Abstract
In racehorses, sudden death (SD) associated with exercise poses a serious risk to jockeys and adversely affects racehorse welfare and the public perception of horse racing. In a majority of cases of exercise-associated sudden death (EASD), there are no gross lesions to explain the cause of death, and an examination of the cardiovascular system and a toxicologic screen are warranted. Cases of EASD without gross lesions are often presumed to be sudden cardiac deaths (SCD). We describe an equine SD autopsy protocol, with emphasis on histologic examination of the heart (“cardiac histology protocol”) and a description of the toxicologic screen performed in racehorses in California. By consistently utilizing this standardized autopsy and cardiac histology protocol, the results and conclusions from postmortem examinations will be easier to compare within and across institutions over time. The generation of consistent, reliable, and comparable multi-institutional data is essential to improving the understanding of the cause(s) and pathogenesis of equine SD, including EASD and SCD.
Introduction
Sudden death (SD) associated with exercise in racehorses is a sporadic event,4,27 but when it occurs, poses a serious health risk to jockeys, and adversely affects racehorse welfare and the public perception of horse racing. SD is the death of a closely observed, apparently healthy animal, in which no clinical signs have previously been observed. 26 The term “exercise-associated sudden death” (EASD) in our report refers to cases of SD that occur during exercise or within 1 h post-exercise. 27 The term “sudden cardiac death” (SCD) refers to those cases of SD or EASD in which there is no apparent reason or cause of death during the postmortem examination and histologic examination of extracardiac tissues. 27 These cases of SCD are then “autopsy-negative” as there are no lesions to explain the cause of death. Numerous studies have recorded postmortem findings in cases of SD or EASD in a variety of equine populations.5,6,11,13,20,23,24,27,35 Recorded causes of SD or EASD include exercise-induced pulmonary hemorrhage, pulmonary edema and hemorrhage, massive thoracic and/or abdominal bleedings, cardiac tamponade as a result of aortic rupture, skull or cervical neck fractures, myocardial fibrosis, and arteriosclerosis. Such studies have various autopsy and histologic sampling methods, and their results and interpretations of the findings are inconsistent. As a result, valid conclusions about the underlying events that lead to EASD and SCD are difficult to attain and, therefore, the pathogenesis and possible cause(s) of EASD and SCD in racehorses are largely unclear.
We describe herein the autopsy protocol used for cases of SD and EASD at the California Animal Health and Food Safety Laboratory System–University of California, Davis, with special emphasis on the sampling protocol for the histologic examination of the heart (“cardiac histology protocol”). The 3 main goals of this autopsy protocol are 1) to perform a postmortem examination of each case to try to establish the cause of EASD, 2) to standardize the histologic examination of the heart in cases of EASD so that results can be compared over time and between institutions, and 3) to describe the toxicology screens performed at our institution on cases of EASD. The protocol was developed based on information from veterinary textbooks,28,37 journal literature,3,7,15,16,39,40 conference proceedings (Maxie G. Cardiovascular necropsy of domestic mammals. Proc ACVP annual meeting; 1991 Dec 10–15; Orlando, FL; Kiryu K. Cardiopathology of arrhythmias in the horse. Proc 26th Am Assoc Eq Pract; 1981 Nov 30–Dec 3; Anaheim, CA), and the expertise of several veterinary anatomic pathologists involved with the autopsy of SD and EASD racehorses over the years.
Postmortem procedure
The autopsy technique will vary depending on personal and institutional preference but, in all instances, a systematic examination of all visceral organs, including the brain, must be performed. In addition, the skull and neck are carefully examined for fatal traumatic injury during a fall. When there is evidence of significant skull or neck trauma to explain the cause of EASD, further examination of the front and hind limbs is performed looking for acute fractures or other articular, ligament, or tendon lesions that may have precipitated the fatal fall. The possibility of acute pelvic fractures must also be investigated. When a severe pelvic fracture occurs, there is often abundant fluid and/or clotted blood present in the large muscular masses surrounding the pelvis, the pelvic cavity, the retroperitoneum, and/or the abdominal cavity. If the cause of death is not apparent during the autopsy (“autopsy-negative cases”), a cardiac examination and histologic sampling is performed, as described in the specific section below. Histopathology samples routinely include brain, heart, lungs, liver, spleen, kidneys, adrenal glands, skeletal muscle, stomach, and gastrointestinal tract. Additional histologic samples, as well as samples for other disciplines, such as bacteriology, virology, parasitology, and immunology are collected at the discretion of the individual pathologist. Samples for toxicology tests are routinely collected, as explained in the specific section below.
