Abstract
A 9-y-old, male ring-necked pheasant (Phasianus colchicus) was autopsied following euthanasia because of acute distress, recumbency, and dyspnea. The bird had experienced a protracted period of neuromuscular disease localized to the left sciatic nerve. Gross and histologic examination of the large intestine revealed intramural nodules predominantly comprised of atypical, whorling spindle cells with small cores of granulomatous inflammation centered on cross-sections of immature adult nematodes. The body structures of these metazoan organisms and clinical disease manifestation are consistent with Heterakis isolonche infection. Nodular spindle cell proliferations without granulomatous inflammation or intralesional nematodes were also found throughout the liver and lungs, suggesting metastasis from the intestine. Immunohistochemical staining of the hepatic and pulmonary tumor tissue with vimentin and S100 suggests a neurofibroblastic origin.
A 9-y-old, male ring-necked pheasant (Phasianus colchicus) from the Columbus Zoo and Aquarium (Columbus, Ohio) was autopsied following euthanasia because of acute distress and dyspnea. Previous medical history included a broken blood feather during molting in August 2012 and a protracted period of neuromuscular disease characterized by proprioceptive deficits localized to the left sciatic nerve beginning in November 2014; neuromuscular disease was treated as needed with meloxicam (1–1.125 mg orally every 24 h for 7–10 d), gabapentin (12.5 mg orally every 12–24 h for 7–14 d), or tramadol (10 mg orally every 24 h for 14 d). Fecal examinations were negative for endoparasitism from the time of procurement in November 2011, until the most recent fecal examination 8 d prior to euthanasia in May 2015.
Increasing leukocytosis characterized by increasing heterophilia and persistent monocytosis were documented beginning 1 mo prior to euthanasia, despite treatment with enrofloxacin (6 mg orally every 24 h for 7 d) and trimethoprim–sulfamethoxazole (48 mg orally every 12 h for 10 d). Twelve days before the bird’s death, the leukogram