Abstract
Effective control and the eventual eradication of Bovine viral diarrhea virus (BVDV) from cattle populations depend on the accurate identification of infected animals. Although typically a disease agent of cattle, BVDV is known to infect a wide variety of nonbovine species, including sheep. However, validation of serologic tests in these nonbovine species, particularly sheep, is lacking. We analyzed 99 sheep sera (57 samples from Pestivirus-naive sheep, and 42 samples from BVDV-inoculated sheep) in order to investigate 3 serologic tests: the agarose gel immunodiffusion (AGID) and 2 commercial enzyme-linked immunosorbent assays (ELISAs) for detection of BVDV antibodies. At the manufacturer’s cutoff thresholds, the AGID performed with 95.2% diagnostic sensitivity; ELISA-A performed with sensitivity of 90.5% and ELISA-B with 69.1%. All 3 tests performed with 100% diagnostic specificity. Two-graph receiver operating characteristic analysis showed that performance characteristics were optimized, such that both diagnostic sensitivity and diagnostic specificity were >95% for both ELISAs, if the thresholds were altered to 34.9% inhibition for ELISA-A and 63.5 signal-to-noise ratio for ELISA-B.
Bovine viral diarrhea virus (BVDV; family Flaviviridae) is a virus of the Pestivirus genus, which also includes Border disease virus (BDV) in sheep and Classical swine fever virus in pigs. 2 BVD is a highly prevalent disease of cattle that is associated with immunosuppression 4 and reproductive losses. 7 It is widely acknowledged that BVDV has significant economic impacts on cattle industries, with estimates ranging from US$40,000 to US$100,000 per infected herd per year. 9 Several studies have shown that systematic control or eradication of BVDV is economically beneficial in endemically infected cattle populations.8,13 Control of BVDV is largely reliant on testing for BVDV-specific antibodies, virus, viral RNA, or viral antigen. Detection of the virus (or its subunits) is critical for the identification of persistently infected (PI) individuals, which are epidemiologically important for the maintenance and spread of the virus. 3 The detection of specific antibodies indicates seroconversion in response to a previous infection in unvaccinated animals. Serologic tests for BVDV antibodies enable naive and exposed herds to be identified. Animals testing negative in a seropositive herd are consistent with PI animals and may warrant further testing for the presence of the virus.
In Australia and New Zealand, there are a number of serologic tests available for the identification of BVDV-specific antibodies in cattle, including agarose gel immunodiffusion (AGID), virus neutralization test (VNT), and enzyme-linked immunosorbent assay (ELISA; Australian and New Zealand Standard Diagnostic Procedures. Ruminant pestivirus infections, 2006, https://goo.gl/NwD5xN). 20 Although it is still thought of as the gold standard for identification of BVDV infection, VNT is relatively expensive and labor intensive when compared to ELISA and AGID, which are rapid and inexpensive, even for large-scale testing. One commercial ELISA has been validated relative to the VNT and was shown to have excellent performance on cattle sera, with diagnostic sensitivity (DSe) of 96.7% and diagnostic specificity (DSp) of 97.1%. 10 That same study showed that the performance of the AGID did not differ significantly from that of the ELISA. 10
Although BVD is most well known as a disease of cattle, many studies have identified infection in a wide range of other ruminant and pseudoruminant species, including sheep, 21 goats, 1 deer, 15 and alpaca. 6 Similarly, BDV infection has been reported in cattle, with a bull in New Zealand identified as being persistently infected with BDV. 14 Infection with BVDV in sheep is of particular importance given their proximity to cattle in some commercial production systems, and the reproductive losses that have been observed following infection of pregnant ewes with BVDV. 5 For example, abortion rates of 52–100% have been observed in ewes infected with BVDV at 25–100 d gestation,5,16,22 and the birth of BVDV PI lambs has also been reported.5,22
In Australia and New Zealand, there is little information available regarding the current state of infections with pestiviruses in sheep. The only known study regarding the prevalence of pestiviruses in sheep in Australia is a postal survey in 1979, which identified only 4% of flocks with Pestivirus infection. 11 However, the prevalence of BVDV in cattle is reported to be >86% in Australia (Taylor L. Findings of an Australia wide serological survey of beef and dairy herds for bovine viral diarrhoea virus conducted between 2007 and 2009. Australian Cattle Vets 2010;57) and 63% in New Zealand. 12 It has also been identified that only the BVDV-1 genotypes can be found in this region,11,23 with a predominance (96%) of BVDV-1c in Australia. 18 It is clear that there is a gap in the knowledge regarding Pestivirus infections in sheep, but, in order to accurately undertake a prevalence survey of this region, the correct detection techniques need to be identified. Therefore, we sought to optimize 3 serologic assays: the AGID and 2 commercially available ELISAs for the detection of BVDV-specific antibodies in sheep serum.