Cardiac autopsy protocol
The approach to the gross examination and dissection of the heart in animals varies according to personal or institutional preference and the literature consulted. It is beyond the scope of this review to describe the different methods of heart dissection in animals, which are available elsewhere (Maxie G. Cardiovascular necropsy of domestic mammals).7,22,37 Regardless of the heart dissection method chosen, all of the major components of the heart, including the pericardium, myocardium, mural and valvular endocardium, great vessels, right and left coronary arteries, and the regions where the main components of the conduction system are located must be carefully examined for gross abnormalities (e.g., changes in color, hemorrhage, fibrosis, valvular thickening, narrowing of coronary arteries, etc.), even when, in our experience, the vast majority of hearts from racehorses are grossly normal. The intact heart and body weight ratios have been reported by other authors.14,37 However, this information seems to be of limited value unless standardized control values are available for racehorses. Moreover, there is evidence that the size and weight of the equine heart varies depending on a number of variables, including, but not limited to, age, breed, sex, and type of training.8,14,42
The histologic sampling protocol of the heart is composed of 11 routine samples (Table 1) plus any areas with gross abnormalities. Section 1 includes the right ventricular free wall, with the right atrial wall, the right coronary artery, and the parietal cusp of the tricuspid valve (Fig. 1). Section 2 includes the pulmonary artery semilunar valve with the right ventricular outflow tract and the pulmonary artery (Fig. 2). Section 3 is a slice of the right atrial appendage (Fig. 3). Section 4 includes the sinoatrial (SA) node region. The SA node is located in the subepicardial region of the superior border of the terminal sulcus (sulcus terminalis) at the junction of the cranial vena cava and the right atrium (Fig. 4a, 4b). The terminal sulcus is the crease that separates the right atrium and adjacent cranial vena cava, and the superior border corresponds internally with the terminal crest (crista terminalis).10,37 Locating the SA node may be problematic (Fig. 4a). To increase the chances of finding the SA node, a ~2 cm wide by 5 cm tall slice that includes the superior border of the terminal sulcus, the terminal crest, the cranial vena cava, and the right auricle must be collected and subsequently bread-sliced perpendicular to the terminal sulcus into 5–6 sections (Fig. 4b). Section 5 includes the left atrial appendage (Fig. 5). Figure 6 shows sections 6–8. Section 6 includes the left ventricular free wall, left atrial wall, left coronary artery, and the parietal cusp of the left atrioventricular valve. Sections 7 and 8 include a portion of each left ventricular papillary muscle. Section 9 includes the atrioventricular (AV) node region (Fig. 7). The AV node is smaller than the SA node and lies subendocardially within the right atrial wall, just cranial to the orifice of the coronary sinus and ~6 mm above the attachment of the septal cusp of the right atrioventricular (tricuspid) valve. It has a flattened, oblong shape, is ~15 mm in depth and 7 mm wide in the adult horse, and is overlaid by ~1–2 mm of subendocardial atrial myocytes. 3 Locating the AV node can also be problematic, possibly even more so than the SA node. To increase the chances of finding this node and associated conduction pathways, a ~3–4 cm wide by 3 cm tall portion of the general area described above and shown in Figure 9 must be collected and subsequently bread-sliced into 4–5 sections. Section 10 includes any portion of the interventricular septum with 1 endocardial surface (the average equine interventricular septum is too thick to fit in a single histology cassette; Fig. 8). Section 11 includes a portion of the aortic semilunar valve, aorta, and left ventricular outflow tract (Fig. 9). Additional sections of heart can be added if gross lesions are observed in other sites or at the discretion of the veterinarian performing the autopsy.
Sections collected for the histologic examination of the equine heart in cases of racehorse sudden death.

