Abstract
Tube cystostomy is a surgical method used for managing obstructive urolithiasis and involves placement of a Foley catheter into the urinary bladder. We identified and evaluated the antibiotic resistance patterns of bacteria isolated from indwelling Foley catheters following tube cystostomy in goats with obstructive urolithiasis. Urine samples collected over a 10-y period from catheter tips at the time of removal were submitted for bacteriologic culture and antibiotic susceptibility testing. Resistance patterns to antibiotics, trends in the resistance patterns over the study period, and the probability of a bacterial isolate being resistant as a function of the identity of the isolate and antibiotic tested were determined. A total of 103 urine samples from 103 male goats with obstructive urolithiasis managed surgically with tube cystostomy were included in the study. Aerococcus (36.9%) and Enterococcus (30.1%) were isolated most frequently. The susceptibility patterns of all bacteria isolated did not change over the study period (p > 0.05). Proportions of isolates resistant to 1, 2, and ≥3 antibiotics were 36.9%, 18.5%, and 23.3%, respectively. Thus, 41.8% of bacterial isolates were resistant to 2 or more antibiotics tested. The probability of Aerococcus spp., Escherichia coli, and Pseudomonas aeruginosa isolates to be resistant to ampicillin, ceftiofur, erythromycin, penicillin, or tetracycline ranged from 0.59 to 0.76.
Obstructive urolithiasis is common in castrated male goats and is typically managed medically and/or surgically. Because of the relatively high success rate, tube cystostomy is one of the most commonly performed surgical procedures for management of this disease. 19 Tube cystostomy involves placement of a temporary indwelling Foley catheter into the urinary bladder through the abdominal wall so that continuous bladder emptying can occur, thereby helping to relieve urethral spasm and permitting reestablishment of normal urethral patency postobstruction. The average duration that a Foley catheter is maintained in the urinary bladder following placement is ~14 d. 19
In human patients that are managed with Foley catheters to treat obstructive urinary tract diseases, Foley catheter–associated urinary tract infections (CAUTI) can occur as a result of bacterial infections originating from patient skin, hospital personnel, and/or fecal contamination.1,5,17 Bacterial pathogens gain entry into the bladder through the catheter lumen or along the catheter–abdominal wall interface. 26 Additionally, the presence of an indwelling Foley catheter can disrupt the normal mechanical defense mechanism of bladder emptying in cases where the presence of the catheter causes bladder overdistension, incomplete voiding, and a residual urine volume that can support bacterial growth. 8 In humans, sequelae to CAUTI range from mild (urethritis and cystitis) to severe (pyelonephritis, renal scarring, urolith formation, and bacteremia). 13 If left untreated, infections secondary to CAUTI can lead to urosepsis and death. 27 Although there are no peer-reviewed studies in goats, it is anticipated that similar complications secondary to CAUTI will be observed in goats.
As a result of potential complications associated with CAUTI following placement of Foley catheters into goat bladders, administration of pre- and postsurgical prophylactic antibiotics that concentrate in urine is performed routinely. However, in our experience, goat patients are still predisposed to CAUTI. Although not proven for isolates from goats, bacterial pathogens involved in CAUTI in humans are more likely to be multidrug resistant because of the formation of biofilms on the catheter. 9 Bacteria within biofilms are able to survive and proliferate in the presence of therapeutic antibiotic concentrations. 4
The objectives of our study were: 1) to identify the types of bacteria isolated from indwelling Foley catheters at the time of removal following tube cystostomy to manage urolithiasis and 2) to characterize the antibiotic resistance patterns of the bacterial isolates. Information on common caprine isolates from Foley catheters, antibiotic sensitivity, and resistance patterns should be useful to veterinarians when managing pre- and postsurgical antibiotic therapy in goats.