Materials and methods
Animals
Fifty-four sheep were used in our study: 25 wethers and 29 ewes; all were Merino × Border Leicester ewes and wethers from the University of Adelaide’s commercial flock at its Roseworthy Campus, South Australia. The flock is known to be Pestivirus naive based on previous testing. All animal experiments were approved by the University of Adelaide Animal Ethics Committee prior to this project (S-2012-240 and S-2012-248).
Sera
Blood samples were collected from all 54 sheep at the beginning of the project. The naive status was confirmed in all of the assays used in our study. Forty-two of the sheep were then given subcutaneous inoculations of 2 mL of freeze–thawed serum previously collected from a South Australian BVDV PI cow. Serum from the PI cow was typed as BVDV-1c. 5 Three sheep were not inoculated, although remained with the flock; the remaining 9 sheep were excluded from further participation in the project for practical reasons.
Blood samples were collected 6 wk postinoculation from the 42 inoculated sheep and from the 3 sheep that were not inoculated. Therefore, in total, 57 blood samples were collected from Pestivirus-naive sheep, and 42 samples from BVDV-inoculated sheep. All blood samples were centrifuged at 3,000 × g for 20 min, and serum was aliquoted into 1.2-mL microtubes before storage at −80°C until assayed.
Detection of BVDV antibodies using AGID and ELISA
The 99 serum samples were tested using an AGID a assay that uses the C24V reference strain (BVDV-1a) 19 as the antigen, with a score of 0 considered negative and scores of 1–3 considered positive. The AGID is recognized to be group reactive and capable of detecting antibodies to both BVDV and BDV (https://goo.gl/15GnYh).
The serum samples were also tested using 2 commercial BVDV antibody ELISAs. Samples were analyzed by the first ELISA (A) b as per the manufacturer’s instructions where results were expressed in percentage inhibition (%Inh). The manufacturer’s recommended cutoff for positive samples was >50%Inh. This ELISA has been stated by the manufacturer to detect antibodies to BVDV-1a, -1b, and -2.
Samples were also analyzed by a second ELISA (B), c wherein results were expressed as signal-to-noise ratio (S/N). The manufacturer’s recommended cutoff for positive samples for that assay is ≤40 S/N (note: as this is a blocking ELISA, low S/N values signify a positive result). This ELISA has been stated by the manufacturer to detect antibodies to both BVDV and BDV.
Statistical analysis
The performance characteristics of the AGID and both ELISAs were calculated using EpiTools (https://goo.gl/NCMJU0). The ELISA results were dichotomized using the manufacturer’s recommended cutoffs, and the true exposure status (inoculated or uninoculated) was used as the reference status. A confidence level of 95% was used.
A 2-graph receiver operating characteristic (TG-ROC) analysis (https://goo.gl/LG9XZx) was performed on the data from each ELISA. Data from the blocking ELISA-B were transformed using the following formula: 100 – (S/N). From the TG-ROC analysis, optimal cutoff values for maximizing test efficiency (minimizing misclassification) or maximizing Youden J statistic were determined, and the corresponding test performance characteristics, with confidence intervals, were calculated. Cutoff values for ELISA-B were extracted from the TG-ROC analysis in the 100 – (S/N) manipulated form, then back-calculated and are presented here as S/N.
Pairwise 2-tailed z-tests (https://goo.gl/xDBbGz) were performed to examine statistically significant differences in performance characteristics between tests, and before and after optimization. A p value of <0.05 was considered significant.