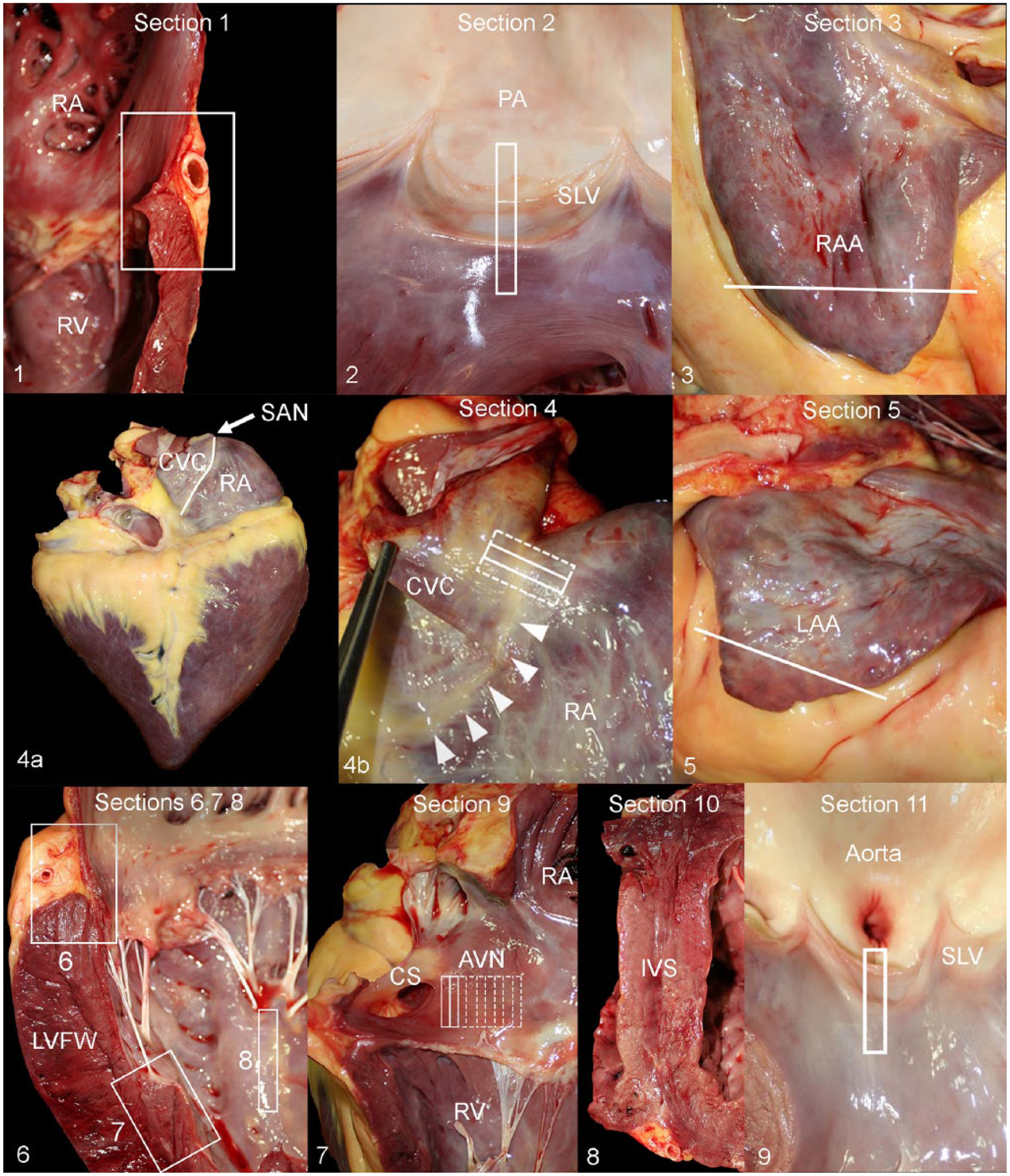
Histologic sampling protocol for enhanced examination of the equine heart in cases of sudden cardiac death.