Medical records of goats that were presented to the University of California–Davis, William R. Pritchard Veterinary Medical Teaching Hospital (UCD-VMTH) for obstructive urolithiasis and that underwent tube cystostomy from January 2000 to December 2010 as the only major surgical procedure to manage urolithiasis were selected. Information obtained from the medical records included signalment (breed, age, and use) and antibiotics administered pre– and post–tube cystostomy. Breed was classified as meat/dairy (Alpine, Boer, Cashmere, Lamancha, Nubian, Oberhasli, San Clemente, Spanish, and Toggenburg), dwarf (pygmy or dwarf Nigerian), or other (Tennessee fainting). Because of the possibility of recurrence of urolithiasis following surgical or medical management, cases were considered only for the first tube cystostomy procedure. At the time of Foley catheter removal, urine samples were collected from the inside and outside of the Foley catheter tip using a sterile swab. At the time of Foley catheter removal, no reported postcystostomy complications were observed, based on history and clinical examination. Urine samples were submitted for microbiologic aerobic and anaerobic culture and susceptibility testing at the time of collection. Sample analysis was performed at the UCD-VMTH Microbiology Diagnostic Laboratory.
Swabs were used to inoculate 5% defibrinated sheep blood (Hardy Diagnostics, Santa Maria, CA) and MacConkey agars (Hardy Diagnostics). Inoculated agars were incubated at 35°C in 5% CO2. Bacteria were identified on the basis of Gram stain, results from catalase testing, indole and oxidase spot testing, fermentative patterns (API 20E, API 20NE, and API 20 Strep; BioMerieux, Durham, NC), and biochemical testing including triple sugar iron (Biological Media Services, University of California, Davis, CA), Christensen urea (Biological Media Services), Simmons citrate (Biological Media Services), or identification strips (BioMerieux). Susceptibility testing was performed through a broth microdilution method (Sensititre, Thermo Fisher Scientific, Pittsburg, PA). Interpretation of test results was performed following the guidelines of the Clinical Laboratory Standards Institute (CLSI).2,3 Briefly, a few isolated colonies were used to inoculate 2 mL of brain–heart infusion broth and incubated at 35°C without CO2 for 2–6 h. Broth culture was added dropwise to 0.85% NaCl to achieve a turbidity equivalent to a 0.5 McFarland nephelometer standard. Ten microliters of this suspension were added to cation-adjusted Mueller–Hinton broth, and plates (Thermo Fisher Scientific) were inoculated with 50 μL of broth per well. Plates were then incubated at 35°C without CO2 overnight, and a minimum inhibitory concentration (MIC) was determined for each antibiotic. Quality control and quality assurance of test result interpretation was based on the CLSI guidelines.2,3 Interpretation of susceptibility of isolates was determined through CLSI guidelines using ruminant interpretations when available and human guidelines when ruminant guidelines were not.2,3 To date, no CLSI guidelines for interpretation exist for some of the bacteria that were isolated, and, consequently, veterinary Streptococcus breakpoints for Aerococcus, human Pseudomonas breakpoints for Flavobacterium, and ruminant Pasteurellaceae breakpoints for nonfermenting group 3 gram-negative bacteria were applied.
Data were checked for normality using the Shapiro–Wilk test. Means and medians were reported for parametric and nonparametric data, respectively. Descriptive statistics were used to summarize data. For the purposes of our study, antibiotic susceptibilities reported as intermediate were classified as resistant. To investigate the association between resistance patterns by bacterial isolates and antibiotics, a χ 2 test was performed. To assess whether there was a trend (increase in resistance patterns) by different bacterial isolates during the study period from 2000 to 2010, a Cochran–Armitage test was performed. For the Cochran–Armitage analysis, the year periods were 2000, 2001, and 2003; 2005–2007; and 2008–2010.
A follow-up logistic regression was performed to predict the probability of an isolate being resistant as a function of its identity and antibiotic tested. In the logistic regression, the dependent variable was defined according to the bacterial isolate’s resistance to an antibiotic. Independent variables considered were the identity of the bacterial pathogen isolated and the antibiotic tested. Initial p to enter into the logistic regression model was set at 0.1 but final comparison for variables to be included into the model was set at p < 0.05. Goodness-of-fit of the final model was assessed using likelihood ratios. All analyses were performed using Proc Genmod SAS v. 9.4 (SAS Institute, Cary, NC).
