Abstract
Postmortem lesions and cause of death were evaluated retrospectively for 963 horses examined as part of the Ontario Racing Commission Death Registry over a 13-y period. The Death Registry was established in 2003 to identify factors leading to death or euthanasia of racehorses in this Canadian province. Postmortem examination was carried out on 56% of horses reported to the Death Registry and included Standardbred, Thoroughbred, and American Quarter Horses. Musculoskeletal injury was the most common reason for death associated with racing or training among all racehorses and involved 68% of horses. A history of sudden death during or immediately following exercise was described for 31% of racing- or training-associated deaths, and in 16% of all horses in the study. Sudden death occurred in horses of all breeds, and our report describes lesions associated with sudden death in a series of Standardbreds. The cause of death in these cases was most frequently attributed to cardiopulmonary lesions. The cause of death was undetermined in 20% of sudden death cases, and it is speculated that cardiac arrhythmia may have contributed to these deaths. Injection-associated death was documented in 4% of the study population. Lesions among horses whose death was not associated with exercise were similar to those in other equine populations, and lesions involving the gastrointestinal system were most common. Standardization of pathology reporting of pulmonary and other lesions in racehorse postmortem cases would allow direct comparison of results among racehorse populations.
Keywords
Introduction
The risk of race-related fatal injury or death in racehorses is recognized worldwide. Among Thoroughbred (TB) horses racing in flat races in various geographic areas worldwide, risks of fatality have been calculated at 0.44–2.95 per 1,000 starts.3,7,33 For the subset of catastrophic musculoskeletal injury, risks of 1.7–1.99 per 1,000 starts have been described in the United States and Canada.7,12 In the past, postmortem (PM) examination of racehorses has not been carried out routinely because of cost and availability. As a result, reports documenting the cause of death or conditions leading to euthanasia of racehorses are limited.3,14,15,18,22 Catastrophic fracture and sudden death during exercise are recognized as significant contributing factors to racehorse death.3,11,22,26 PM documentation of the cause of death in racehorses of various breeds and race types, and in different geographic regions, provides important background information and directs research efforts, with the ultimate goals of reducing fatalities and overall improvement in the welfare of racehorses.
The Ontario Racing Commission (ORC) was established in 2000 as an independent government agency overseeing horse racing at 13 Standardbred (SB) tracks, 2 TB flat-racing tracks, and 1 Quarter Horse (QH) track in the province of Ontario, Canada. In April 2016, the ORC merged with the Alcohol and Gaming Commission of Ontario (AGCO). The ORC, now AGCO, Death Registry was established on January 1, 2003 in part to identify factors leading to death or euthanasia of racehorses in this province. Under the program, owners, trainers, and/or veterinarians are required to notify ORC (AGCO) of the death of any racehorse that has been entered or qualified to race, or completed an official workout, in the province in the 60 days prior to death. For each reported death, a requirement for PM is at the discretion of the Registrar of AGCO. Our retrospective descriptive study evaluated PM lesions and cause of death among Ontario racehorses over a 13-y period, and compares these findings to those of similar studies in other jurisdictions.
Materials and methods
PM cases submitted through the ORC Death Registry to the Animal Health Laboratory (AHL), University of Guelph from January 1, 2003 to December 31, 2015 were identified retrospectively using the 2 laboratory information management systems (LIMS) in place sequentially at the AHL during this time period (2003–2007: VADDS; Advanced Technology, Ramsay, NJ; 2007–2015: Sapphire; LabVantage Solutions, Somerset, NJ). All PM cases submitted by the ORC were included in the study. Problem-oriented PM examinations were conducted from 2003 to 2007, with ancillary test selection and tissue sampling at the discretion of individual case pathologists. Beginning in 2008, a standard protocol for PM examination of Death Registry cases was instituted, which included gross PM and histopathology on a standard set of tissues based on the clinical category of each case (fracture; unexpected death; or death associated with a specific clinical syndrome; Supplementary Table 1). The need for ancillary microbiology was determined by the pathologist for each case. Toxicology for selected cases was carried out at the discretion of the ORC.
Submission information and pathology reports were reviewed for each case, and the following data were captured: date of PM; pathologist; breed; sex; lip tattoo, freeze brand, and/or age; clinical history, including whether the animal died or was euthanized; association of death with racing or training; and PM diagnosis, including morphologic diagnosis(es) and/or cause of death. In those cases for which age was not provided in the history, the horse’s age was determined from the lip tattoo (for TB) or freeze brand (for SB) using schedules published by the Jockey Club (http://www.jockeyclub.com/) and Standardbred Canada (http://www.standardbredcanada.ca/), respectively. For each case, the primary diagnosis or lesion responsible for death or euthanasia was subsequently categorized based on the body system involved, as follows: fractured limb; axial fracture; non-fracture musculoskeletal; gastrointestinal (GI); respiratory; cardiovascular (CV); central nervous system (CNS); renal; injection-associated; other miscellaneous conditions; or cause of death undetermined.