The SA and AV nodes are relatively easily recognized microscopically in tissue sections stained with hematoxylin and eosin, as the histology of nodal conduction cardiomyocytes is different from that of the adjacent, contractile atrial cardiomyocytes. The SA node can be recognized at low power at the junction of the cranial vena cava and atrial myocardium based on the characteristic histologic appearance of the nodal cardiomyocytes and the presence of 1 or more nodal arteries within the node (Fig. 10). As the body of the node tapers cranially into the cranial crus and caudally into the caudal crus, nodal arteries are not found within the node. The node occupies approximately two-thirds of the wall at the cranial vena cava–right atrial junction. 3 The SA node is composed of irregularly arranged, interwoven, thin, branching cardiomyocytes embedded in an abundant network of connective tissue (Fig. 11). Nodal cardiomyocytes are often less eosinophilic than contractile atrial cardiomyocytes, have less prominent cross-striations, no intercalated discs, and some individual cardiomyocytes resemble Purkinje cells (Fig. 12). Toward the margins of the node, long strands of nodal cardiomyocytes run between and connect with contractile atrial cardiomyocytes (Fig. 13).3,10,19,28,39
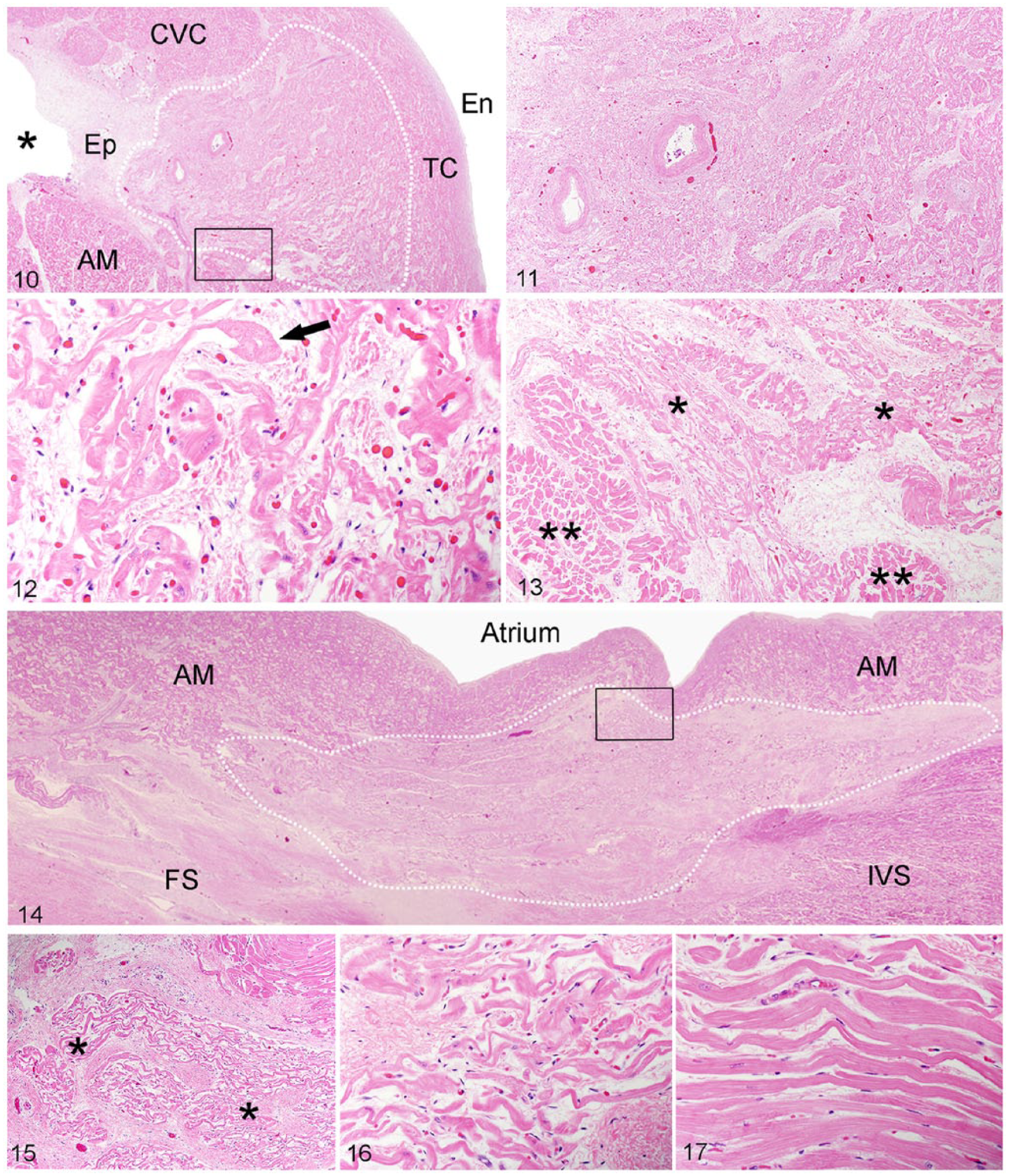
Histology of the sinoatrial node (SAN). H&E.