revealed a white blood cell count of 69.8 × 109/L, a heterophil count of 44.67 × 109/L, and a monocyte count of 13.96 × 109/L. At that time, differential diagnoses included aspergillosis, chlamydiosis, mycobacteriosis, or salmonellosis. A 2-wk course of doxycycline (30 mg orally every 24 h) and itraconazole (6 mg orally every 24 h) treatment was initiated. Four days later, when the bird became recumbent and weak with open-beak breathing, euthanasia was elected.
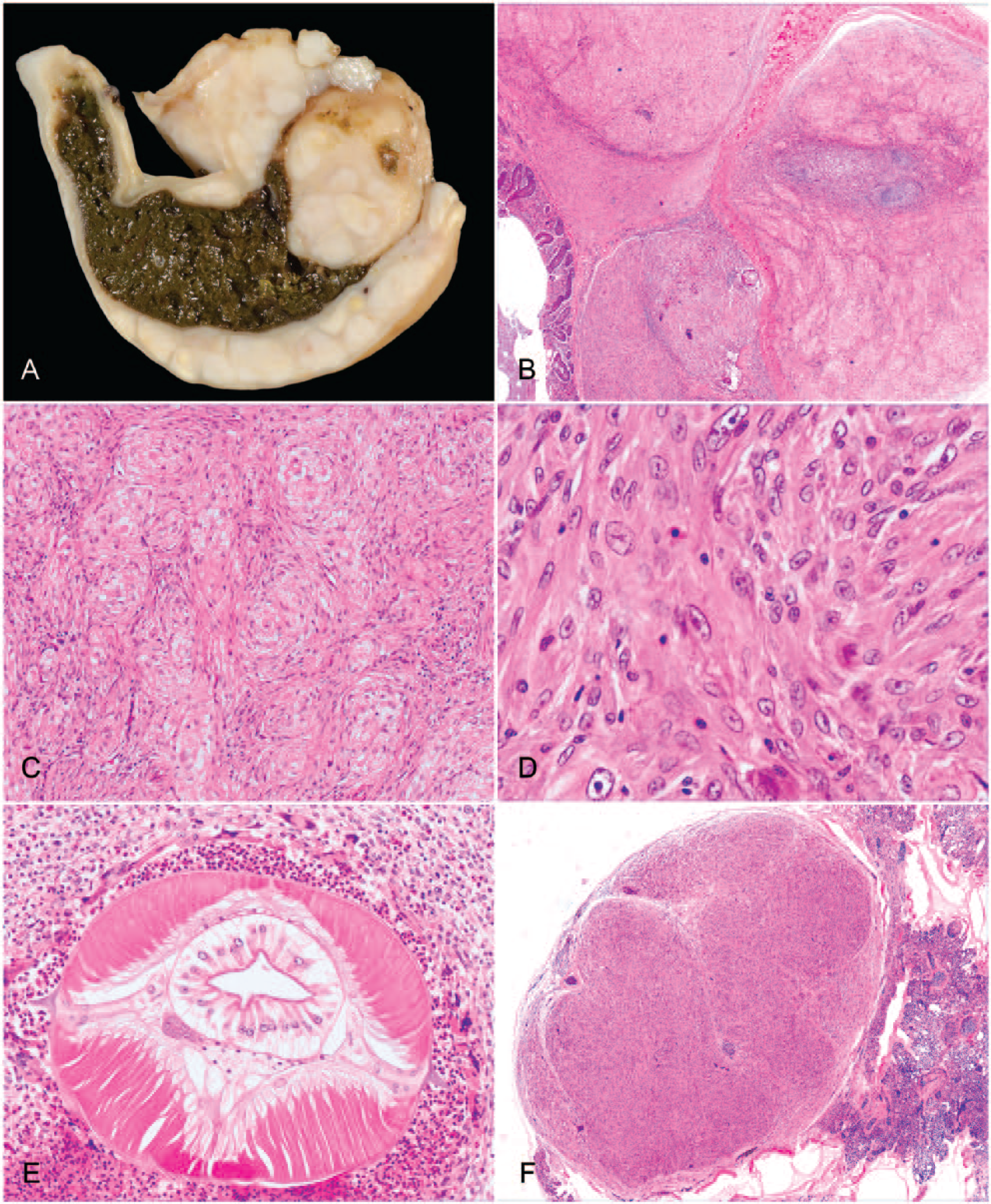
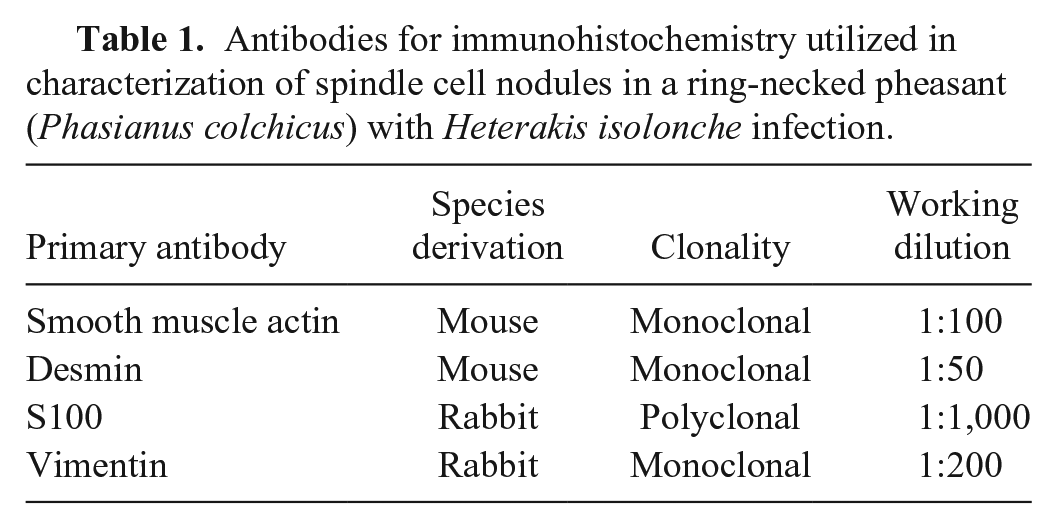
An autopsy was performed by attending zoo veterinarians, and a complete set of formalin-fixed tissues was submitted to the surgical biopsy service at The Ohio State University Veterinary Medical Center (Columbus, OH). Gross postmortem examination revealed confluent, large, ~5–15 mm diameter, invasive, white, nodular masses in the wall of the large intestine (Fig. 1A) as well as small, ~2–5 mm diameter, white nodules in the liver and lung that were visible on the serosal surfaces and extended an equal distance into the organs’ parenchyma. There were no other significant gross findings. Differential diagnoses following autopsy were mycobacteriosis (i.e., Mycobacterium avium or Mycobacterium genavense) or lymphosarcoma caused by avian leukosis virus. Tissues were routinely trimmed, embedded, and stained with hematoxylin and eosin for initial evaluation. After examination of the histopathology findings, immunohistochemistry was performed using antibodies against smooth muscle actin (SMA; Dako North America, Carpinteria, CA), desmin (Dako), S100 (Dako), and vimentin (Abcam, Cambridge, MA; Table 1). The SMA antibody had been validated by the manufacturer to recognize avian α-SMA. All antibodies were also validated in non-lesioned tissues from the same bird.
Antibodies for immunohistochemistry utilized in characterization of spindle cell nodules in a ring-necked pheasant (Phasianus colchicus) with Heterakis isolonche infection.
