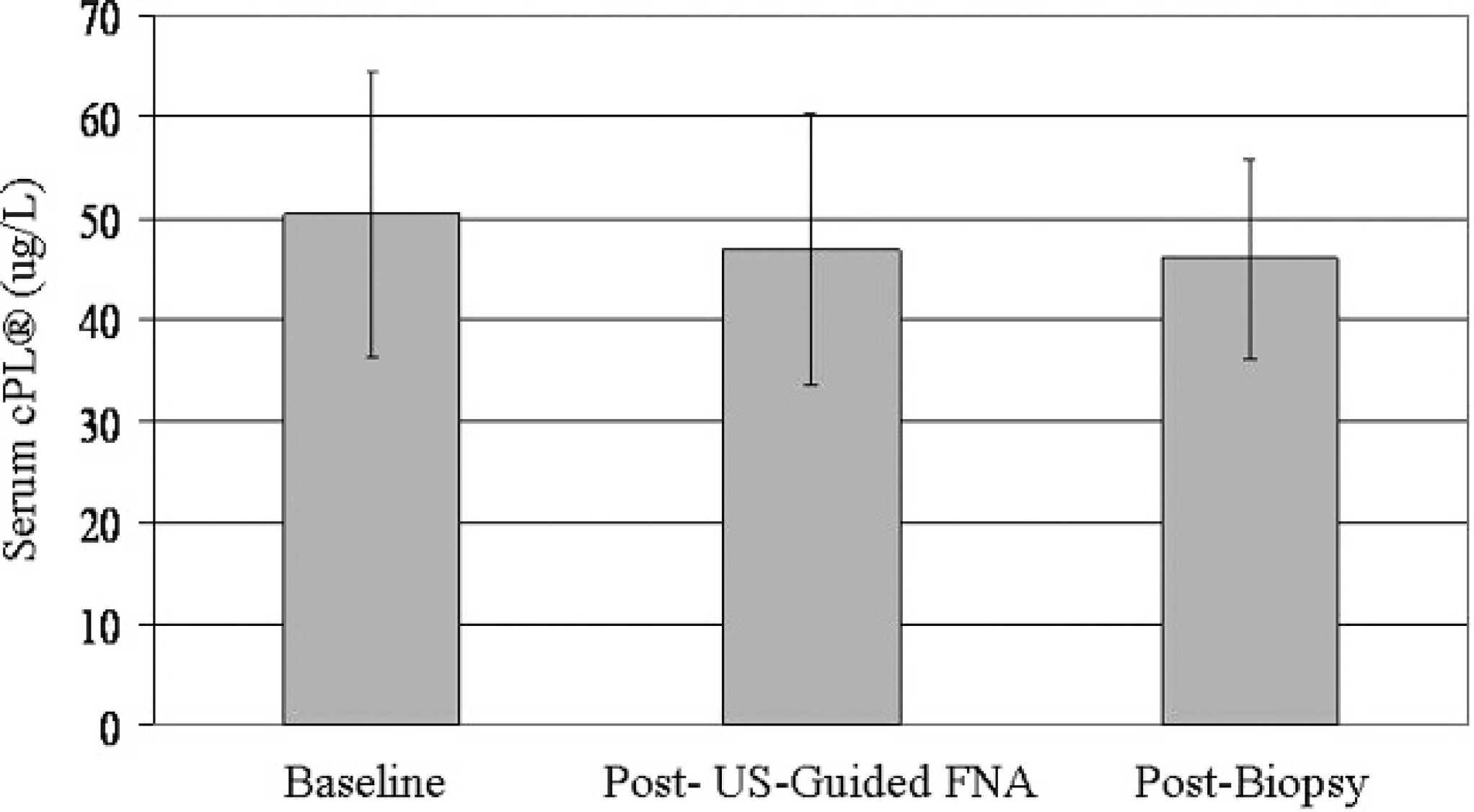
Serum canine–specific pancreatic lipase immunoreactivity (cPLI) assay a levels: values ≤200 μg/l are within the normal range, and values ≥400 μg/l are considered to be consistent with pancreatitis. There were no significant differences between serum cPLI levels among the 3 time points (P > 0.05).
Statistical analysis
Descriptive statistics of numerical data are reported as median and range. Paired sample t-tests were used to compare changes in serum pancreatic enzyme levels over time. Spearman correlations were used to assess relationships among age, pancreatic enzyme levels, and the 8 histopathologic lesions. Statistical calculations were performed by using a commercially available software package, n with the significance level set at P < 0.05 for t-tests and P < 0.01 for the correlations, given the large number of correlations performed.