The AV node is slightly more difficult to detect at low power than the SA node, partly because of the lack of large nodal arteries within the node. The AV node has an oblong, flattened shape and lies subendocardially surrounded by right atrium and interventricular septum cardiomyocytes and the fibrous septum (Fig. 14). Similar to the SA node, AV node cardiomyocytes are haphazardly arranged, interconnected, thinner, and smaller than atrial contractile cardiomyocytes, have fewer cross-striations, no intercalated discs, and are supported by abundant connective tissue (Figs. 15–17). The AV node continues cranially and drops into the fibrous septum just below the cardiac cartilage, where it transitions into the atrioventricular (or common) bundle (AVB). As the AV node transitions into the AVB, cardiomyocytes become larger and more widely separated by the collagen of the fibrous septum, have less branching, and are less tortuous than in the AVN and, albeit smaller, have some semblance of the specialized Purkinje cells observed in the downstream portions of the conduction system (Fig. 18).19,28,40
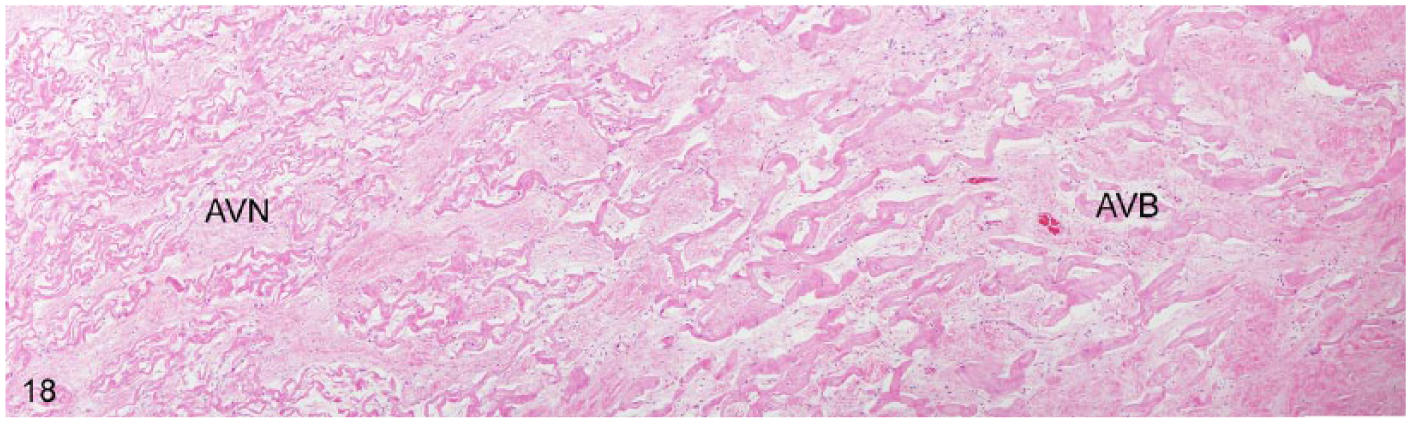
Atrioventricular node (AVN) to atrioventricular bundle (AVB) transition. From left to right, cardiomyocytes gradually become larger, more widely separated by collagen, are less branching and tortuous, and resemble Purkinje cells.
Toxicology testing
Toxicology testing in cases of SD is challenging because, in the majority of cases, there is no history of specific toxicant exposure prior to death. In some cases, specific testing might be directed by anecdotal history of use of specific substances, presence of postmortem lesions compatible with specific chemical exposures, or an a priori hypothesis regarding the role of a particular chemical in causing SD. In such cases, the samples analyzed and the analytical methodology employed is driven by the kinetics or chemical properties of the toxicant of interest. For example, in some SD cases, large amounts of unclotted blood have been found on postmortem examination without any evidence of a cause. In such cases, liver samples have been tested and found positive for low concentrations of anticoagulant rodenticides (ARs). 9 This analysis was developed and optimized for sensitivity specifically for commonly available ARs. However, in most cases, toxicology testing is directed toward searching for “unknowns.” The latter approach requires using analytical methods that are designed to extract and analyze chemicals with diverse chemical characteristics.