Results
A total of 1,708 horse deaths were reported to the Death Registry between January 1, 2003 and December 31, 2015. PM was requested on a subset of 963 of 1,708 (56%) horses, with an annual range of 26–81% of all reported deaths (Table 1). These included all horses with a history of sudden death during or immediately following exercise. Death Registry PMs contributed to 963 of 4,538 (21%) of all equine PM cases at the AHL during this time period. Beginning in 2008, PM was requested for fewer fracture cases, and emphasis was placed on submission of non-fracture cases, resulting in a lower number of total submissions for 2008–2015 than in previous years. The combined effects of a decrease in the racehorse population in Ontario over the 13 y of the study and a reduction in the annual number of racing dates in the province is reflected in the decrease in the number of reported deaths over time.
ORC Death Registry, 2003–2015: total and PM cases.
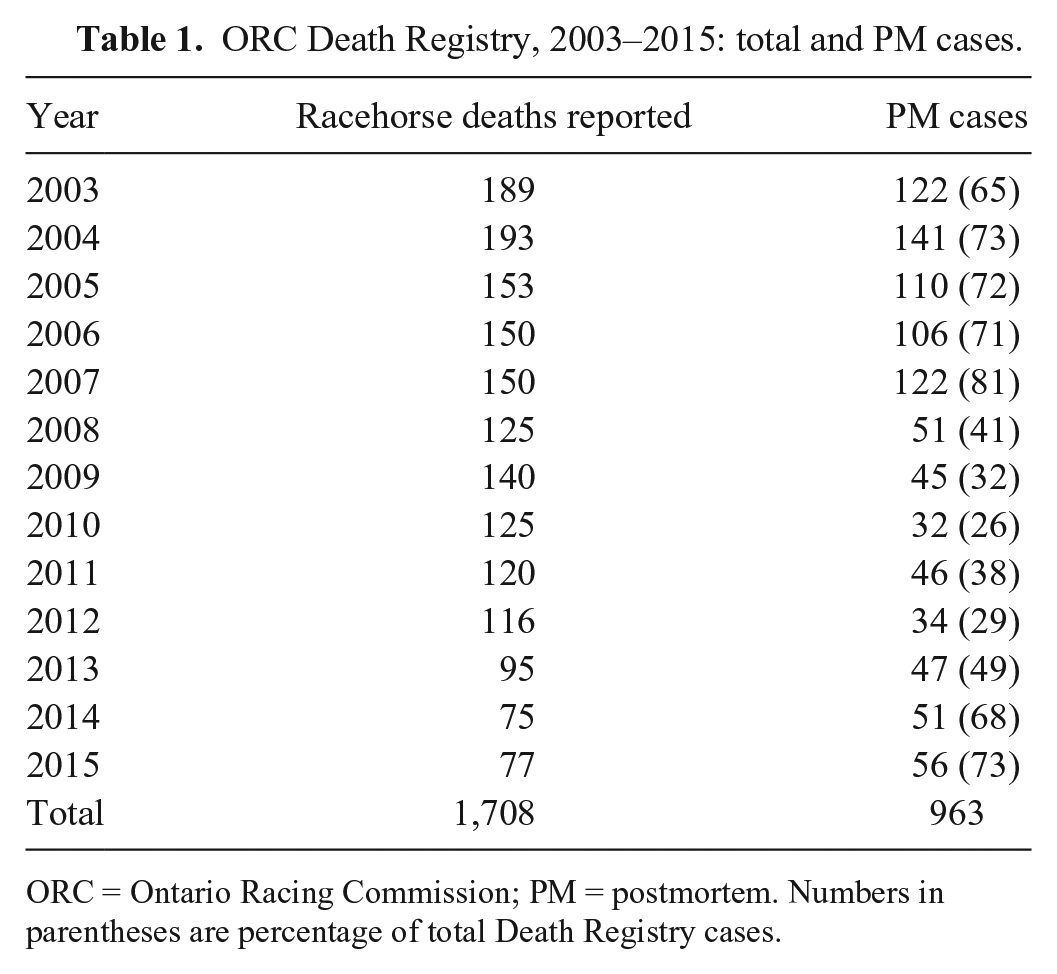
ORC = Ontario Racing Commission; PM = postmortem. Numbers in parentheses are percentage of total Death Registry cases.