Histologically, large, confluent spindle cell nodules expanded transmurally and effaced the large intestine from the level of the lamina propria through the serosa (Fig. 1B). These nodules were composed largely of whorling and interlacing spindle cells in a loose collagenous stroma that had ovoid nuclei and abundant streaming eosinophilic cytoplasm (Fig. 1C). Neoplastic spindle cells demonstrated cytologic features of malignancy, including anisocytosis, anisokaryosis, nuclear pleomorphism, binucleation, and multiple nucleoli (Fig. 1D). There were 4 mitotic figures in 10 consecutive high-power (400×) fields. In the center of these nodular proliferations within the large intestine, there were often small cores of heterophilic and granulomatous inflammation with prominent foreign body–type multinucleate giant cells centered on nematode organisms. Cross-sections of metazoan parasites were identified as heterakid nematodes by their lateral alae, thin cuticle, lateral chords, polymyarian coelomyarian musculature, a pseudocoelom, a digestive tract composed of uninucleate columnar cells, and an immature gonad (Fig. 1E). In the lung and liver, there were smaller nodules consisting solely of mesenchymal proliferations identical to those observed in the intestine (Fig. 1F).
Immunohistochemistry was performed on both the primary intestinal and putative metastatic hepatic and pulmonary nodules to further characterize the spindle cell composition, and staining profiles were similar in all sites examined. Photomicrographs in Figure 2 depict the immunohistochemical staining characteristics of the pulmonary mass in Figure 1F. Neoplastic cells diffusely stained positive for vimentin (Fig. 2A) and S100 (Fig. 2B) but negative for SMA (Fig. 2C) and desmin (Fig. 2D), which labeled only intratumoral vessels. Coelomyarian musculature of the nematode parasite was immunopositive for SMA and S100. Based on histopathology and immunohistochemistry, the pheasant was diagnosed with nodular, heterophilic, and granulomatous typhlocolitis with intralesional immature adult heterakids, as well as malignant mesenchymal neoplasia of neurofibromatous immunophenotype with pulmonary and hepatic metastases.
