Abstract
Visceral toxicosis of catfish (VTC) syndrome was recognized in the late 1990s and recently has been associated with exposure to Clostridium botulinum type E neurotoxin. Tentative diagnosis is based on clinical presentation and gross findings, and is confirmed by bioassay. In April 2009, channel catfish (Ictalurus punctatus) from 2 different farms presented with abnormal swimming behavior and mortalities. Nine fish were submitted to the Aquatic Research and Diagnostic Laboratory (Stoneville, Mississippi) for evaluation. Bacterial cultures from these fish were negative. Necropsy findings included intestinal intussusceptions, ascites, pale proximal intestines with engorged serosal blood vessels, splenic congestion, and a reticular pattern to the liver. Significant histopathologic findings were limited to cerebral, splenic, and hepatic congestion, splenic lymphoid depletion and perivascular edema, vascular dilation and edema of the gastrointestinal tract, and perivascular edema in the anterior and posterior kidneys. Intoxication from C. botulinum type E neurotoxin was suspected based on the clinical signs and lack of gross and microbiological evidence of an infectious disease process. The toxicosis was confirmed with a positive bioassay using serum collected from the submitted fish.
Visceral toxicosis of catfish (VTC) was first recognized in the spring of 1998 when channel catfish (Ictalurus punctatus) producers in the southeastern United States began experiencing acute mass mortalities in food or brood-sized fish (Avery J, Khoo L, Steeby J, et al. 1999, Early spring losses of foodfish and broodfish. National Warmwater Aquaculture Center News 2(1):1). This syndrome occurs primarily in the spring and fall when water temperatures are below 20°C. Initial clinical signs include abnormal behavior with fish porpoising or swimming in large schools. As the disease progresses, fish are found listless in close proximity to the pond bank, and sometimes display muscular fasciculations or quivering barbels when handled. Occasionally, there is eversion of the stomach into the oral cavity and exophthalmia. Internal lesions are chylous abdominal ascites, intestinal intussusceptions, pale proximal intestines with prominent blood vessels, splenic congestion, and a reticular pattern to the liver. 12 Tentative diagnosis is based on clinical presentation and gross findings and is confirmed using a catfish bioassay and mass spectrometric-based endopeptidase methodology. 5
In early investigations, an infectious cause for the mortalities was placed lower on the list of differentials when bacterial and viral cultures on different cell lines were negative. A toxin was suspected as the causative agent based on the following preliminary unpublished investigations. Placing sentinel fish in cages in affected ponds gave equivocal results, and injecting fish with centrifuged, concentrated pond water did not cause mortalities or lesions. Gavaging test fish with minced whole affected fish did not reproduce similar clinical signs or symptoms. However, injecting fingerling catfish with serum collected from affected fish reproduced the syndrome usually within 24 hr, and filtered (0.2 µm) or ultraviolet-irradiated serum was still toxic. Sequential passage of serum collected from fish used in bioassays showed a diminution of clinical signs indicating a dilution of the affecting agent. Based on these findings, a toxin was suspected. Size fractionation experiments utilizing centrifugal devices a as well as high-performance liquid chromatography revealed that the active fraction was approximately 30–50 kDa. It was also shown that toxin was heat labile as its activity was lost when serum was heated to 56°C for 30 min. Although botulism was suspected, efforts to confirm this using the conventional mouse bioassay were unsuccessful. In 2007, a channel catfish serum neutralization bioassay was paired with endopep mass spectrometry to confirm that botulinum type E exposure is associated with this toxicosis. 5 The current report describes clinical signs, and gross and histopathological lesions from 2 cases of VTC confirmed via catfish bioassay.