PM cases included 516 of 963 (53.6%) SB, 422 of 963 (43.8%) TB, and 25 of 963 (2.6%) QH. Age range for SB was 2–12 y (median: 5 y, mean: 5.2 y); 1–13 y for TB (median: 3 y, mean: 3.8 y), and 2–6 y for QH (median: 5 y, mean: 4.3 y). Age was undetermined because of illegible or untraceable tattoo or freeze brand, and was not provided in the clinical history for 45 of 516 SB, 99 of 422 TB, and 22 of 25 QH. A total of 339 female (157 SB, 169 TB, 13 QH), 598 male (stallion or gelding; 348 SB, 239 TB, 11 QH), and 1 intersex (SB) horses were included. Sex was not recorded for 10 SB, 14 TB, and 1 QH. A total of 24 veterinary pathologists and pathology trainees contributed to PM examinations.
An overall summary of PM lesions is listed in Table 2. Among all cases, 514 of 963 (53%) deaths were causally associated with racing or training and included 224 of 516 (43%) SB, 274 of 422 (65%) TB, and 16 of 25 (64%) QH. Musculoskeletal injury was the most common reason for death associated with racing or training, involving 348 of 514 (68%) cases, and the majority of these horses were euthanized (331 of 348, 95%). Most cases involved limb fractures (299 of 348, 86%), and these were identified in 110 of 516 (21%) SB, 184 of 422 (44%) TB, and 5 of 25 (20%) QH. Anatomic locations of limb fractures are listed in Table 3. In SB, fractures involving the first, or proximal, phalanx (P1) were most common (62 of 110, 56%). In TB, fractures involving bones contributing to a forelimb fetlock joint (third metacarpal bone, MC3; 1 or both proximal sesamoid bones; and P1) were most common (106 of 184, 58%), with MC3 most frequently involved. Carpus was the second most common fracture site in TB (38 of 184, 21%).
Summary of postmortem findings in Ontario racehorses by breed, 2003–2015.
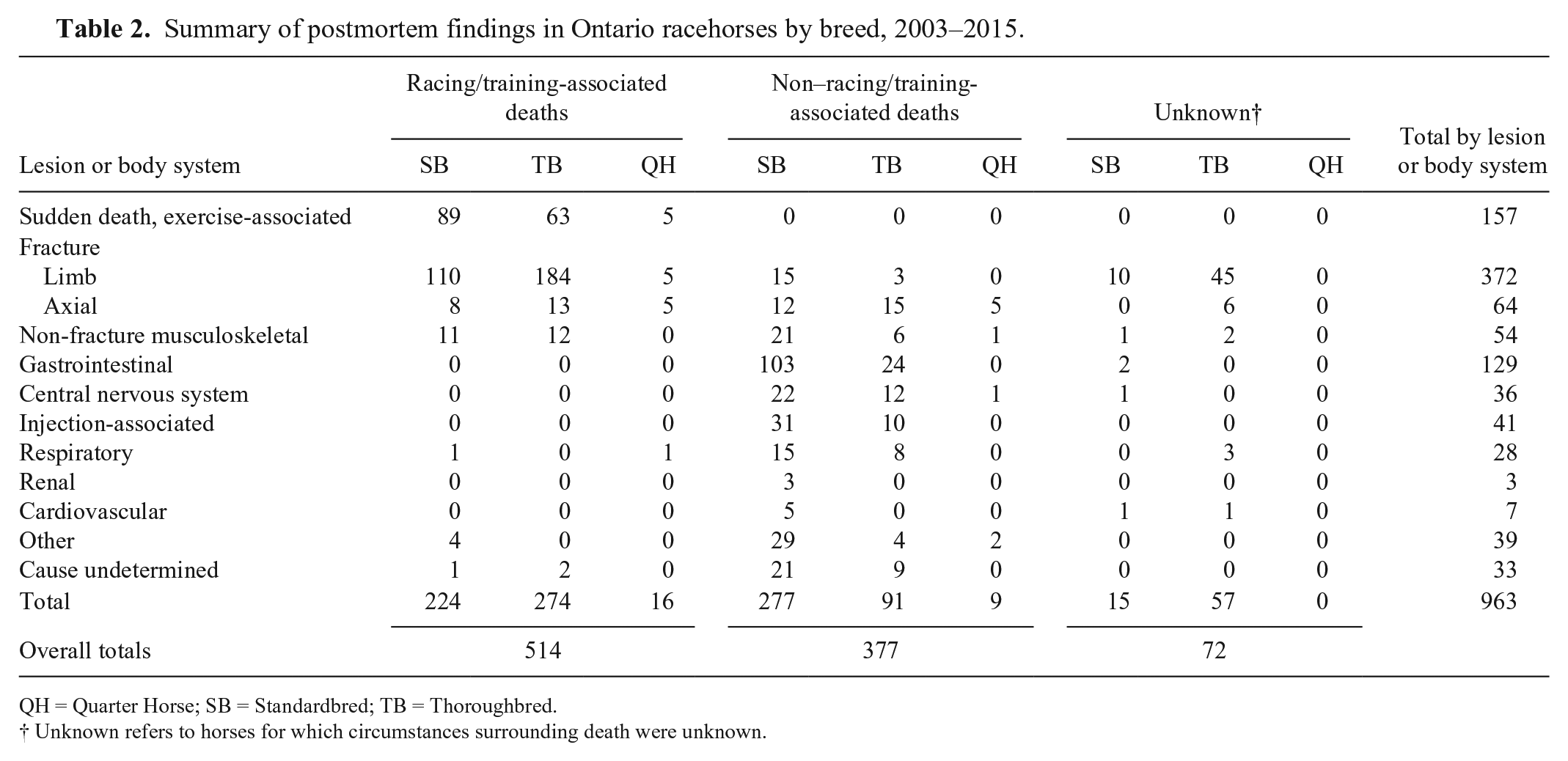
QH = Quarter Horse; SB = Standardbred; TB = Thoroughbred.