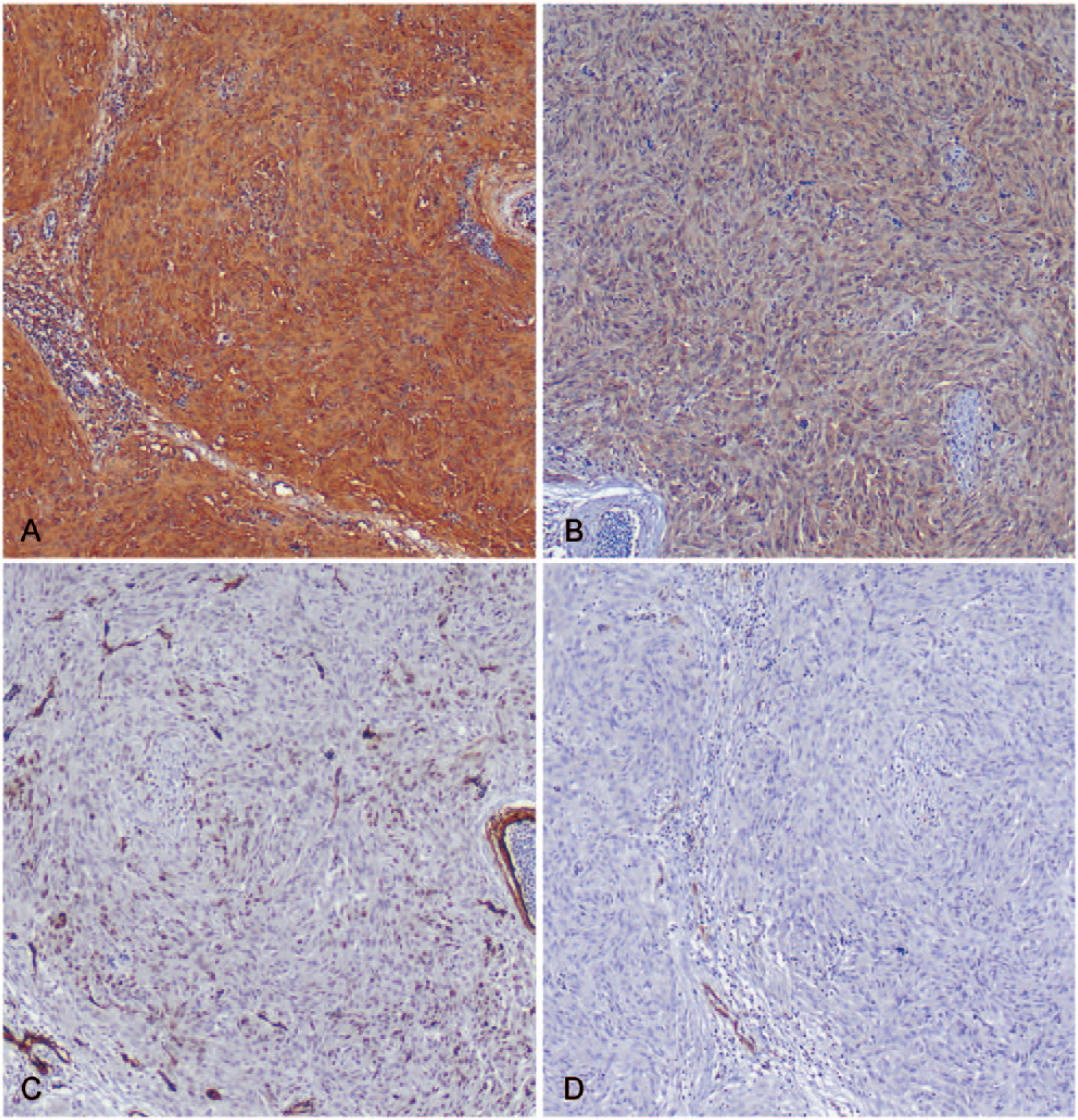
A pulmonary mass (H&E shown in Fig. 1F) demonstrates positive immunostaining for vimentin (
The nature of the proliferative spindle cell population associated with nodular typhlitis in pheasants has long been debated. Nomenclature such as “sarcoid leiomyoma,” 9 “parasitic neoplasia,” 9 “leiomyoma,” 11 “parasitic granuloma,” 5 “verrucose typhlitis,” 11 “nodular typhlitis,” 15 and “pseudoneoplastic” 17 have been used to describe this condition. Extra-cecal involvement interpreted as metastasis in our case leads us to favor a diagnosis of true mesenchymal malignancy. Although the possibility of aberrant migration of the larval parasites outside the cecum cannot be excluded, no nematodes were identified outside the intestinal tract, and the nodules of spindle cell proliferation within the liver and lung lacked heterophilic or granulomatous centers. Furthermore, because migration of second-stage Heterakis isolonche larvae into the cecal wall and back into the lumen as adults is a normal part of parasitic development in its definitive host, 3 aberrant migration outside the cecum—and in multiple foci—in its definitive host is considered less likely. The related heterakid Heterakis gallinarum has not been associated with such nodular proliferations, and is only considered pathogenic insofar as its role as an intermediate host in the transmission of Histomonas meleagridis, the causative agent of blackhead, which is not known to be transmitted by H. isolonche.6,17 Although pulmonary or hepatic nodules interpreted as granulomas were also observed in 3 of the 7 gallinaceous birds with nodular typhlitis reported previously, 8 the nature of these lesions was not characterized histologically, and the authors concluded that the spindle cell component of the coincident cecal nodules represented “severe reactive parasitic fibroplasia.” 8
Our report characterizes the microscopic and immunophenotypic composition of the extraintestinal lesions in nodular typhlitis associated with H. isolonche infection. Histochemical, immunohistochemical, and electron microscopic techniques utilized in previous studies suggest that the spindle cells comprising the bulk of these cecal masses are of fibroblastic 8 or myogenic histogenesis. 