Results
The baseline samples from the 57 uninoculated sheep all tested negative for BVDV antibodies in all 3 assays at the manufacturer’s cutoffs. Conversely, of the 42 exposed sheep, 27 of these tested positive on each of the 3 assays and all but 1 (41) sheep were found to be positive in at least 1 of the assays. The 3 assays (interpreted at manufacturer’s cutoffs) all performed with 100% DSp but lower DSe (Table 1), with ELISA-B showing significantly lower sensitivity than the AGID (p = 0.0018) and ELISA-A (p = 0.0146).
Performance characteristics of the agarose gel immunodiffusion (AGID) and 2 enzyme-liked immunosorbent assays (ELISAs) at the manufacturer’s cutoffs.*
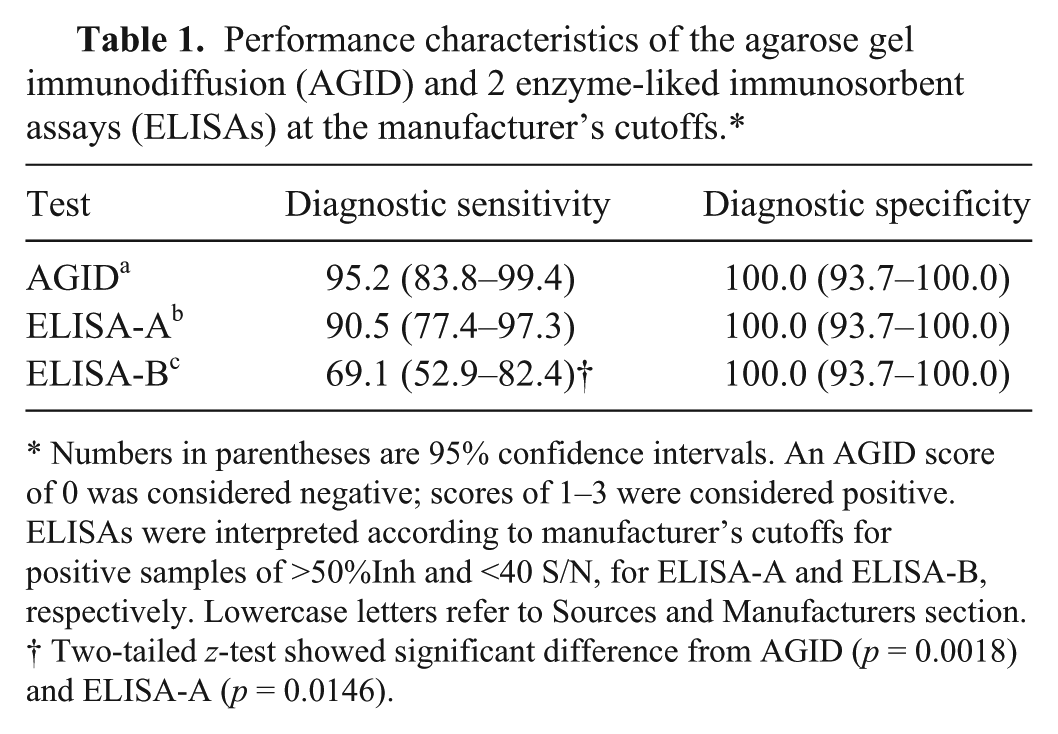
Numbers in parentheses are 95% confidence intervals. An AGID score of 0 was considered negative; scores of 1–3 were considered positive. ELISAs were interpreted according to manufacturer’s cutoffs for positive samples of >50%Inh and <40 S/N, for ELISA-A and ELISA-B, respectively. Lowercase letters refer to Sources and Manufacturers section.
Two-tailed z-test showed significant difference from AGID (p = 0.0018) and ELISA-A (p = 0.0146).
At the manufacturer’s cutoff value for each of the 3 tests, DSp was optimal (100%) but DSe was limited, particularly for ELISA-B (Table 1). AGID had the highest sensitivity (95.2%), followed by ELISA-A (90.5%) and ELISA-B (69.1%).
