Abstract
A case of malignant melanoma in a 7-year-old, intact, black, male Huacaya alpaca with a history of a chronic, nonhealing wound involving the left external nostril, weight loss, and inappetence is described. Malignant melanoma was diagnosed by histology of punch biopsy specimens from a mass on the maxilla associated with the nonhealing wound and from a mass in the submandibular region. The alpaca was humanely euthanized 10 days after the diagnosis on the basis of the poor prognosis and rapid clinical deterioration. At postmortem examination, the alpaca had an ulcerated, multilobulated, black pigmented mass (8.0 cm × 6.0 cm × 4.0 cm) that infiltrated the left rostral maxilla extending into the marrow space and into the left nasal cavity. Numerous, discrete, coalescing masses were present in the subcutaneous tissue of the submandibular area, peritracheal connective tissue, pericardium, and diaphragmatic parietal pleura and were disseminated throughout the pulmonary parenchyma. The masses were diffusely black on cut surface and exuded black pigment. Histologically, all masses were composed of spindloid to polygonal cells with indistinct cell borders and moderate amounts of cytoplasm that contained abundant fine, black granules (melanin), confirming metastasis of a primary mucocutaneous melanoma. To the authors' knowledge, this is the first report of a malignant melanoma involving bone in a New World camelid.
A 7-year-old, 56-kg, intact, black, male Huacaya alpaca presented with a 2-month history of a nonhealing wound on the left external nostril and a 10-day history of left submandibular and retropharyngeal swelling. Before referral, the alpaca had received local wound care and several courses of antibiotics without clinical improvement. The alpaca was housed in a paddock and fed a diet of grass hay and pellets. The animal was treated with anthelmintics and vaccinated (Clostridium perfringens types C and D and Clostridium tetani) regularly. No prior history of illness was reported by the owner. Physical examination revealed a normal temperature (38°C), heart rate (52 beats/min), and respiratory rate (32 breaths/min). The alpaca was thin, with a body condition score of 1.5 out of 5. The mucous membranes were moist, but capillary refill time could not be assessed because of dark pigmentation. Abnormalities were not noted on auscultation of the cardiovascular or respiratory system. Fewer than 2 first-compartment borborygmi were auscultated per minute (reference [ref.] range: 3–4 per min). A raised mass (6.0 cm × 6.0 cm × 3.0 cm) was present on the left cranial-lateral maxilla at the caudal aspect of the nares. The mass was firm, nonmovable, and intimately associated with the overlying black-pigmented skin, which was centrally ulcerated and firm with an irregular, bumpy surface and areas of mild hemorrhage. The mass appeared to extend into the nasal cavity in that the left nasal passage was completely obstructed by a black expansile mass similar in appearance to the external mass. Airflow from the left external nares was absent. The left mandibular lymph node was firm and enlarged, measuring 3.0 cm × 3.0 cm × 1.5 cm. A third, fluctuant subcutaneous mass, measuring 6.0 cm × 4.0 cm × 2.5 cm, was identified just caudal to the ramus of the left mandible.
Initial blood work revealed a marked leukocytosis (25,500 leukocytes/μl, ref. range: 8,000–21,400 leukocytes/μl), characterized by a mature neutrophilia (19,700 neutrophils/μl, ref. range: 4,700–14,900 neutrophils/μl). The serum biochemical profile revealed hyperglycemia (194 mg/dl, ref. range: 89–132 mg/dl), hypoalbuminemia (2.9 g/dl, ref. range: 3.3–4.4 g/dl), hyperglobulinemia (3.3 g/dl, ref. range: 1.9–3.1 g/dl), hyponatremia (141 mmol/l, ref. range: 147–158 mmol/l), and low total CO2 (17.9 mEq/l, ref. range: 19–25 mEq/l). Ultrasound of the left mandibular lymph node revealed a well-encapsulated mass (3.0 cm × 3.0 cm) with a hypoechoic center. Sonographically, the retropharyngeal mass measured 6.0 cm × 4.0 cm and had a loculated center. A fine-needle aspirate of the retropharyngeal mass was performed and yielded a cloudy, viscous, dark-gray fluid.