Urine samples from 103 male goats were submitted for microbiologic testing. Eight, 7, 2, 5, 3, 36, 31, 8, and 3 samples were submitted in 2000, 2001, 2003, 2005, 2006, 2007, 2008, 2009, and 2010, respectively. No samples were submitted in 2002 and 2004. Forty-eight samples (46.6%) were collected from meat/milk dairy breeds, 38 (36.9%) from dwarf breeds, 2 (1.9%) from mixed breeds, and 2 (1.9%) from other breeds. The breed was not reported for 13 samples (12.7%). The animal age was recorded in samples from 86 goats and not reported for 17 goats. The median (range) reported age was 3 y (3 mo to 7 y). Samples were collected from 89 castrated goats but information on whether a patient was castrated was missing for 14 goats. The antibiotic administered pre– and post–tube cystostomy was reported for 90 goats and not recorded in 13. When considering only cases for which the antibiotic administered was recorded, third-generation cephalosporins were administered in 61 (67.8%) goats. Thirteen (14.5%), 12 (13.3%), 2 (2.2%), 1 (1.1%), and 1 (1.1%) goats were administered gentamicin, florfenicol, ampicillin, penicillin, and tulathromycin, respectively. Mean ± standard deviation time to removal of Foley catheters was 14.7 ± 0.9 d.
All submitted samples had a single significant bacterial genus. Species of bacteria isolated were Actinobacillus spp., Aerococcus (viridans, and other species), Enterococcus (durans, faecalis, faecium, gallinarum, and other species), Escherichia coli, Flavobacterium, nonfermenting group 3 gram-negative bacteria, Pseudomonas aeruginosa, Staphylococcus (aureus and coagulase-negative species), and Streptococcus (bovis, viridans, and other species). Proportions of genera of bacteria isolated from urine samples are summarized in Table 1. As a result of the small number of isolates identified, Actinobacillus, Flavobacterium, and nonfermenting group 3 gram-negative bacteria species were not included in the χ2 and logistic regression analyses. Susceptibility test results of P. aeruginosa to ampicillin, erythromycin, and penicillin were excluded in the data analysis.