Unknown refers to horses for which circumstances surrounding death were unknown.
Anatomic location of limb fractures associated with racing or training in ORC Death Registry postmortem cases, 2003–2015.
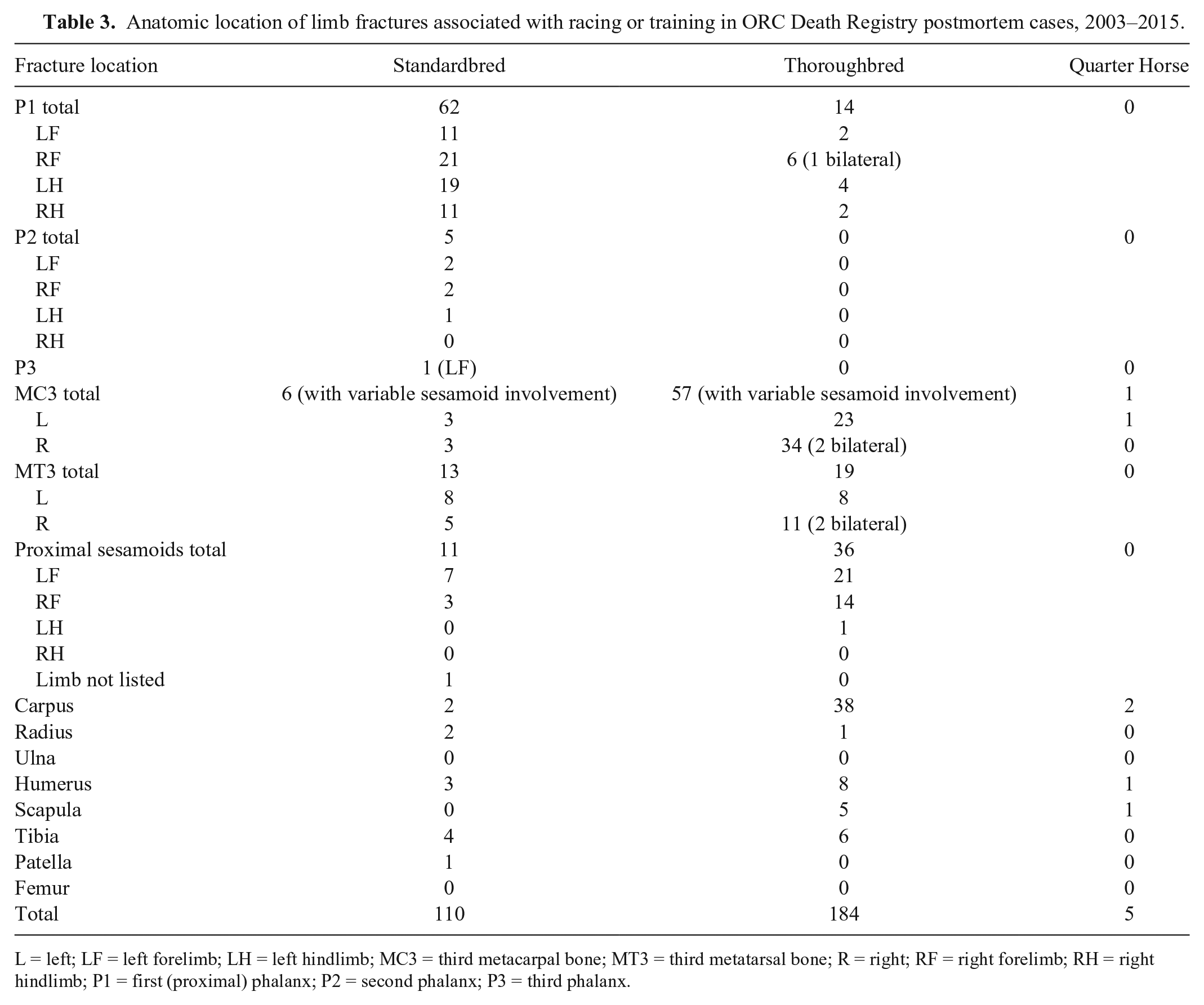
L = left; LF = left forelimb; LH = left hindlimb; MC3 = third metacarpal bone; MT3 = third metatarsal bone; R = right; RF = right forelimb; RH = right hindlimb; P1 = first (proximal) phalanx; P2 = second phalanx; P3 = third phalanx.