Differential diagnoses included neoplasia, foreign body, infection (Corynebacterium pseudotuberculosis, tooth root abscess), and trauma. Organisms were not identified on Gram stain of the fluid from the retropharyngeal mass, and aerobic and anaerobic bacterial culture yielded no growth. The alpaca was sedated with butorphanol a (0.09 mg/kg intravenously) to facilitate obtaining punch biopsies of the skin and underlying maxillary mass and the mandibular lymph node, as well as exudate from the retropharyngeal mass. The biopsy specimens were fixed in 10% neutral buffered formalin and submitted to the Washington Animal Disease Diagnostic Laboratory (WADDL; Pullman, WA) for evaluation. The alpaca was discharged to the owner's care with instructions for daily cleaning of the maxillary mass and for use of systemic antibiotics (florfenicol, b 20 mg/kg intramuscularly, q48hr).
The biopsy specimens were processed routinely and embedded in paraffin, 4-μm sections were cut, placed onto glass slides, and stained with hematoxylin and eosin (HE). Initial histological examination revealed highly pigmented tissue. Selected sections were bleached with 0.25% potassium permanganate, treated with 5% oxalic acid, and then stained with HE to facilitate accurate evaluation. Light microscopy of the biopsy specimen from the maxilla revealed a highly cellular mass that infiltrated an overlying band of dense fibrous tissue that was covered by a thin serocellular crust. The absence of epidermis indicated that the biopsy was taken from an ulcerated area. The mass was composed of polygonal to spindloid cells separated into nests and bundles by thin strands of collagenous stroma. The cells had distinct margins and moderate amounts of eosinophilic cytoplasm, which contained variable amounts of fine, black pigment granules (melanin). Nuclei were round to oval with coarsely clumped chromatin and variably distinct nucleoli. Anisocytosis and anisokaryosis were marked, with a few giant nuclei and an average of 25 mitotic figures per 10 high-power (400×) fields of view (Fig. 1). The biopsy from the mandibular lymph node included a wide band of fibrous connective tissue separated from a small nodule of neoplastic cells by a discontinuous rim of remnant lymphoid tissue consisting of sheets of mature lymphocytes and a few small venules. The nodule was composed of sheets of pigmented cells similar to those described above, mixed with large numbers of epithelioid cells containing uniform nuclei and large amounts of coarse, black pigment granules within the cytoplasm, with extensive infiltration of the adjacent connective tissue. A diagnosis of mucocutaneous malignant melanoma of the left external nares with metastasis to the mandibular lymph node was made. Two weeks after the initial examination, the alpaca was presented for tachypnea and open-mouth breathing and was euthanatized on the basis of a guarded prognosis and the rapid progression of clinical signs.
A nonmovable, raised, ulcerated mass (8.0 cm × 6.0 cm × 4.0 cm) originated in the skin overlying the maxilla just caudal to the left nostril. The mass invaded and replaced the underlying maxillary bone and extended into the nasal cavity. The mass completely occluded the left nasal cavity and was in contact with the nasal septum, which was slightly deviated to the right. The mass extended 4 cm caudally within the marrow space of the maxilla (Fig. 2). The mass was firm and homogeneously black on the cut surface. Numerous, similar, discrete to coalescing black masses ranging from 0.2 to 7.0 cm in diameter were scattered along the left ventral neck, throughout the pulmonary parenchyma, and along the pericardium and diaphragmatic parietal pleura (Fig. 3). The mass in the retropharyngeal area had a soft necrotic center. No abnormalities were identified within the abdominal cavity. After the complete postmortem exam, selected tissues were fixed in 10% neutral buffered formalin and submitted along with the entire head and neck to WADDL for histopathology. Sections from the maxillary mass, mandibular lymph node, retropharyngeal mass, trachea and associated masses, pericardial and pleural masses, lung, liver, kidney, spleen, and brain were processed and evaluated histologically as previously described. Maxillary specimens were also decalcified for 24–48 hr before processing and paraffin embedding. All masses evaluated consisted of neoplastic cells with a similar arrangement and morphology to those described in the original biopsy specimens. Where not ulcerated, the external portion of the mass extended from the dermal-epidermal junction (without infiltration of the epidermis) through the maxillary bone and into the nasal cavity with complete obliteration of adnexal structures and normal dermal and subcutaneous architecture. Neoplastic cells infiltrated into and partially replaced the bone marrow of the maxilla adjacent to the tumor and extended a few centimeters caudally. The mandibular lymph node, retropharyngeal mass, and multiple masses from along the trachea had a narrow rim of compressed, marginal lymphoid tissue in some areas and portions of a capsule, which identified them as lymph nodes. The masses in the lungs were discrete and nonencapsulated and replaced approximately 30% of the parenchyma.