In the same week of April 2009, market-size channel catfish (Ictalurus punctatus) from 2 different farms in the Mississippi River Delta presented with abnormal swimming behavior and significant mortalities. Four fish from farm X and 5 fish from farm Y were submitted to the Aquatic Research and Diagnostic Laboratory at the Thad Cochran National Warmwater Aquaculture Center (Stoneville, Mississippi). Gill biopsies were performed and examined by light microscopy for evidence of parasites or bacterial infection. Blood was collected from vessels at the level of the caudal peduncle, allowed to clot in sterile tubes a with no additives at 4°C, and centrifuged at approximately 1,000 × g for 10 min at 4°C. Fish were then euthanized utilizing tricaine methanesulfonate b (250 mg/L). Serum was collected and placed in sterile 15-ml polypropylene tubes and stored at 4°C. Bioassays were performed by injecting 0.25 ml of the serum intraperitoneally into fingerling channel catfish (approximately 10–20 g) utilizing a 1-ml syringe with a 27-guage (0.406-mm) needle. The fish were monitored at least once a day for the next 3 days for clinical signs. Brain and posterior kidney were subjected to bacterial culture on blood agar plates incubated at 27 ± 2°C. Plates were checked daily for bacterial growth for 4 days. Internal gross lesions were noted, and samples of gill, brain, muscle, skin, spleen, heart, gastrointestinal tract, liver, and anterior and posterior kidney were collected and fixed in 10% neutral buffered formalin solution. Tissues were fixed for at least 24 hr, processed routinely, embedded in paraffin, sectioned at approximately 4 µm, stained with hematoxylin and eosin or periodic acid–Schiff stain, and examined using a microscope equipped with a digital camera and image analysis software. c In addition, archived tissues from VTC-suspected cases in 1999 from Arkansas were also examined. d
Gill biopsies from farm X contained moderate numbers of the protozoan parasites Ambiphyra sp., Trichodina sp., and Henneguya sp., while only Trichodina sp. were present in fish from farm Y. Serum from all 4 fish from farm X produced positive bioassay results within 24 hr. Only serum from fish no. 3 from farm Y resulted in a positive bioassay 48 hr post-injection. No bacterial pathogens were evident in the cultures from all 9 fish.
Gross lesions are summarized in Table 1. Only fish no. 1 from farm X had all 5 lesions typically associated with VTC, which included intestinal intussusceptions, ascites, pale proximal intestines with engorged blood vessels, splenic congestion spleen, and a reticular pattern to the liver (Figs. 1, 2). All fish from farm X had at least 2 lesions while some in farm Y had only 1 lesion. All fish from both farms had empty stomachs or scant amount of algae except for fish no. 4 from farm Y that had semi-digested fish parts and bones in the stomach.
Summary of gross internal lesions in suspect visceral toxicosis of catfish–affected channel catfish (Ictalurus punctatus) from 2 different farms.*
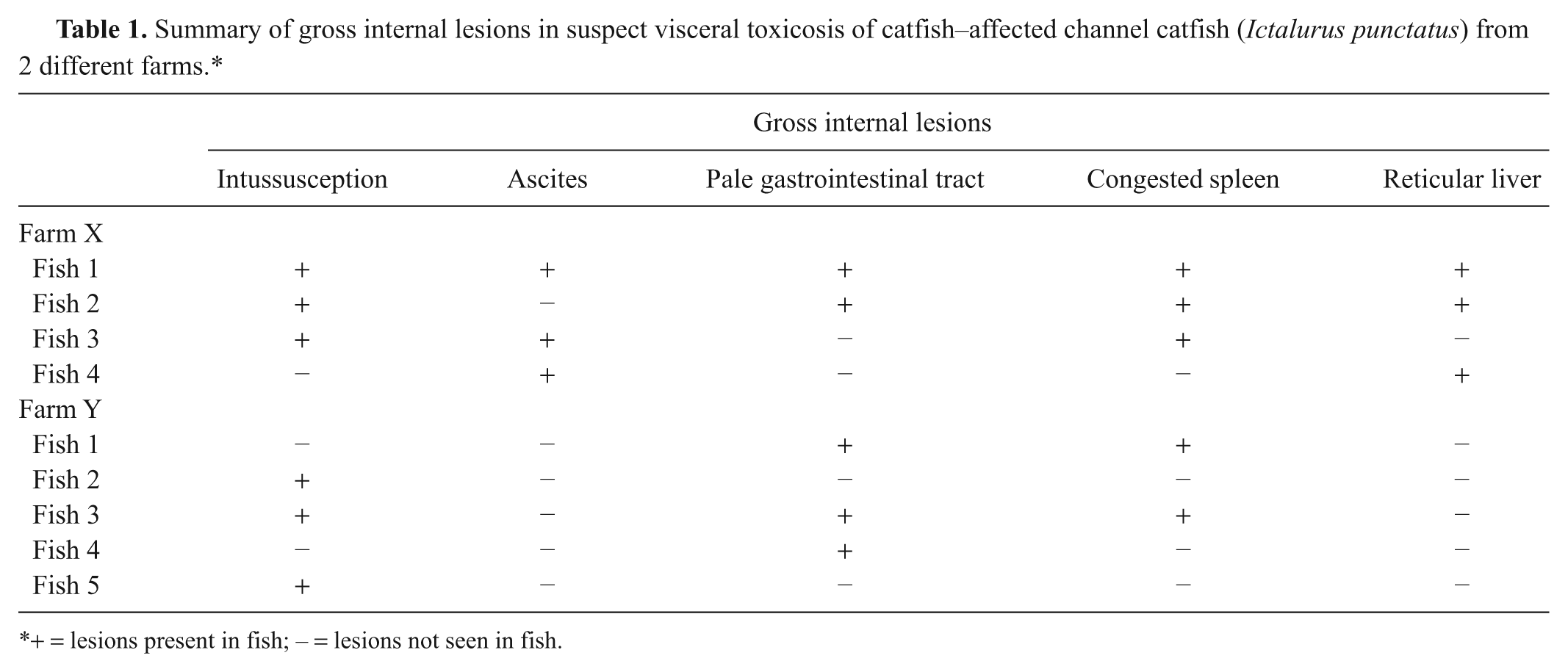
+ = lesions present in fish; – = lesions not seen in fish.