Fractures involving the axial skeleton were identified in 26 of 348 (7%) horses with racing or training musculoskeletal injuries, and involved pelvis (16 of 26, 62%), skull (6 of 26, 23%), vertebrae (3 of 26, 12%), or ribs (1 of 26, 4%). On-track accidents involving other horses or stationary objects were responsible for injuries in 7 of 31 (23%) of these cases. Racing- or training-associated non-fracture musculoskeletal injuries resulted in euthanasia of 11 SB and 12 TB because of a variety of soft tissue injuries including rupture of flexor or extensor tendons, suspensory ligaments, or other ligaments. One 5-y-old TB horse died as the result of exertional rhabdomyolysis following training. In this animal, there was no histologic evidence of previous episodes of muscle injury, associated polysaccharide storage myopathy, or of other concurrent disease.
Unexpected (sudden) death occurred during or immediately following racing or training in 157 of 963 (16%) of all horses in the study, representing 157 of 514 (31%) of horses for which death was causally associated with these activities (Table 4). These included 89 of 516 (17%) SB, 63 of 422 (15%) TB, and 5 of 25 (20%) QH. Considering all deaths reported to the Death Registry during this time period, the proportional mortality rate for exercise-associated sudden death among all breeds was 9% (157 of 1,708). The cause of death was undetermined in 32 of 157 (20%) of these horses. Of horses with a history of exercise-associated sudden death that had significant PM findings, lesions involving respiratory tract were most frequent and were considered the definitive or likely cause of death, or to have contributed to death, in 79 of 157 (50%) of animals. Severe pulmonary hemorrhage, interpreted by the case pathologist as consistent with exercise-induced pulmonary hemorrhage (EIPH), was the most common respiratory tract lesion identified. EIPH was considered the definitive or likely cause of death in 60 of 79 (76%) sudden death cases with respiratory lesions, representing 60 of 157 (38%) of all sudden death cases. Concurrent thoracic lymphoma was identified in 1 horse with EIPH. Pulmonary hemorrhage (13 of 157, 8%) and pulmonary edema and congestion (3 of 157, 2%) were described in other horses whose deaths were associated with exercise, but no definitive relationship was made by the pathologist between these lesions and the cause of sudden death. Although SB and TB were similarly represented among exercise-associated sudden death cases, both pulmonary hemorrhage and EIPH-associated pulmonary hemorrhage were more frequently described among SB (49 of 89, 55%) than among TB (23 of 63, 37%) that died suddenly while racing or training.
Cause of death or significant postmortem lesions in horses dying suddenly during exercise.

EIPH = exercise-induced pulmonary hemorrhage.
CV lesions were considered to have caused or contributed to death in 34 of 157 (22%) exercise-associated sudden death cases. Hemopericardium with cardiac tamponade resulted in death in 15 of 157 (10%) cases, of which 9 of 15 (60%) were SB, 5 of 15 (33%) were TB, and 1 of 15 (7%) was a QH. A definitive site of aortic rupture was identified in 13 of 15 (87%) of these horses. Hemoabdomen, hemothorax, or mediastinal and/or pulmonary hemorrhage caused or contributed to death in 13 of 157 (8%) animals. In 7 of 13 (54%) of these horses, the site of vascular rupture was identified, and the source of hemorrhage remained unknown in 6 of 13 (46%) animals. Other gross or histologic lesions involving the heart were considered significant in 6 of 157 (4%) horses and included valvular endocarditis in 2 horses, and 1 horse with each of endocardial fibrosis, mild myocarditis, focal myocardial degeneration and fibrosis, and cardiac hypertrophy leading to acute heart failure.
Skull fractures were present in 5 of 157 (3%) horses that died suddenly during exercise, although whether this was the actual cause of death or a perimortem lesion resulting from collapse could not be determined. Sepsis (3 of 157, 2%); mild encephalitis (2 of 157); disseminated intravascular coagulation (1 horse), and multi-organ hemorrhage (1 horse) were also identified in horses with a history of exercise-associated sudden death, and were suggested as contributing factors to death in these animals.