Melanocytic tumors arise from genetically altered epidermal melanocytes or melanoblasts derived from embryonal neuroectoderm. 1,5,7,10 The biological behavior of malignant melanoma in both humans and dogs is variable but is often characterized as highly aggressive with rapid spread from the primary tumor site to advanced-stage metastatic disease. Metastasis occurs hematogenously or via lymphatics to regional lymph nodes, liver, and lung. Advanced-staged metastatic melanoma is considered largely refractory to surgical, chemotherapeutic, or radiation therapy. 1,5,11 In humans, genetic predisposition, DNA mutations, and exposure to ultraviolet radiation are thought to contribute to the development of malignant melanoma. Evidence is mounting in humans that a mutation at the v-raf murine sarcoma viral oncogene homolog B1 (BRAF) kinase gene is associated with the development of malignant melanoma in sun-protected sites on the body. 1 The etiopathogenesis of canine melanoma is not well described but is suspected to be similar to the human disease process. 5,10,11 Four primary forms of malignant melanoma have been described in dogs, including dermal, uveal, oral, and mucocutaneous. 5
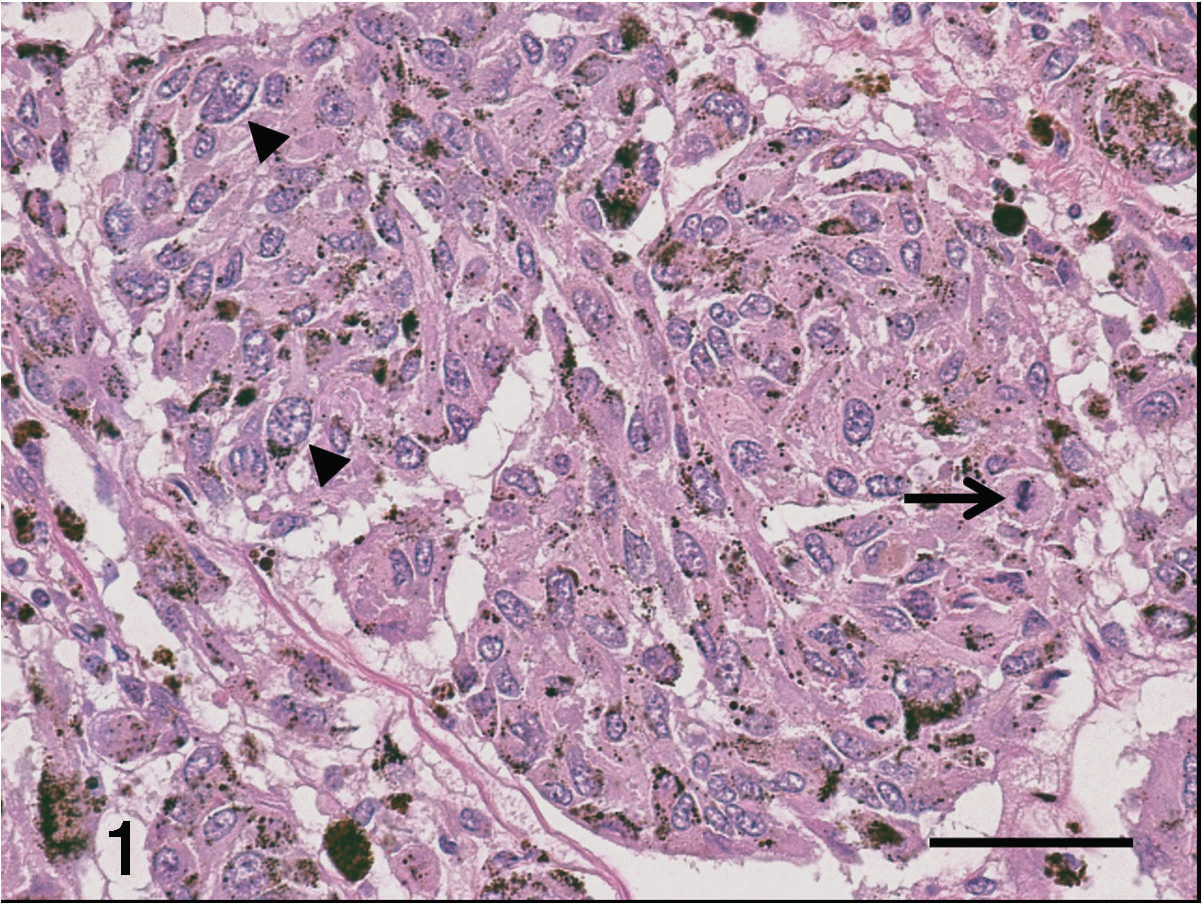
Hematoxylin and eosin–stained sections of the mucocutaneous mass showing melanin granules within the cytoplasm of neoplastic cells, a mitotic figure (arrow), and marked anisokaryosis with a few very large nuclei (arrowheads). Bar = 50 μm.

Transverse section of the rostral skull at the level of the carnasal teeth (

Gross specimen of thoracic cavity with the lungs in situ demonstrating multifocal to coalescing metastatic melanoma involving the pericardium and diaphragmatic parietal pleura and disseminating throughout the pulmonary parenchyma.