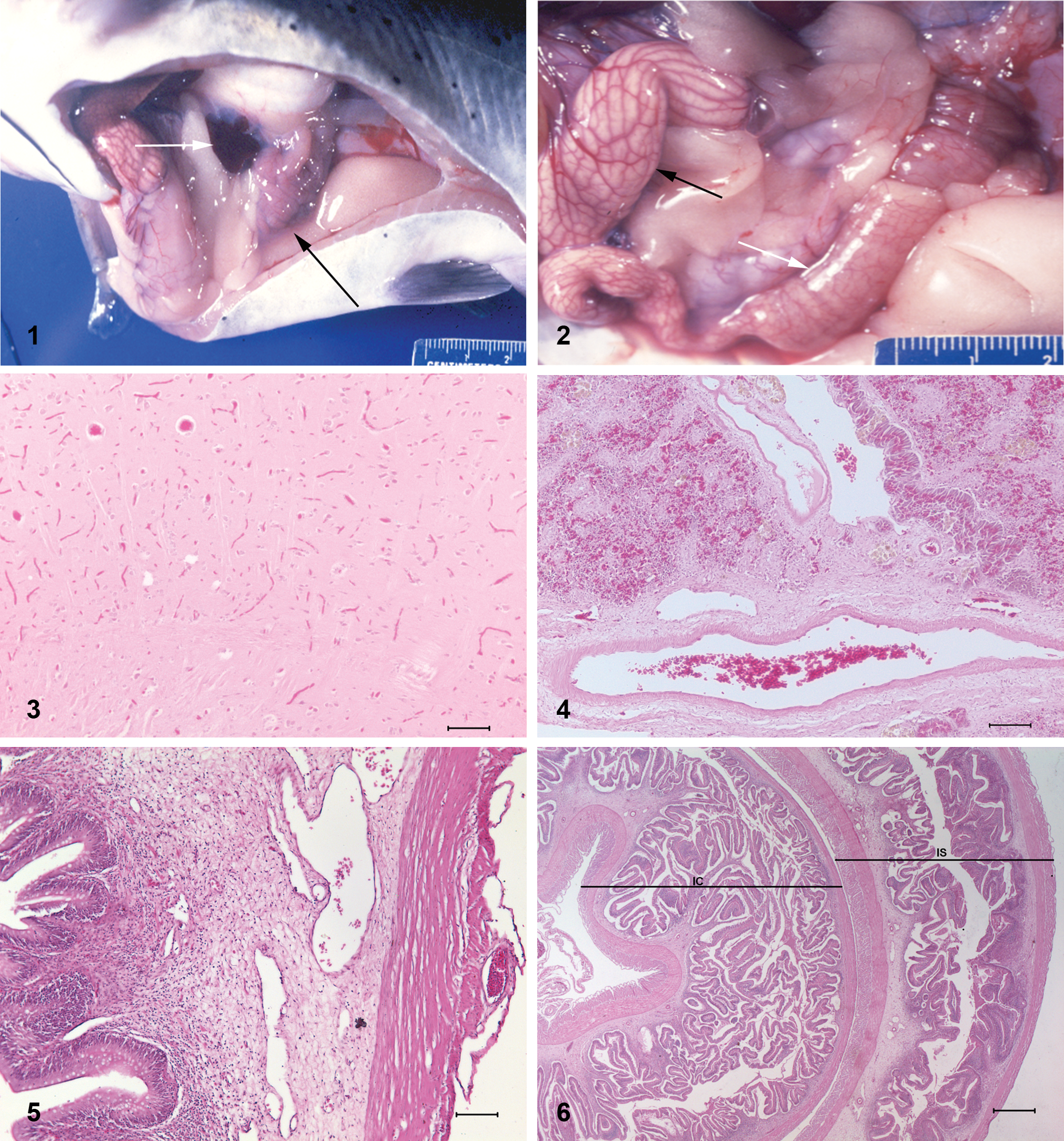
Typical gross internal lesions in a visceral toxicosis of catfish–affected channel catfish (Ictalurus punctatus). Black arrow indicates the chylous ascites; white arrow indicates a mildly congested spleen.