Results
Twenty-seven Beagle dogs with a median age of 15 months (range: 11–33 months) were studied (12 intact males, 1 neutered male, 14 intact females). All the dogs had a body condition score of 4 or 5 on a 9-point scale, and median body weight was 10.5 kg (range: 7.9–16.2 kg). The results of the baseline physical examination, CBC, serum biochemistry profile, and urinalysis tests documented a clinically healthy population. The pancreas was accessible for FNA in 20 of 27 dogs. Of the 7 dogs in which aspirates were not obtained, 5 had a visible pancreas but no clear window for aspiration, and, of the 2 remaining dogs, the pancreas was not visible ultrasonographically. During surgery, pancreatic FNA and clamshell biopsy specimens were obtained from all the dogs.
Clinically evident acute complications of ultrasound-guided FNA were not observed on physical examination before general anesthesia for the second part of the study. Vital parameters remained within normal limits for sedated individuals in 24 of 27 dogs. One of these 24 dogs had a prolonged effect of sedation but was hemodynamically stable, with no free fluid in the abdomen. The 3 remaining dogs did not have post-aspirate vital parameters recorded.
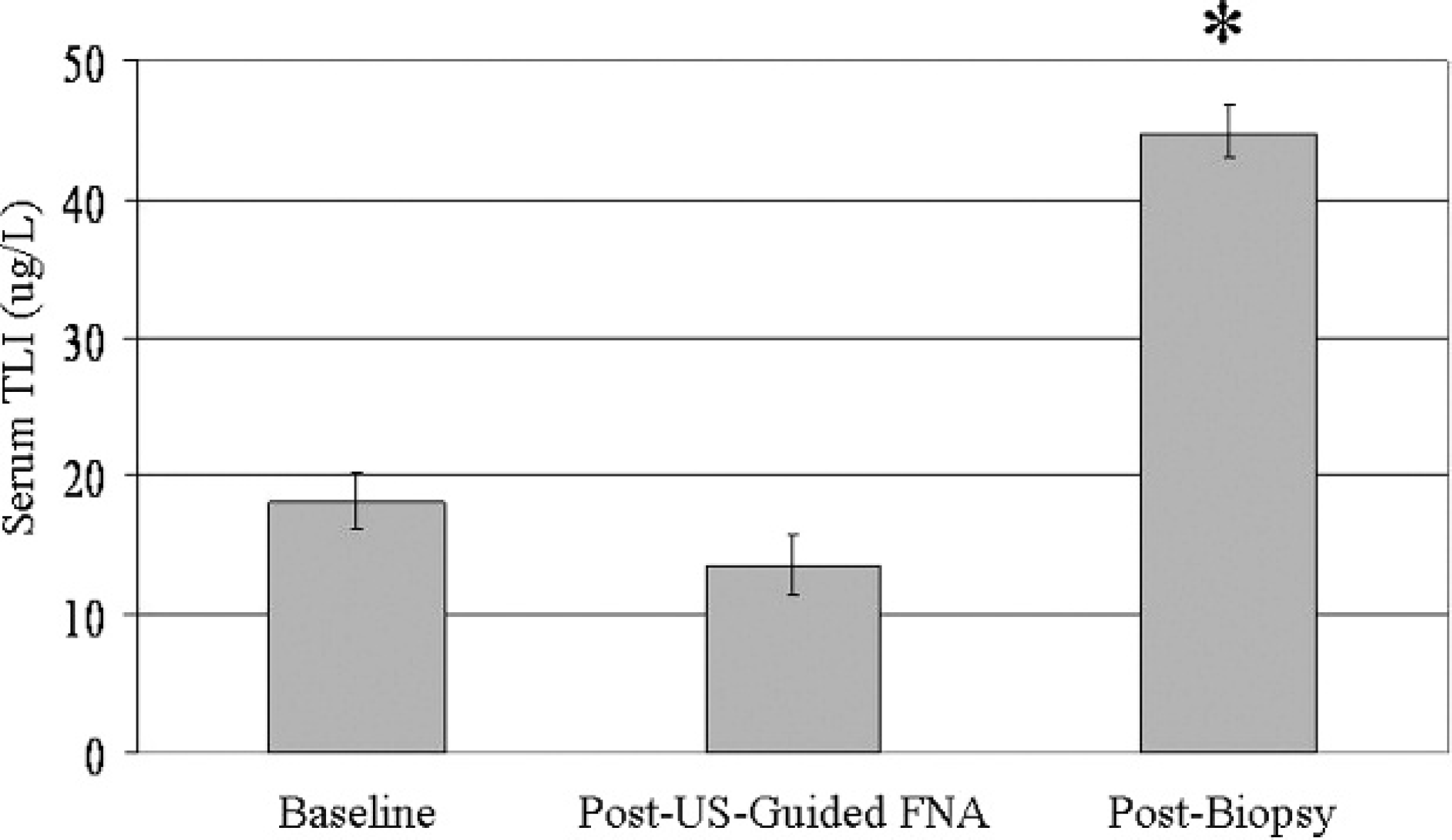
Serum trypsin–like immunoreactivity (TLI) levels. All the dogs had a normal serum TLI (<35 μg/l) at baseline, and ultrasound-guided fine-needle aspirate (FNA) did not affect the TLI value (P > 0.05). In contrast, serum TLI levels increased significantly after concurrent intraoperative FNA and biopsy (P < 0.0005).