The TG-ROC analysis identified 2 possible optimal cutoffs for each of the ELISAs, in order to maximize test efficiency (minimize misclassification) and maximize Youden J statistic, respectively (Figs. 1, 2). For ELISA-A, DSe improved at both optimized cutoffs relative to the manufacturer’s cutoff, but the improvements were not statistically significant. No changes in DSp were observed for ELISA-A. For ELISA-B, significant improvements in DSe were observed at both the adjusted cutoff for maximal test efficiency (p = 0.0005) and the cutoff for maximized Youden J statistic (p < 0.0001), with no significant reductions in DSp. The optimal cutoffs and corresponding test characteristics are presented in Table 2. There were no significant differences in performance characteristics among the 3 assays (AGID, ELISA-A, and ELISA-B) when the optimal ELISA thresholds were used.
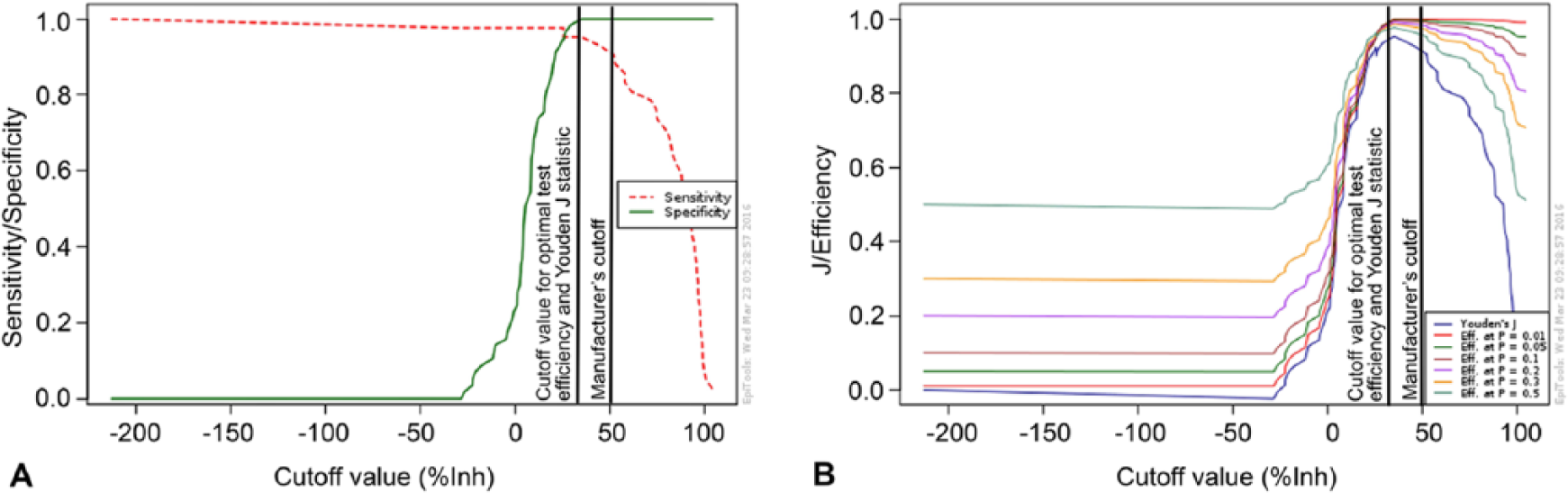
Output of a 2-graph receiver operating characteristics analysis (
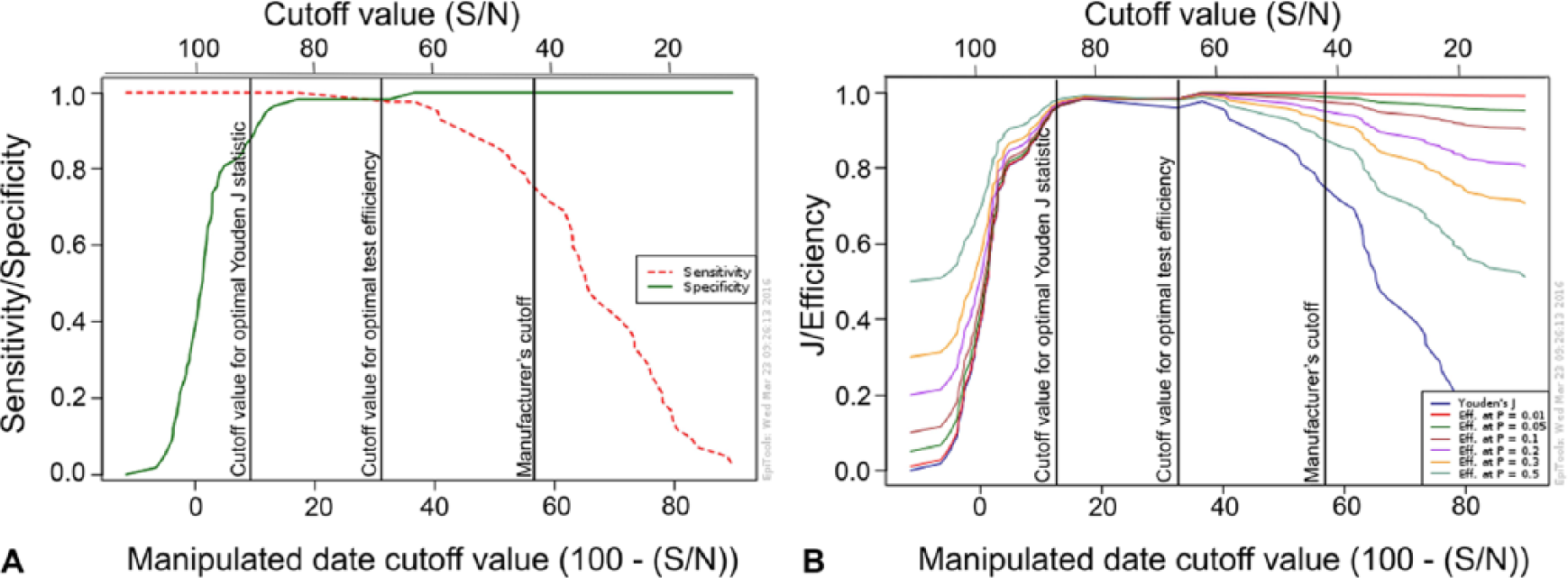
Output of a 2-graph receiver operating characteristics analysis (
Optimal cutoff values as determined by 2-graph receiver operating characteristic analysis for maximizing test efficiency (minimizing misclassification) and maximizing Youden J statistic and the corresponding test performance characteristics for ELISA-Ab and ELISA-B.c,*
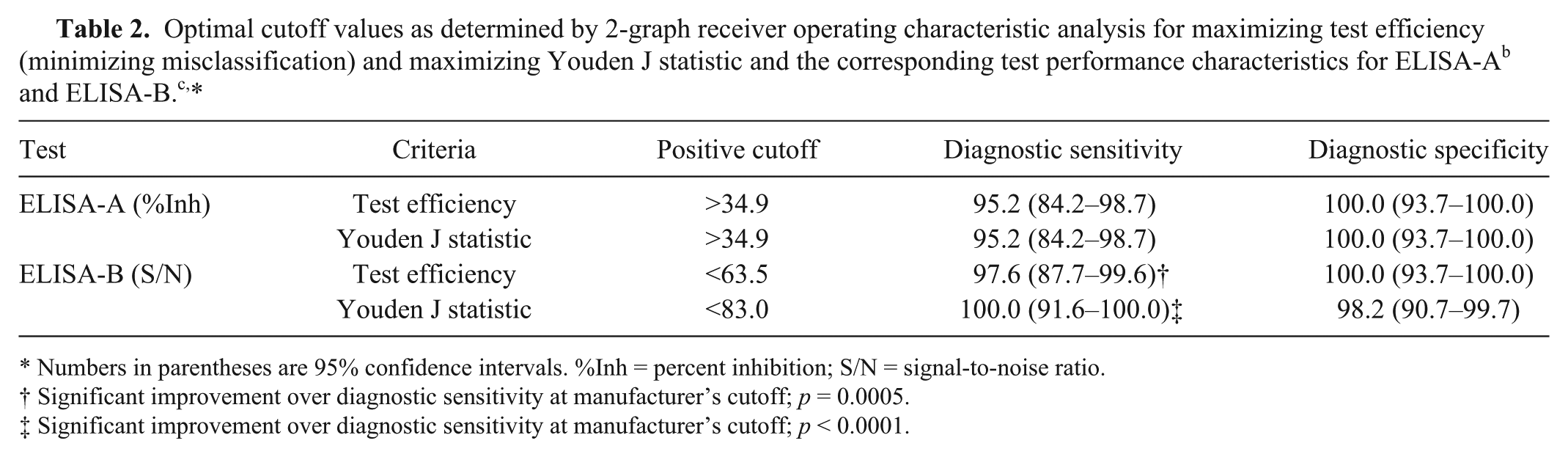
Numbers in parentheses are 95% confidence intervals. %Inh = percent inhibition; S/N = signal-to-noise ratio.