Bacterial isolates identified and relative proportions from male goat urine samples collected from Foley catheters (n = 103).
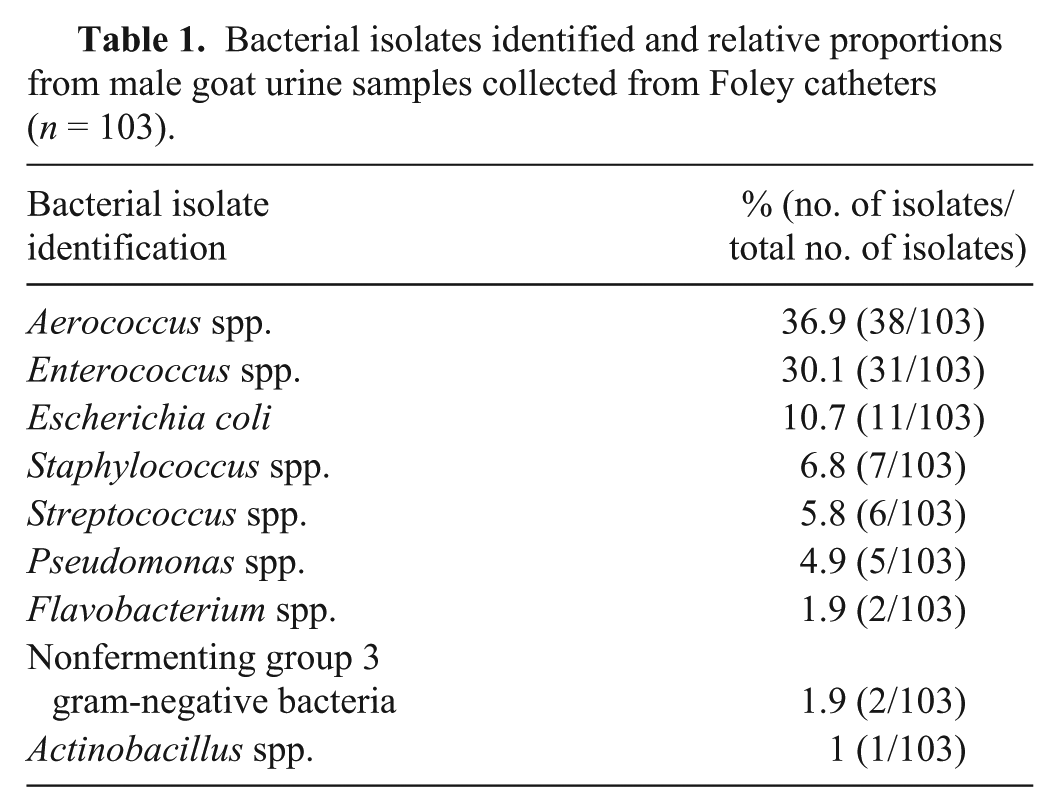
All bacterial isolates were tested for sensitivity to amoxicillin–clavulanate, ampicillin, ceftiofur, gentamicin, erythromycin, penicillin, and tetracyclines. Twenty-two isolates (21.3%) were susceptible to all antibiotics tested. Proportions of isolates resistant to 1, 2, or ≥3 antibiotics tested were 36.9% (38 of 103), 18.5% (19 of 103) and 23.3% (24 of 103), respectively. Thus, 41.8% of bacterial isolates were resistant to 2 or more antibiotics tested. There was no evidence for increased trends in bacterial resistance from 2000 to 2010 (p = 0.194).
Results of the logistic regression predicting the probability of an isolate being resistant as a function of the isolate identified and antibiotic tested are summarized in Table 2. Aerococcus spp., E. coli, and P. aeruginosa isolates were significant predictors of resistance to an antibiotic (p < 0.05; Table 2). Ampicillin, ceftiofur, gentamicin, tetracycline, and penicillin were significant predictors of resistance when tested on the isolates (p < 0.05; Table 2). Probabilities of an isolate being resistant as a function of its identity and the antibiotic tested derived from the logistic regression are summarized in Table 3.
Probability of an isolate being resistant as a function of the identity of the isolate and the antibiotic tested in male goat urine samples (n = 103).*
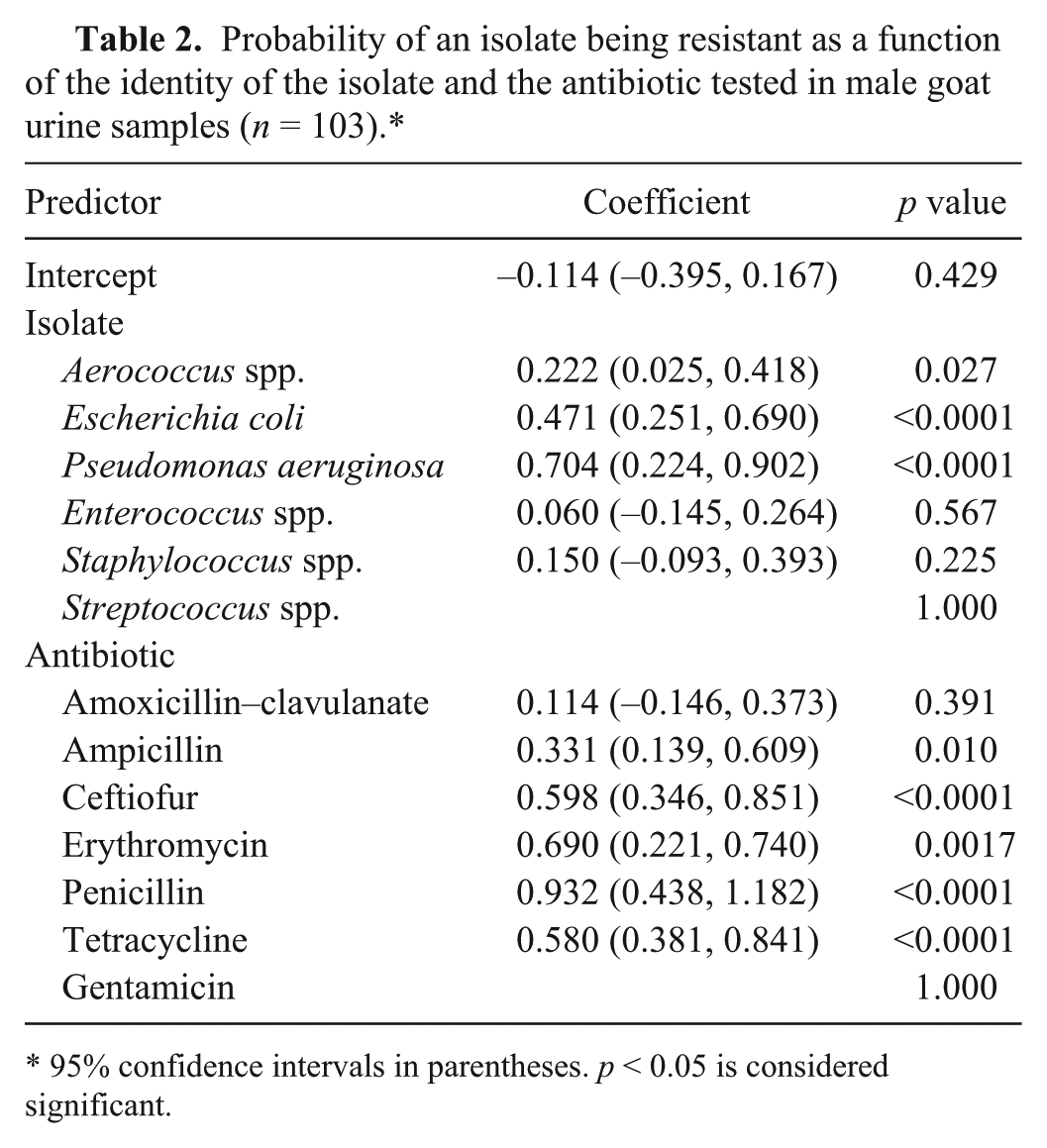
95% confidence intervals in parentheses. p < 0.05 is considered significant.
Probability of bacterial isolate resistance as a function of the identity of the isolate and the antibiotic tested in male goat urine samples (n =103).*

Only isolates and antibiotics that were significant (p < 0.05) in the logistic model were included.