Of the remaining 9 horses in which death was associated with exercise but was not categorized as sudden death or the result of musculoskeletal injury, 3 horses were euthanized because of post-exercise lameness but lacked PM lesions indicative of the cause; 3 horses were euthanized as a result of serious accidents on the racetrack; 2 horses had lesions compatible with EIPH but did not die during or immediately after exercise; and 1 horse had lesions consistent with septicemia.
The circumstances surrounding death or leading to euthanasia were not described in the clinical history provided for 72 of 963 (7%) cases. Of these, 64 of 72 (89%) horses had fractures involving limbs (55 of 72, 76%) or axial skeleton (6 of 72, 8%), or severe non-fracture musculoskeletal injuries (3 of 72, 4%), suggesting that injuries among the majority of these horses were likely associated with exercise. Significant lesions in the remaining 8 horses with unknown exercise history included severe pulmonary hemorrhage compatible with EIPH (3), hemoabdomen (2), cervical vertebral stenosis (1), mesenteric torsion (1), and typhlocolitis (1).
Death was unassociated with racing or training in the remaining 377 of 963 (39%) cases, the majority of which were SB (277 of 377, 73%). Lesions considered significant to the cause of death involved a range of organ systems including GI, CNS, musculoskeletal, respiratory, CV, and renal systems. GI lesions were most frequently identified in this group, contributing to 127 of 377 (34%) non–exercise related deaths. Enteritis and/or colitis (55 of 127, 43%) and intestinal accident (36 of 127, 28%) accounted for the majority of GI lesions. No lesions indicative of the cause of death were identified in 30 of 377 (8%) horses whose death was not related to racing or training. Toxicology analysis of PM blood samples was carried out in 7 of these cases, with identification of various stimulants potentially contributing to the death of 2 horses. Strychnine toxicosis was confirmed by toxicology testing as the cause of death in 1 horse.
Death was associated with drug injection in 38 of 377 (10%) horses whose deaths were unrelated to exercise, and in 38 of 963 (4%) of all horses in the study. Severe clinical reaction to drug administration led to euthanasia of 3 additional horses. In all but 1 of these 41 injection-associated cases, collapse followed by seizure or death of the horse occurred during or immediately after injection. Intravenous drug administration was described in 14 of 41 (34%) cases; the route of drug injection was not provided for the remaining 27 cases. Numerous drugs or drug combinations were associated with suspected drug reactions and death, including amino acid supplements, antibiotics (penicillin, trimethoprim–sulfadiazine, gentamicin), unspecified iron preparations, dimethyl sulfoxide (DMSO), mannitol, vitamin E–selenium preparations, other nonspecified vitamins, testosterone, other anabolic steroids, estrogen, calcium, pentosane polysulfate, Sarapin (High Chemical, Levittown, PA), trichlormethiazide–dexamethasone acetate, n-butyl alcohol, and prednisone. No PM lesions indicative of a definitive cause of death were identified in the horses. In 16 of 41 (39%) horses with a history of injection-associated death, pulmonary edema and/or congestion with variable hemorrhage were identified PM; multi-organ congestion was present in 3 of 41 (7%) horses; and laryngeal edema was evident in 1 horse. Cerebral edema, malacia, and vasculitis in 1 horse that died following drug administration were considered to have resulted from intracarotid injection. Septicemia and death was confirmed or suspected to be the result of contaminated intravenous solutions in 3 additional cases.
Discussion
The horseracing industry contributes significantly to the economy of many geographic regions worldwide. Few other spectator sports depend so intrinsically on animal participation. The risk of injury among racehorses is well recognized and has been documented.3,33 In order to identify factors predisposing to injury and death in racehorses, and to improve the health status and welfare of these animals, racing bodies in some jurisdictions have instituted a requirement for standardized PM to investigate fatalities.22,30 Racehorse PM studies to date have predominately involved TB racehorses, with inclusion of QH in a few reports.3,14,15,18,22 Horseracing in Ontario, Canada is unique in that TB, SB, and QH all contribute to the racing industry in this province. The ORC Death Registry provides information on the cause of fatalities among individuals of each breed. Following the introduction of the Death Registry in 2003, PMs of Ontario racehorses have contributed to a significant number of all equine PM cases at the AHL (21%), and have provided valuable data for multiple equine research projects at the Ontario Veterinary College, University of Guelph. Our retrospective evaluation of Death Registry cases provides a comprehensive summary of PM findings in a large number of racehorses, and includes information on SB racehorse fatalities.
Limb fracture was the single most common lesion causally associated with death among all of the racehorses in our study, and was the most common diagnosis overall, involving 39% of horses for which PM was carried out. Distribution of fracture sites in TB horses was similar to that described in other reports, with fractures most frequently involving bones of the fetlock joint or carpus.11,26 In contrast, P1 was the most common fracture site in SB. Predominance of limb fracture as the cause of race-related fatality has been described in studies of TB racehorses in the United States and Australia.3,18 An accurate prevalence rate for limb fractures in general, and for fractures at specific anatomic sites, among Ontario racehorses cannot be determined from our study given the discretionary requirement for PM in the study population, with PM of only 56% of horses reported to the Death Registry. However, the results suggest that these features are likely similar among Ontario TB racehorses to those described in other regions.