Significant histopathological lesions were confined to the brain, liver, spleen, gastrointestinal tract, and anterior and posterior kidney. The majority of the cerebral blood vessels were congested (Fig. 3). Hepatic sinusoidal vessels, especially near the capsular surface, were mildly congested. Most hepatocytes had moderate, microvesicular vacuolation. There was mild to moderate splenic congestion and lymphoid depletion with increased prominence of the reticular framework. In addition, there was pallor around several arteries with separation of the connective tissue and smooth muscle fibers (edema; Fig. 4). Splenic mesothelial cells were occasionally hypertrophic with a “tombstone” effect. Changes in the proximal intestinal tract were pallor of the lamina propria with separation of the cellular elements by clear spaces (edema) as well as marked dilation of blood vessels within the lamina propria. There was also dilation of vessels on the serosal surface that were devoid of blood (lymphatics presumptive) as well as a few smaller congested blood vessels on the serosal surface (Fig. 5). There were low numbers of mononuclear cells within the lamina propria and submucosa. In sections through the intussusception, there was submucosal edema, especially within the intussuscipiens with little or no inflammation or necrosis of the intussusceptum (Fig. 6). Similar edematous changes were present in the stomach with expansion of and separation of the cells within the lamina propria. Gastric serosal vessels were dilated and congested. In the posterior kidney, there was mild to moderate separation of smooth muscle fibers of small arteries as well as the connective tissue and smooth muscle fibers of the larger collecting ducts and the opisthonephric ducts. Similar but less severe changes were also present in arteries of the anterior kidney. There were high numbers of pigmented macrophage centers in the spleen and the posterior kidney with fewer in the liver and anterior kidney.
Clostridium botulinum is a Gram-positive, anaerobic bacterium with 7 serotypes based on toxin production. 13 Botulism caused by C. botulinum neurotoxin type E (BoNT/E) in cultured salmonids has been reported in the United States, Britain, and Denmark.2,3,7 In Europe, the disease was originally referred to as “bankruptcy disease” because of its devastating effects. Since the recognition of VTC as a disease entity in the late 1990s, it has occurred annually with variable prevalence (Khoo, L, Gaunt P, Mauel M: 2008, 2007 Aquatic Diagnostic Laboratory Summary Report. National Warmwater Aquaculture Center News 10:6–7). It is a particularly devastating disease as it usually affects the larger more valuable fish. Unfortunately, there are no current treatments or management strategies for VTC as the epidemiology of this disease in channel catfish has yet to be fully understood.
Reports of atypical behavior in VTC-affected catfish are not unusual. Rainbow trout (Oncorhynchus mykiss), walleye (Sander vitreus), and yellow perch (Perca flavescens) experimentally exposed to BoNT/E swam in a head up/tail down orientation and breached the surface of the water, 13 a behavior also observed in channel catfish. In the experimental studies, changes were attributed to the inhibition of cholinergic mechanisms controlling the swim bladder and/or the motor control of the fins used to stabilize and maintain postural balance. It was hypothesized that gobies (members of the Gobiidae family) did not display this behavior as they lacked a swim bladder. Hyperpigmentation was observed in some of the species tested, and it was proposed that because chromatophores are under nervous control, acetylcholine either caused melanophore dispersion (resulting in darkening) or aggregation (resulting in lightening). 13 Although not always a consistent finding, VTC-affected catfish often appear darker than normal but this was not noted in the catfish described herein.