Enzyme levels
Serum TLI and cPLI a values were measured at baseline, 181 min (range: 121–251 min) after ultrasound-guided FNA, and 71 min (range: 9–185 min) after intraoperative FNA and biopsy. The lower limit of the cPLI assay is <30 μg/l, and the upper limit of the TLI assay is >50 μg/l. Values obtained outside of these limits were recorded for statistical analysis as an absolute value of 30 μg/l or 50 μg/l, respectively. None of the dogs had a cPLI level that was suggestive of pancreatitis at any time point during the study, with all values being well below 200 μg/l (Fig. 1). Furthermore, in the 20 dogs in which ultrasound-guided FNA was feasible, there were no differences among cPLI levels at any of the 3 sampling time points (P > 0.05; Fig. 1). In these 20 dogs, serum TLI did not change from baseline (18.2 ± 2.1 μg/dl; mean ± standard error) after ultrasound-guided FNA (13.6 ± 2.2 μg/dl) but increased significantly after intraoperative sampling (44.8 ± 1.9 μg/dl; P < 0.0005; Fig. 2). After surgical biopsy, the 20 dogs that had both ultrasound-guided FNA and intraoperative sampling had a higher mean (SE) serum TLI (44.8 ± 1.9 μg/dl) than the 7 dogs that had only intraoperative samples taken (36.4 ± 4.1 μg/dl; P < 0.05; Fig. 3).
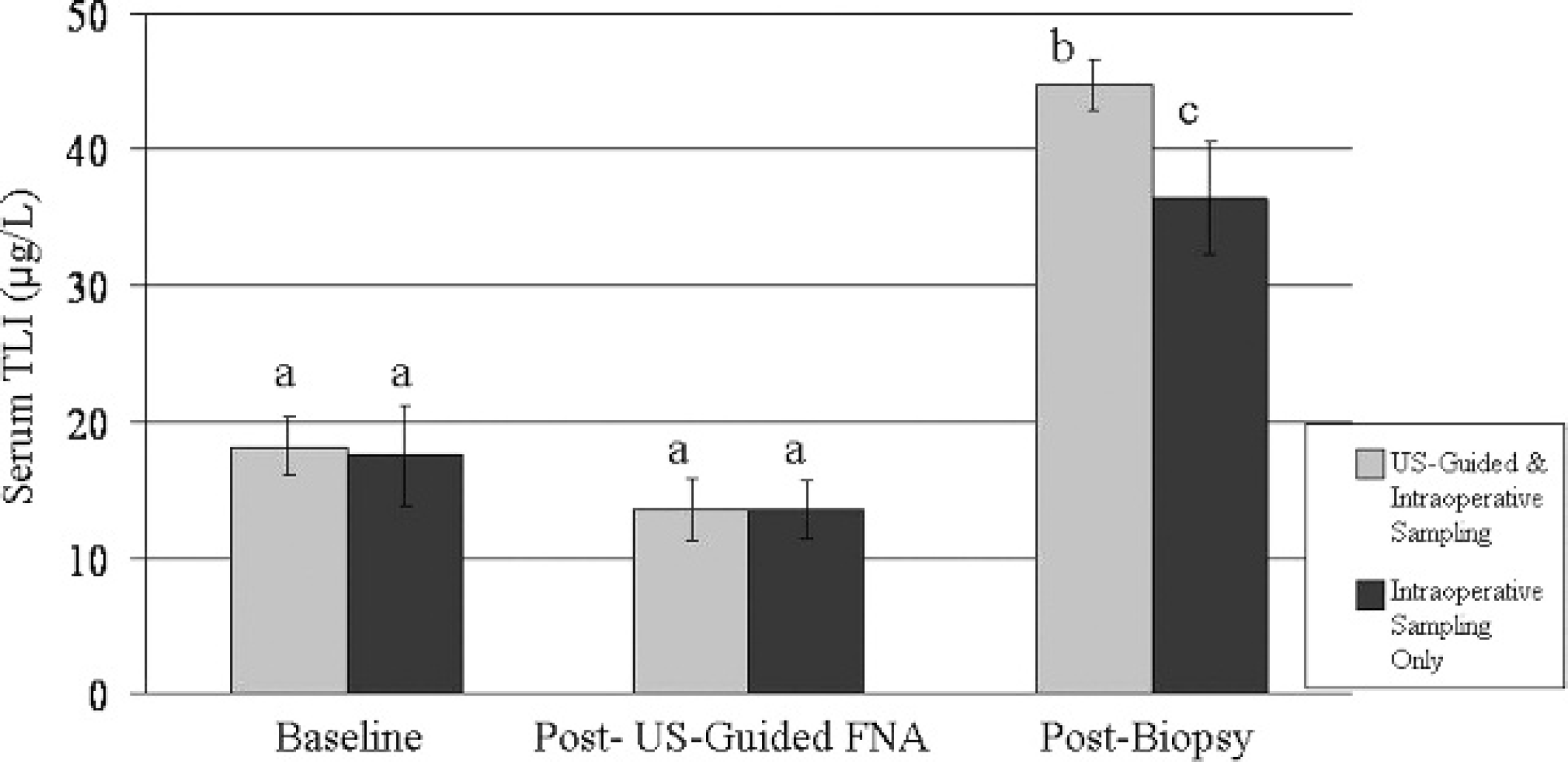
Serum trypsin–like immunoreactivity (TLI) increased with sampling. After intraoperative fine-needle aspirate (FNA) and biopsy, the group that had previously had ultrasound-guided FNA had a higher mean serum TLI than the group that had only intraoperative sampling performed (P < 0.05). Columns that do not have a common superscript letter differ significantly.
Cytologic yield