Sudden death has been defined as acute collapse and death, either during or immediately after a race, in an apparently healthy horse, 23 and sudden death in TB racehorses is recognized worldwide.3,4,14,15,18,22,23 Both racing- and training-associated sudden death were included as a category in our study. Sudden death was described in the clinical history of 31% of horses in which death was causally associated with these activities, and in 16% of all horses in the study. Unlike the situation involving fracture cases, PM was completed on all Death Registry horses with a history of sudden death. As a result, the proportional mortality rate of 9% (157 of 1,708 total Death Registry cases) for exercise-associated sudden death among Ontario racehorses is considered accurate, with an approximately equal rate of sudden death among TB, SB, and QH populations. In comparison, proportional mortality rates for sudden death of 19% among TB in flat races in Australia, and 4% in TB in the United States, have been documented.3,22
Similar to other published results, 22 cardiopulmonary lesions were considered the cause of death in the majority of exercise-associated sudden death cases in our study, and pulmonary hemorrhage was the most common lesion identified overall in these cases. In conjunction with a history of exercise, the lesion was considered by pathologists in the majority of these cases to be consistent with EIPH. Significant pulmonary hemorrhage, whether interpreted to represent EIPH or other cause of hemorrhage, was more frequently identified among SB than in TB with exercise-associated sudden death, despite the approximately equal number of horses of each breed among these cases. In other studies, sudden death among TB has also been attributed to pulmonary hemorrhage.4,15,22 Subjectivity is recognized in the PM definition and interpretation of pulmonary hemorrhage both in our study and in others. 22 In addition, lung autolysis is common in racehorse PM cases and may be severe, interfering with interpretation of both gross and histologic lesions in lung. A 2015 consensus statement of the American College of Veterinary Internal Medicine (ACVIM) suggests that minimal evidence exists in the literature for a clear association between EIPH and an increased risk of sudden death in racehorses. 17 “Equine exercise-associated fatal pulmonary hemorrhage” has been presented as a more accurate term to categorize these lesions, the etiology of which is not well understood. 6 Acute cardiac failure or vascular rupture are suggested causes. Fatal pulmonary hemorrhage and EIPH could potentially occur together in sudden death cases. 6 Adoption by pathologists of well-defined criteria for describing pulmonary lesions in racehorse sudden death cases may aid in better defining this condition. These could include consistent inclusion in the pathology report of the gross distribution of lung lesions (hemorrhage) and percentage of lung parenchyma involved, and histologic examination of standard sections from multiple regions of lung to correlate gross and histologic distribution of lesions. Consistent PM evaluation for the presence and anatomic distribution of histologic lesions associated with EIPH could also support contribution of this condition in cases of fatal pulmonary hemorrhage. These lesions include pleural, interlobular septal, and interstitial fibrosis; regional remodeling of pulmonary veins with fibrosis; and hemosiderophage accumulation in pulmonary interstitium and airway lumens.6,9,24,25,32 Standardization of reporting for pulmonary and other lesions in racehorse PM cases would facilitate direct comparison of results between and among racehorse populations.