Melanocytic neoplasia, both malignant (melanoma) and benign (melanocytoma), has been described in all domesticated large animal species. 4,13 A higher prevalence of melanocytic tumors has been reported in animals with pigmented mucosa and in horses, cattle, dogs, goats, and pigs with dark skin. 4,7,8,13 In the horse, melanomas most frequently occur in gray horses, and it is believed that a disturbance in melanin metabolism occurs during the graying process, leading to overproduction and accumulation of melanoblasts in the dermis. 4,13
Retrospective studies 2,9,12 reported the prevalence of neoplasia in the New World camelid to be 1.0–8.3%. To date, there has been 1 report of nonmetastatic intraocular melanoma in a 2-year-old female alpaca, 3 2 reports of benign cutaneous melanocytoma in llamas, 6,12 and 1 abstract describing cutaneous metastatic malignant melanoma and melanocytoma in an alpaca with metastases to the local lymph nodes, lung, and liver (Avakian A, Parry NM: Cutaneous and metastatic melanoma and melanocytoma in an alpaca [Abstract]. Combined 57th and 41st Annual Meeting of the American College of Veterinary Pathologists and the American Society for Veterinary Clinical Pathology, Tucson, AZ, December 2–6, 2006. Vet Pathol 43:817). None of the aforementioned reports made reference to the animal's skin color.
The malignant melanoma in the case here originated in the skin at the mucocutaneous junction of the left nares and was locally invasive, with metastasis to the regional lymph nodes and lung. To the authors' knowledge, this is the first detailed clinical report of malignant melanoma in an alpaca and the first involving bone. Melanocytoma or melanoma should be considered as a differential diagnosis for cutaneous masses or nonhealing wounds in the New World camelid, especially involving mucocutaneous junctions. This case demonstrates that mucocutaneous melanoma in the camelid has the potential to be locally invasive with metastasis to regional lymph nodes and lung.
Footnotes
a.
Dolorex®, Intervet Inc., Millsboro, DE.
b.
NUFLOR® Injectable Solution, Schering-Plough Animal Health Corp., Kenilworth, NJ.