Cytologic yield varied, depending on the technique used to acquire the samples. Although some pancreatic cells were seen on many slides, 0 of 20 ultrasound-guided aspirates had sufficient cellularity to be of diagnostic quality. Intraoperative aspirates (18/27 [67%]) and touch impressions from biopsy samples (21/27 [78%]) were more often of diagnostic quality to evaluate cellular characteristics.
Histopathology
During surgery, all 27 pancreata appeared grossly normal, with no evidence of trauma from ultrasound-guided FNA. Histopathologic evaluation revealed normal pancreatic tissue in the majority (19/27) of clamshell biopsy samples. The remaining 8 biopsy samples had at least 1 lesion present. In order of decreasing frequency, the lesions in the biopsy samples were pancreatic necrosis, hyperplastic nodules, edema and inflammation (neutrophilic and/or lymphocytic), and fibrosis and atrophy (Table 1). Necropsy specimens contained more lesions than biopsy samples (Table 1). Two additional lesions were also detected in the necropsy samples that were distinct from the lesion types described in the previously published histopathology protocol. 7 These additional lesions were hemorrhage and fibrin deposition (Table 1). Overall, the majority of pancreatic tissue in the study dogs was free of histopathologic lesions. All pancreata had at least 1 lesion present from the grading protocol, but the mean frequency scores were low. Less than 1–11% of all tissue were affected by each lesion. Lastly, all lesions were graded as mild, except 1 dog that had moderate nodular hyperplasia.
Because the majority of the pancreatic biopsy specimens and cytology samples were normal, there were not enough lesions to explore correlations between these techniques. There were no significant correlations among age, pancreatic enzyme levels, or specific histopathologic lesions (P > 0.05).