The cause of death was undetermined because of the absence of significant PM lesions in 20% of exercise-associated sudden death cases, similar to results of a 2011 study. 22 Spontaneous, fatal cardiac arrhythmia has been speculated as the likely underlying condition in sudden death cases for which no lesions are identified. 22 Fatal arrhythmia is also suggested in some sudden death cases in which minimal inflammatory or fibrotic lesions are identified microscopically in myocardium. 20 However, confirming the significance of these lesions is not possible after death, and death ascribed to this cause remains presumptive. In humans, sudden death during exercise is often attributed to fatal arrhythmia or to previously undetected congenital cardiac structural anomalies. 13 Significant and potentially fatal cardiac arrhythmias have been identified in racing SB horses in the period immediately following racing.27,29 Cardiac arrhythmias have also been associated with treadmill exercise in TB, 19 and in some horses experiencing non-fatal episodic collapse. 21 It has been hypothesized that, in racehorses, acute heart failure and pulmonary hypertension resulting from post-exercise arrhythmia may contribute to pulmonary hemorrhage, cardiopulmonary failure, and sudden death. 27
Cardiac tamponade resulting from hemopericardium, and blood-loss anemia or shock resulting from hemoabdomen and/or hemothorax were the most frequent CV lesions contributing to death in exercise-associated sudden death cases. A definitive site of aortic rupture was identified in 87% of horses with cardiac tamponade; however, the source of hemorrhage was confirmed in only 54% of cases with body cavity hemorrhage. A 2015 study has suggested an association between anticoagulant rodenticides detected in trace amounts in liver of racehorses, and exercise-associated sudden death caused by massive body cavity hemorrhage. 5 Potentiation of the effect of these anticoagulants by other drugs or by other, unknown factors has been suggested. 5 Toxicology testing for anticoagulant rodenticides was not carried out on any of the horses in our study; however, this should be considered for cases in which there is significant hemorrhage in the absence of obvious vascular rupture. Aortic rupture is a well-recognized condition in horses and is presumed to result from increased aortic pressure during strenuous physical activity. 28 A 2008 report evaluating variably severe, localized mural mineralization in pulmonary artery and other large blood vessels suggests that the lesion may predispose to vascular rupture at affected sites. 1 In pulmonary artery, mineralized foci are associated with mural stress as a result of the physical shape of the artery and increased pulmonary luminal pressure at these sites. 31 Some sites of vascular mineralization may be detectable only microscopically, 1 emphasizing the importance of histologic examination of the vascular wall at sites of rupture. In a single horse in our study with pulmonary hemorrhage, focal mural mineralization was adjacent to a site of pulmonary artery aneurysm; however, a definitive site of vascular rupture was not identified.
The death of 4% of horses in our study was associated with parenteral administration of various pharmaceuticals or supplements. Although the number of affected horses is fewer compared to the number of fatalities caused by fracture or exercise-associated sudden death, these deaths are of concern and potentially easily preventable. A clinical history of collapse and death during or immediately after injection was provided for many of these adverse drug reactions and is compatible with type I hypersensitivity (anaphylaxis) or an anaphylactoid reaction triggered by the injected substance. 10 In horses, anaphylaxis is often clinically expressed as hypotension and collapse, although bronchoconstriction, laryngeal and pharyngeal edema, and cardiac dysrhythmia may also contribute to cardiopulmonary arrest and/or asphyxia.8,10 Inadvertent intracarotid injection of a drug or supplement, or massive endotoxemia resulting from contaminated solutions, are other potential explanations for rapid injection-associated death. As part of the PM protocol used in our study in 2008–2015, carotid arteries and pericarotid soft tissue were routinely examined for evidence of recent hemorrhage or puncture supportive of intracarotid injection. Intracarotid injection was not confirmed in any horses in our study, although CNS lesions of malacia and vasculitis in 1 horse that survived for 24 h after drug administration were suspected to reflect cerebral vascular spasm associated with intracarotid drug delivery. Sepsis resulting from injection of contaminated medications or supplements also contributed to death and euthanasia of several horses in our study, and has been described previously. 16
The frequency of PM lesions related to other body systems in both euthanized and non-euthanized horses in our study is similar to that described in PM surveys of the general equine population. 2 This suggests that with the exception of catastrophic musculoskeletal injuries requiring euthanasia, exercise-associated sudden death, and injection-associated deaths, factors contributing to death among racehorses are similar to those in other equine populations.
This descriptive study provides baseline information regarding the cause of death and PM lesions among SB, TB, and QH racehorses in Ontario. Limb fracture and exercise-associated sudden death were the most common reasons for euthanasia or death, respectively, among both SB and TB in Ontario. This is in agreement with previous studies of the cause of death in racing TB, but has not been documented previously in SB, to our knowledge. PM is especially important for evaluation and understanding of cases of sudden death in exercising horses, many of which may be the result of arrhythmia and acute cardiac failure. Adverse reactions and sepsis resulting from drug injection also contribute to death among racehorses. Other causes of death and lesions in this population are similar to those in the general equine population. Standardization of reporting of pulmonary and other lesions in racehorse PM cases would facilitate direct comparison of results between and among racehorse populations.
Footnotes
Acknowledgements
The following pathologists performed postmortem examinations on cases included in this study: Murray Hazlett, Peter Lusis, Tony van Dreumel, Beverly McEwen, Margaret Stalker, Maria Spinato, Janet Shapiro, Gary Thomson, Sameh Youssef, Andrew Vince, Kate Welch, Gaylan Josephson, Brian Binnington, Andrew Brooks, Phil Watson, Grant Maxie, Brandon Lillie, Robert Foster, Fannie Dessureault, Susan Newbigging, Noel Harrington, Tara Arndt, Patricia Turner, and Josepha DeLay. Drs. Bruce Duncan and Adam Chambers of the Alcohol and Gaming Commission of Ontario provided data and additional information regarding the ORC (AGCO) Death Registry program. This manuscript is dedicated to the memories of Dr. Kate Welch and Dr. Peter Lusis, our colleagues and friends.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