1 In our case, immunohistochemistry was also performed on metastatic pulmonary and hepatic nodules to exclude possible confounders in the intestinal lesions, such as granulation tissue and entrapped or regenerative smooth muscle. Positive immunohistochemical staining for vimentin and S100 and negative staining for SMA and desmin among the neoplastic spindle cell population suggest a neurofibroblastic derivation,4,10 which may include Schwann cells and/or perineurial fibroblasts. 14 Strong, diffuse S100 staining in intestinal neurofibromatous tumors is associated with Schwann cell differentiation; 14 negative staining for SMA and desmin rule out other differentials such as hemangiopericytoma or leiomyosarcoma. 4
From a comparative pathology perspective, others have cited the neoplastic inductive capacity of endoparasites such as the nematode Spirocerca lupi in canids and the trematode Schistosoma spp. in primates. 9 Esophageal nodules associated with S. lupi reportedly evolve primarily from inflammatory or reactive proliferations through malignant transformation to fibrosarcoma or extraskeletal osteosarcoma. 7 In veterinary medicine, parasite-associated neoplasms are considered host-derived; however, intriguingly, there has been a report of parasite-derived malignant neoplasia of the cestode Hymenolepis nana metastasizing throughout its host in an immunocompromised human patient. 12 In domestic species, neu oncogene point mutations have been associated with malignant schwannomas and peripheral nerve sheath tumors. 16 A similar pathogenesis of progression from reactive to malignant spindle cells may be involved in the present case, with metastasis from the intestine to the lung and liver being the positive proof of malignancy. The histopathology in this pheasant is not unlike that of malignant peripheral nerve sheath tumors, which have been best characterized in dogs and cattle, but are only rarely reported in wild species, including damselfish, a muskrat, 2 and a water moccasin. 13 In this advanced case of large intestinal H. isolonche infection in a pheasant, nodular typhlocolitis progressed to neurofibromatous malignant mesenchymal neoplasia with pulmonary and hepatic metastases.
Footnotes
Acknowledgements
We thank Dr. Paul Stromberg for his assistance in parasite identification in histologic sections.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