Susceptibility not included because of intrinsic resistance by P. aeruginosa.
A major finding of our study was that the majority (78.7%) of pathogens isolated from urine collected from Foley catheter tips were resistant to at least 1 antibiotic. Additionally, 41.8% of the isolates were resistant to 2 or more antibiotics. Based on the logistic regression (Table 2), the probability that Aerococcus spp., E. coli, and P. aeruginosa were resistant to ampicillin, ceftiofur, erythromycin, penicillin, or tetracycline was relatively high (probability range: 0.59–0.76; Table 3). The findings suggest that the tested antibiotics are less likely to be effective (at least in vitro) against Aerococcus spp., E. coli, and P. aeruginosa isolates. It should be noted that P. aeruginosa is intrinsically resistant to ampicillin, penicillin, erythromycin, and extended spectrum cephalosporins, 10 and E. coli is less likely to be susceptible to penicillins or erythromycin. 25 The presence of resistant bacterial isolates from urine samples despite antibiotic administration prior to and after surgery suggests that goats are susceptible to CAUTI following tube cystostomy. Third-generation cephalosporins were the most frequently administered prophylactic antibiotic pre- and post-tube cystostomy. Reasons for a veterinarian to choose third-generation cephalosporins are likely to include the ability of the antibiotic to concentrate in urine, dosing frequency, and attending clinician preference. It should be noted that third-generation cephalosporins were prohibited for extra-label use in major food-producing species including, cattle, pigs, and poultry in April 2012 (U.S. Food and Drug Administration. Animal Medicinal Drug Use and Clarification Act of 1994 (AMDUCA). Available from: http://www.fda.gov). However, as goats are considered a minor species, the extra-label use of third-generation cephalosporins is not specifically prohibited in this species.