Significant improvement over diagnostic sensitivity at manufacturer’s cutoff; p = 0.0005.
Significant improvement over diagnostic sensitivity at manufacturer’s cutoff; p < 0.0001.
Discussion
In order to improve the DSe of both ELISA kits, TG-ROC analysis was undertaken. TG-ROC results for ELISA-A indicated that by lowering the manufacturer’s cutoff from 50%Inh to 34.9%Inh, specificity remained optimal but sensitivity increased from 90.5% to 95.2% for both test efficiency and Youden J statistic. In comparison, TG-ROC analysis for ELISA-B returned different cutoff values for test efficiency and Youden J statistic. Results of the TG-ROC analysis indicated that in order to increase test efficiency and minimize the number of misclassified samples, increasing the manufacturer’s cutoff from 40 S/N to 63.5 S/N maintained an optimal specificity while increasing sensitivity from 69.1% to 97.6%. However, in order to maximize the Youden J statistic, the manufacturer’s cutoff had to be increased to 83 S/N. At this cutoff, DSe was 100%; however, specificity decreased from 100% to 98.2%. Both identified cutoff values for ELISA-B are valid options.
Which cutoff value is preferable is dependent on the purpose of the test and the costs associated with false-negatives relative to false-positives. If the accurate identification of seropositive individuals is the objective, then the 83 S/N cutoff is appropriate wherein sensitivity is maximized. By contrast, if the accurate identification of negative animals is critical—for example, if antibody screening is being used for identification of negative individuals that are potential PI animals as a precursor to virus testing—then specificity is critical and the 63.5 S/N is appropriate. The correct identification of these individuals is critical in most control and eradication programs, as the elimination of a PI animal removes the most epidemiologically important source of infection. 17 However, in areas that use vaccination, PI animals may be antibody-positive as a result of an immunologic response to a heterologous strain. Therefore, caution should be taken when interpreting results from herds that use vaccination as a control method.
The AGID and 2 commercial ELISAs assessed in our study have useful performance characteristics for detection of BVDV-specific antibodies in sheep sera. We suggest that in order to optimize performance characteristics to >95% for both DSe and DSp of both ELISAs, the thresholds be altered to 34.9%Inh for ELISA-A and 63.5 S/N for ELISA-B. It is, however, important to be aware of limitations in the ability of serologic tests to discriminate between Pestivirus species. To ensure that the identification of BVDV-infected or non-infected sheep is correct, further work is required on the cross-reactivity of specific antibodies against other Pestivirus species, particularly BDV, with the assays used in this study.
Footnotes
Acknowledgements
We thank John Matheson, Grant Jarvis, Caitlin J. Jenvey, and Brenden Johansson for their technical assistance throughout this project.
Authors’ contributions
CA Evans and SR Lanyon contributed to acquisition, analysis, and interpretation of data, and drafted the manuscript. MP Reichel contributed to analysis and interpretation of data. All authors contributed to conception and design of the study, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
AGID, Elizabeth Macarthur Agriculture Institute (EMAI), New South Wales, Australia.
b.
PrioCHECK BVDV Ab P80 ELISA, Prionics, Martinsried, Germany.
c.
BVDV p80 protein antibody test kit, IDEXX Laboratories, Rydalmere, New South Wales, Australia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the University of Adelaide, with IDEXX ELISA kits donated by IDEXX Laboratories Inc.