Because information regarding kinetics is not available when searching for unknowns, the selection of appropriate samples for testing can present a challenge, particularly when resources are limited and multiple samples cannot be analyzed routinely. Samples for toxicologic testing typically focus on tissues involved in metabolism, accumulation, or elimination of foreign substances. Liver is the most common postmortem sample tested, given its initial exposure to, and metabolism of, diverse chemical classes. Other samples that might be useful for postmortem collection and testing include stomach contents, kidney, urine, available blood, and hair. Our current protocol calls for collecting several different samples, including liver, kidney, urine, aqueous humor, and stomach content, although liver is most frequently tested. Although not routinely collected at our laboratory, synovial fluid or synovial membrane could be of potential toxicologic interest for some laboratories or regulatory agencies, especially to investigate the presence of substances injected intra-articularly that may not reach detectable systemic levels. Samples are stored at −20°C until processed or discarded. Our laboratory currently does not have validated methods to test formalin-fixed (FF) tissues for toxic or prohibited substances. Two important limitations to testing FF tissues are the usually small sample size and the high risk of false-negative results, as there is currently not enough knowledge about what substances may be lost or masked during the tissue fixation process.
The sophistication of analytical systems has enabled diagnosticians to look for literally thousands of chemical compounds with a high degree of specificity and sensitivity. In cases in which there is no history of exposure to a specific chemical, powerful screening tools such as gas chromatography–mass spectrometry (GC-MS), liquid chromatography–mass spectrometry (LC-MS), inductively coupled argon–plasma emission spectroscopy (ICP-AES), and inductively coupled plasma–mass spectrometry (ICP-MS) are available to detect a broad array of organic and inorganic (metal) compounds. In general, GC-MS is designed to detect volatile, lower-molecular-weight organic chemicals, whereas LC-MS is designed to detect non-volatile, higher-molecular-weight organic chemicals. Although there is some overlap with regard to chemical detection, the combined use of both provides more comprehensive screening. An advantage of GC-MS is that mass spectra from unknowns can be compared with hundreds of thousands of compounds in commercial databases, whereas similar libraries for use with LC-MS are not available and have to be established by each laboratory. ICP-AES and ICP-MS techniques are designed to detect metals. Multiple metals can be detected with a single analysis by either technique using a variety of sample types and across a range of concentrations. Laboratories vary with regard to what metals are included in a typical screen. ICP-MS is considered to be more sensitive and is better suited for the detection of many trace elements. Because metals such as cobalt and arsenic are currently of interest in performance horses and are generally present at very low concentrations in liver tissue, we routinely screen postmortem liver samples using ICP-MS. Our current reporting limits for cobalt and arsenic in liver are 10 ppb and 50 ppb, respectively. Other metals included in our routine screening protocol include lead, manganese, iron, mercury, molybdenum, zinc, copper, cadmium, and selenium.
Discussion
In many cases of equine EASD, the cause is not apparent during autopsy and it is then generally presumed to be acute cardiac, cardiovascular, or cardiorespiratory failure. Therefore, an examination of the cardiovascular system is warranted. The equine SD autopsy protocol presented herein includes enhanced histologic examination of the heart and an extensive toxicologic screen. By consistently using this standardized protocol, the results and conclusions from the postmortem examinations at different institutions and over time within the same institution will be easier to compare. The future generation of consistent, reliable, and comparable multi-institutional data will hopefully contribute to improved understanding of the cause(s) and pathogenesis of equine SD, including EASD and SCD.
The majority of SD and EASD autopsies are performed in standard diagnostic laboratories. Therefore, this autopsy protocol was designed to be practical and easy to follow by busy pathologists with a diverse background and specialty focus. The 11 histologic sections of heart included in the enhanced cardiac histology examination aim at evaluating a representative, yet not excessive, number of samples that include relevant anatomic and histologic structures, such as the conduction system (SA and AV node regions), mural and valvular endocardium, epicardium, atrial and ventricular wall myocardium, coronary and pulmonary arteries and aorta.
The histologic examination of the conduction system is problematic, as there is considerable variability in the size, shape, and exact location of the sinus node among animal species and even within individuals of the same species. 30 Therefore, it is difficult to consistently locate and histologically identify the SA and AV nodes, for which some sections from the nodal regions may sometimes not show nodal cardiomyocytes or there may be just a small portion of the nodes.