Histopathology findings (N = 27). *
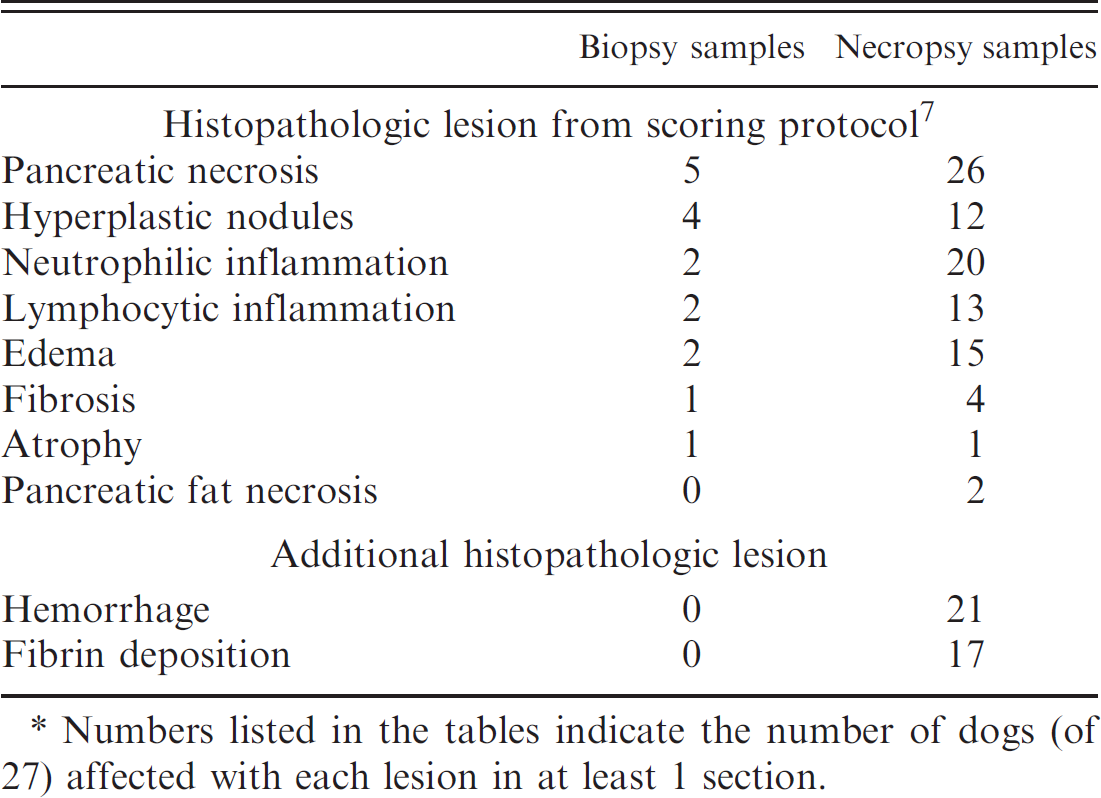
Numbers listed in the tables indicate the number of dogs (of 27) affected with each lesion in at least 1 section.
Discussion
The results of the present study suggest that pancreatic aspiration and biopsy can cause an increase in serum TLI levels and sporadic, mild, focal peracute pancreatic necrosis, inflammation, hemorrhage, and fibrin deposition. The tissue damage from ultrasound-guided FNA and intraoperative sampling, however, was mild enough to avoid a short-term rise in cPLI. a
Cytosolic enzymes can be released from exocrine pancreatic epithelial cells after trauma, and intra-organ enzyme release can result in inflammation, as seen in cases of acute pancreatitis. 2 The absence of significant increases in serum levels of pancreatic enzymes after ultrasound-guided pancreatic aspiration suggests that the trauma to the pancreas during this procedure is minimal. Intraoperative sampling, however, was associated with a change in the TLI. The fact that the dogs that had both ultrasound-guided and intraoperative sampling had a higher TLI than those that had only intraoperative sampling suggests that disruption to the pancreatic tissue from these procedures was additive. It is unclear why the TLI but not the cPLI a was affected by the procedures in the current study. The TLI may be more sensitive than cPLI at detecting acute insult or may be more abundant in acinar cells. Alternatively, it is possible that general anesthesia, associated changes in pancreatic perfusion or blood pressure (not monitored in the present study), or other student manipulations in surgery differentially affected the TLI. It has been suggested that the half-life is rather short (approximately 1–2 hr), so it is unlikely that changes in cPLI were missed with the sampling interval that was used (J. M. Steiner, personal communication, 2009).