Gross lesions in previous reports of piscine botulism are limited to a viscous, amber fecal material in the distal 10–15 cm of the intestinal tract. As botulism is reported to produce gut stasis in mammals,8,10 it was hypothesized that this was due to constipation often associated with animal and human botulism. 3 This lesion is not seen in channel catfish with VTC. In VTC-affected channel catfish, the gastrointestinal tract gross lesions include intestinal intussusceptions and gastric eversion into the mouth. Intussusceptions are most likely acute or possibly agonal given the relative lack of necrosis and inflammation in the intussusceptum. Rather than the distal intestine, in channel catfish, it is the proximal intestine that is often pale with engorged blood vessels. This is most likely due to the effects of the BoNT/E on the transport of chyle and blood flow, which most likely also results in the chylous effusion in the abdominal cavity. The dilated serosal and submucosal vessels in this pale white portion of the intestine lend credence to this hypothesis. Since the presence of lymphatics in fish is steeped in controversy, 11 the vessels devoid of blood cells have been referred to as vessels rather than lymphatics in the current report. Dilated vessels were also a consistent finding in archived histology slides from VTC-suspected cases in 1999 from Arkansas. Also, the catfish may have consumed a recent meal that contained lipids such as a dead fish, as seen in fish no. 4 from farm Y. In studies to replicate botulism in coho salmon (Oncorhynchus kisutch), live fish developed the toxicosis and died after cannibalizing dead fish, which had their intestinal tracts inoculated with BoNT/E spores (presumably leading to sporulation and toxin formation), while those fed feed laced with BoNT/E spores did not develop the toxicosis. 3 Likewise in catfish ponds, fish may become affected after consuming toxic decomposing fish from the bottom of the pond. Anoxic conditions at the bottom of the pond allow for growth of anaerobic bacteria such as C. botulinum on the dead fish. Algae in the stomachs of fish may also serve as a media for growth of C. botulinum. The green alga Cladophora glomerata has been shown to support the growth of C. botulinum, and blooms of this alga have coincided with massive avian die-offs due to BoNT/E in Lake Michigan. 1 Although botulinum C2 toxin produced by certain strains of C. botulinum types C and D has been shown to have enterotoxic effects on gastrointestinal tracts resulting in fluid accumulation, as well as degenerative and necrotic changes in the intestinal mucosa, 9 none of these changes except for submucosal edema were observed in the multiple sections of gastrointestinal tract of the VTC-affected channel catfish in the current study.
Perivascular edematous changes in other peritoneal organs are thought to be the result of cholinergic effects of BoNT/E on the vasculature in these organs. Although a consistent finding in the current report, these changes were not observed in the archived histology slides. This may reflect the possible difference in dose of toxin and the duration of the condition. The edema may also be a result of endotoxins released from the gastrointestinal tract as a sequela to the botulinum toxicosis.
Cerebral congestion is consistent with reports of the effects of botulism in mammals.4,6 Congestion, microthrombi, and petechial hemorrhages are reported in mammals. The latter is thought to be due to breaching of the delicate vessels of the central nervous system by endotoxins released from the gastrointestinal tract in affected animals. 4 This was not seen in the VTC-affected channel catfish in the current study.
Although parasites were noted in the gill biopsies, this level of parasitism is not unusual for sick, inactive fish and is unrelated to the lesions reported in the present study. Periodic acid–Schiff staining confirmed the presence of glycogen within hepatocytes. This is a common finding in well-fed farmed fish.
It is unfortunate but not surprising that the histopathologic lesions seen in VTC-affected fish are nonspecific and cannot form the basis of the definitive diagnosis of this disease syndrome. Routine presumptive diagnosis of this syndrome thus requires the observation of the suite of clinical signs and gross lesions as described above and the exclusion of other infectious diseases. This information together with the catfish bioassay allows for confirmation of the disease while specialized tests such as the endopep mass spectrometry are currently required for definitive diagnosis. The observed lesions in VTC-affected fish are consistent with previous reports of lesions due to botulism. However, these changes are nonspecific and could represent agonal changes (e.g., terminal shock rather than direct damage due to the toxin).
Footnotes
a.
Centricon®, Vaccutainer®; Millipore Corp., Bedford, MA.
b.
Tricane-S®, Western Chemical Inc., Ferndale, WA.
c.
Olympus BX-50 microscope, Spot insight QE digital camera, Spot Basic 3.1 image analysis software; Becton Dickerson, Franklin Lakes, NJ.
d.
Kindly provided by Dr. A. Goodwin, Aquaculture and Fisheries Center, University of Arkansas at Pine Bluff, Pine Bluff, AR.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This research was supported by the College of Veterinary Medicine, Mississippi State University and the Mississippi Agricultural and Forestry Experimental Station (MAFES). This is MAFES Publication J11965.