The guidelines for the location and identification of the SA and AV nodes in our report are based on descriptions of the location of the SA and AV nodes in horses 3 and humans.1,10,19,29 Since the landmark study by Bishop and Cole in 1967, there have been no other detailed studies, to our knowledge, concerning the anatomy, morphology, and histology of the conduction system of the equine heart. Various authors have studied the anatomy and morphology of the conduction system in other mammalian and avian species including, but not limited to, cattle, 18 rabbits,17,33 cats, 34 dogs, 36 sheep, 38 goats, 31 pigs, 32 chickens,21,25 and camels. 12 Studies have shown that the conduction system has a different embryologic origin than that of the working myocardium, and that the system is more extensive than originally thought. The newly described structures of the conduction system include atrioventricular rings, a third node (retroaortic node), and pulmonary and aortic sleeves.10,41
Despite a gross and histologic examination of the heart, the results of the clinical and histologic examinations of the conduction system do not always correlate. For example, horses with diagnosed clinical arrhythmias and/or heart block may have no demonstrable histologic lesions in the conduction system or, vice versa, lesions may be present in the absence of clinical arrhythmia or abnormal conduction. 39 It is our experience that apparently healthy racehorses euthanized because of catastrophic musculoskeletal lesions often have mild, and sometimes moderate, lesions in the heart, including inflammatory lesions near the SA and AV nodes. This poses a great challenge to the pathologist investigating a case of SCD, because finding microscopic lesions in the heart does not necessarily confirm causality of death. Although one author describes a clinicopathologic correlation between atrial and atrioventricular junction fibrosis with either atrial fibrillation, SA block, or paroxysmal ventricular tachycardia, 24 the current scientific knowledge of SCD in racehorses seems insufficient to attain reliable clinicopathologic correlations. In the future, as we continue to gather clinical and pathology information from cases of SCD in racehorses, more conclusions may be drawn in this regard.
Based on our experience, most racehorses with SD or EASD have microscopic heart lesions, albeit usually minimal or mild. These lesions include inflammation of the myocardium, epicardium, endocardium, and valves, fibrosis, acute myocardial degeneration and necrosis, myocardial dropout, as well as miscellaneous lesions. However, it is very important to emphasize that the interpretation of the clinical significance of these lesions is very challenging, given that many horses without a history of SD or EASD that are euthanized because of catastrophic musculoskeletal injuries will have similar lesions. Characterization of microscopic lesions in hearts of racehorses with SD or EASD is ongoing, and detailed results will be available in the near future.
The cardiac histology protocol presented herein should serve as a baseline standardized protocol. As research on equine SCD continues, this protocol will likely evolve to adapt to the changing needs of the specialty, as well as to the different laboratories and veterinarians involved in the investigation of SCD in horses. In humans, the autopsy investigation of cardiac SD is often accompanied by the so-called “molecular autopsy,” which entails sampling for genetic testing to search for inherited arrhythmogenic disorders that are invisible grossly and histologically. 2 The cardiac molecular autopsy is not currently available for horses, but researchers have started working toward developing genetic tests to screen for potential inherited arrhythmogenic disorders that may lead to SCD in horses. This diagnostic tool will hopefully help to determine if inherited cardiac arrhythmogenic disorders may play an important role in horses, as they do in humans.
Toxicology testing is performed routinely on cases of EASD and SCD at our institution. Unfortunately, there is no single analytical technique or combination of techniques that can screen for every chemical. The goal is to cover as many chemical classes as possible. Often the trade-off between specific testing and general screening for organic compounds is the sensitivity of the test for a given chemical. Screening techniques are not optimized for a specific chemical or class of chemicals, and it is possible that very low concentrations of a chemical might not be detected by the screen. If nothing of interest is detected using our screening protocols, all we can say is that a chemical exposure could not be identified within the limits of the analytical procedures performed and the samples tested.
Footnotes
Acknowledgements
We thank all of the CAHFS pathologists who contributed to improving this protocol, and the autopsy, histology, and toxicology technicians for providing critical help during the study of cases of equine SD.
Authors’ contributions
SS Diab and FA Uzal contributed to conception and design of the study. R Poppenga contributed to conception of the study. All authors contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