Another major finding of our study was that the majority of bacterial pathogens isolated from the Foley catheters consisted mainly of commensal bacteria of which Aerococcus spp. and Enterococcus spp. were isolated most frequently. Sources of Aerococcus spp. and Enterococcus spp. likely include skin, feces, the lower genitourinary tract, or the environment. 11 The major pathogens associated with CAUTI in humans include E. coli14,16 and Proteus.13,21 In our study, however, E. coli represented only 10.5% of the isolates. The presence of resistant gram-negative bacteria isolated from urine samples as found in our study population was expected because initiation of bacterial attachment to the epithelial surface of the cells of the urinary tract or to the Foley catheter requires the presence of bacterial adhesins. 13 Gram-negative bacteria produce various adhesins, including fimbriae, pili, and nonfimbrial adhesins. 13 Once attached to the cell surface epithelium or catheter, the bacteria produce exopolysaccharides that entrap and protect them. 13 Consequently, they are able to replicate and form microcolonies that eventually mature into biofilms. 20 Once formed, biofilms protect the constituent bacteria from antibiotics and host immune responses. 24 Spread to the urinary bladder or to other parts of the Foley catheter is achieved by shedding of actively dividing bacterial cells and shearing of biofilm aggregates from the mature biofilm. 13 Within the biofilm, there is exchange of genetic material between the bacterial cells that is greater than planktonic cells, thereby allowing potential spread of antibiotic resistance genes.9,20
In human patients, several methods have been suggested to prevent CAUTI. Attempts have been made to improve Foley catheter design material, including the use of silicone, polyurethane, composite biomaterial, or hydrogel-coated materials, but no single biosurface has been found to be effective enough in preventing biofilm formation.7,18,24 Other reported methods for preventing CAUTI in human patients include the use of Foley catheters impregnated with antibiotics or antiseptics such as silver compounds 15 and the use of probiotics such as nonpathogenic strains of E. coli 83972 to prevent colonization by uropathogenic bacteria.6,12 Although not demonstrated on Foley catheters in other veterinary species, urinary catheters coated with sustained-release varnish of chlorhexidine were effective enough in reducing biofilm formation and crystal formation in dogs. 22 In vitro studies demonstrated reduction of biofilm formation by sustained-release varnish of chlorhexidine on urethral catheters 23 and ureteral stents. 29 Studies evaluating effectiveness of sustained-release varnish of chlorhexidine on biofilm formation on Foley catheters are warranted.
Several noteworthy limitations of our study exist and may call into question the significance of the identified isolates and the resistance patterns reported herein. Isolation of a bacterium with a resistance pattern does not necessarily lead to cystitis. Urine samples were not collected from the urinary bladder prior to the placement of the Foley catheters to determine if bacteria isolated were already present in the upper or lower urinary tract prior to Foley catheter placement. Additionally, no follow-up urine cultures were performed when antibiotics were discontinued, and thus the resolution of a resistant bacterial infection could not be determined. Finally, in vitro antibiotic susceptibility may not indicate in vivo efficacy of an antibiotic when considering the presence of biofilms. 28
Goats undergoing tube cystostomy to manage obstructive urolithiasis may be predisposed to CAUTI. In our study, the major pathogens isolated from urine collected from Foley catheters were predominantly gram-positive commensal pathogens, with Aerococcus spp. and Enterococcus spp. isolated most frequently. Approximately 41.8% of the bacterial isolates were resistant to at least 2 antibiotic classes. Aerococcus spp., E. coli, and P. aeruginosa isolates had a significantly high probability (range: 0.59–0.76) of being resistant to ampicillin, ceftiofur, erythromycin, penicillin, or tetracycline.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