In the current study, the yield of diagnostic quality cytology slides varied, depending on the technique used to acquire the sample. The slides from ultrasound-guided samples were relatively acellular, despite clear ultrasonographic visualization of the needle penetrating the pancreatic capsule and entering the parenchyma. There was no cytologic evidence of other tissue, such as mesenteric fat, being inadvertently aspirated. From these results, it was concluded that, when using this technique, ultrasound-guided aspiration of the normal canine pancreas is unlikely to yield an adequate sample for cytologic interpretation. Normal pancreatic tissue has a tightly associated fibrovascular network 2 that may diminish exfoliation of cells from normal pancreata more so than in diseased organs with disrupted tissue architecture. Operator and/or technique dependency in the collection of samples may also be a factor in determining yield.
Histopathology lesions in the current study were mild and, although present in each pancreas, were noted in a small percentage of the overall organ. Except for biopsy-related lesions, histopathology findings confirmed essentially normal pancreatic tissue, which make the samples from the present study acceptable for characterization of normal canine pancreatic cytology. Necropsy specimens showed more lesions than biopsy samples, presumably because of trauma from the biopsy procedure and the ability to evaluate the entire organ at postmortem. The types of lesions that were present were similar to those known to be present in dogs randomly presented for postmortem. 5,11 Nodular hyperplasia was found in fewer samples than has been previously reported, 6 and this finding is likely because of the young age of the dogs in the present study. The extent of necrosis, hemorrhage, and fibrin present in the necropsy samples adjacent to the biopsy sites was striking and likely represents an acute change induced by biopsy sampling that has not been previously documented.
There were several limitations to the current study. First, the ultrasound-guided aspirates were performed by an internist with limited experience with performing this type of aspiration. If cytologic yield is affected by experience level with ultrasound-guided aspirates of the pancreas, then the yield may have been better had a board-certified radiologist performed the procedure, because that is the typical process at the University of Minnesota Veterinary Medical Center. Secondly, because of the constraints of timing imposed by the student surgical laboratory, there was only a brief interval between pancreatic sampling procedures and euthanasia. The short sampling interval prevented serial evaluation of changes in pancreatic enzyme levels and/or the development of other complications over a longer postprocedure period. Thirdly, manipulations other than pancreatic sampling were performed during surgery, including ovariohysterectomy, jejunal resection and anastomosis, and cystotomy. Although students were instructed not to handle the pancreas during surgery, it is possible that these other manipulations could have influenced the results. Lastly, because the dogs in the current study were young adults, there were very few naturally occurring lesions, which made it difficult to investigate correlations between age and histopathology or between histopathology and cytology.
Overall, pancreatic sampling by FNA or surgical biopsy appeared not to cause significant damage to the pancreas as reflected by stable cPLI a levels within the reference range. More studies are needed to determine longer-term effects of pancreatic sampling on enzyme levels and clinical outcome. High-quality cytologic samples of normal pancreas can be obtained by intraoperative FNA and biopsy but not necessarily with ultrasound guidance.
Acknowledgements
The authors thank IDEXX Laboratories for performing all laboratory work and pancreatic tissue sectioning and preparation for histopathology. The authors would also like to thank Dick Hermes, CVT, for his assistance in obtaining ultrasound-guided fine-needle aspirates from the dogs in this study and Drs. Nathan Rose and Duane Robinson for assistance with blood sample acquisition.
Footnotes
a.
Spec cPL®, IDEXX Laboratories Inc., Westbrook, ME.
b.
Acepromazine maleate 10 mg/ml, Vedco Inc., St. Joseph, MO.
c.
Butorphanol tartrate 10 mg/ml, Fort Dodge Animal Health, Fort Dodge, IA.
d.
Vivid 7, GE Healthcare, Little Chalfont, Buckinghamshire, United Kingdom.
e.
Disposable 22 g × 9 cm spinal needles, Jorgensen Laboratories, Loveland, CO.
f.
Monoject 6 ml syringes, Kendall/Covidien, Mansfield, MA.
g.
Ultra-Pro II Needle Guidance System, CIVCO Medical Solutions, Kalona, IA.
h.
Modified Wright Giemsa, Wescor Inc., Logan, UT.
i.
Atropine sulfate 1/120 grain, Vedco Inc., St. Joseph, MO.
j.
Pentothal, thiopental sodium 1 g vial, Hospira Inc., Forest Lake, IL.
k.
IsoFlo®, Abbott Animal Health, Abbott Laboratories, Abbott Park, IL.
l.
Laparoscopy biopsy cup forceps 66321DZ, KARL STORZ Veterinary Endoscopy America Inc., Goleta, CA.
m.
10% phosphate buffered formalin, Mallinckrodt Baker Inc., Phillipsburg, NJ.
n.
SPSS 13.3, SPSS Inc., Chicago, IL.
