Abstract
Forty-seven cattle management groups from 36 herds in a regional Bovine viral diarrhea virus (BVDV) eradication program were selected to evaluate serology as a tool to detect herd infection with BVDV. Serum samples were obtained from 5 non-vaccinated sentinel calves ≥ 6 months old in each management group and virus neutralizing (VN) antibody titers against BVDV genotypes 1 and 2 were determined. A herd was considered positive if 2 or more sentinel calves had VN antibody titers ≥ 128 to either genotype. Results were compared to individual animal testing of all available calves by reverse transcription polymerase chain reaction (RT-PCR) on skin biopsy samples. In 1 management group from 1 herd (n = 24), 3 sentinel calves had VN antibody titers ≥ 128. Three ear notch samples from that herd were positive for BVDV on RT-PCR assay. All other management groups were negative for BVDV. In the present study, the herd sensitivity of sentinel serology was 100% (95% confidence interval [CI]: 0.05–1.0) and herd specificity was 100% (95% CI: 0.90–1.0). The κ value for agreement between sentinel serology and RT-PCR was 1.0 (95% CI: 1.0–1.0). Preliminary results suggest that sentinel animal serology can be utilized in a BVDV eradication program to provide an accurate and efficient evaluation of herd status.
Control of Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) has been a worldwide challenge for decades. The most well-known source for transmission of BVDV are persistently infected (PI) cattle. 7,8,13 Persistent infection occurs when calves are exposed to BVDV in utero before the fetal immune system is developed, thus allowing the developing fetus to become immunotolerant to the virus. 4 Persistently infected cattle continuously shed large amounts of virus into the environment and are an important source of virus transmission within and between herds. Because of the clinical and economical importance of BVDV, many European countries (including Denmark, Sweden, Norway, Finland, Austria, Germany, and Switzerland) have implemented BVDV eradication programs. 11,12,15 In Michigan, the Upper Peninsula BVDV Eradication Program was launched in 2008 with the goal of eradicating BVDV from a region within the United States. This integrated program includes education, identification, and elimination of persistent infections, as well as vaccination and biosecurity.
A key step in eradicating BVDV from a herd is identification and removal of all PI cattle. Multiple diagnostic tests have been developed to identify PI cattle including virus isolation (VI), antigen capture enzyme-linked immunosorbent assay (ACE), immunohistochemistry (IHC), and reverse transcription polymerase chain reaction (RT-PCR). Unfortunately, whole-herd testing using any of these tests can be a major investment of both time and money for producers. Herd screening tests are desirable for detection of BVDV infection at the herd level before embarking on individual animal testing.
Serological evaluation of small groups of young non-vaccinated calves using virus neutralization (VN) or enzyme-linked immunosorbent assay has been proposed as a method for identifying herds with PI cattle. 5,6,9,10,16,18 The objective of the current study was to evaluate the application of sentinel serology as a screening tool to detect herd infection with BVDV as part of an eradication program in the Upper Peninsula of Michigan.
Thirty-six herds (mean herd size = 109; range: 17–371) consisting of 47 management groups were included in the study. Herds enrolled in the Michigan Upper Peninsula BVDV Eradication Program were asked to voluntarily participate and agree to a sentinel testing scheme as well as a retest of the whole herd in 2009. Twenty-nine herds had tested negative for BVDV in 2008, 4 herds had at least 1 calf persistently infected with BVDV diagnosed in 2008, and 3 herds were new to the study in 2009. Thirty-four of the herds were beef, 1 was dairy, and 1 was mixed dairy and beef. Some (n = 6) of the 36 herds had multiple management groups, which were defined as groups of animals on the farm that were managed separately from other animals or groups.
Ear skin biopsy (ear notch) samples were collected over the course of 2009 from 2,206 calves shortly after birth or at weaning by use of a 0.794-cm ear notcher and then placed in individually labeled bags. These samples were placed on ice or in a refrigerator for storage before being submitted within 1 week of collection to the Michigan State University Diagnostic Center for Population and Animal Health (DCPAH; Lansing, Michigan). Serum samples were collected from 5 non-vaccinated calves ≥ 6 months old in each herd or, in the case of herds with multiple management groups, each management group within a herd. A total of 395 serum samples were collected in the summer and fall of 2009. Serum samples were collected from the oldest calves in each management group based on information provided by the producer.
Skin biopsy samples were analyzed at DCPAH using a pooled (n ≤ 10 ear notches per pool) RT-PCR assay; a hydrolysis probe-based real-time RT-PCR assay with an internal RNA control to monitor inhibition of the reaction was used to detect RNA from BVDV. The BVDV-specific primers and probe were designed from conserved sequences within the 5’-untranslated region (UTR). The assay amplifies both BVDV genotypes 1 and 2 (BVDV-1 and -2, respectively) based upon testing of a validation panel provided by Dr. J. Ridpath (National Animal Disease Center [NADL], Ames, Iowa). 17 The detection limit of the assay is 0.8 50% tissue culture infective dose (Wise AG, Wu P, Benson C, et al.: 2006, A TaqMan-based real-time RT-PCR with internal control for the detection of BVD virus in ear notch samples. In: Proceedings of the 49th annual meeting of the American Association of Veterinary Laboratory Diagnosticians, p. 35. Abstract). If a pool of ear notches was positive, individual samples contained in the pool were retested using a gel-based RT-PCR that amplified a region of the 5’-UTR that spanned base 139–394 of the genome of the BVDV-NADL reference virus. Any animal positive on initial testing was retested a minimum of 14 days later to confirm persistence of the virus. This was done by collecting a blood sample and performing virus isolation and subsequent genetic sequencing of the virus.
Serum samples were tested for VN antibodies against BVDV-1 and -2 using standard microtitration assay procedures (Carbrey E, et al.: 1971, Recommended standard laboratory techniques for diagnosing infectious bovine rhinotracheitis, bovine viral diarrhea and shipping fever (parainfluenza-3). In: Proceedings of the 75th annual meeting of the U.S. Animal Health Association, pp. 629–648. Abstract). Cytopathic Singer strain was used as the BVDV-1 reference strain and cytopathic 125C strain as the BVDV-2 reference strain. The VN test was conducted using bovine turbinate cells that were free of adventitious BVDV. 1-3 Fetal bovine serum supplement for cell culture medium was also free of live adventitious BVDV, RNA from BVDV, and antibody against BVDV. Serial 2-fold dilutions ranging from 1:4 to 1:4,096 were made for each sample of serum. Antibody titer was considered to be the highest serum dilution at which the cytopathic effect of the BVDV reference strain was completely inhibited.
A herd was considered to be infected with BVDV if 2 out of 5 calves in any management group had VN antibody titers ≥ 128 for either BVDV-1 or -2. Conversely, if 4 out of 5 calves in all management groups had VN titers ≤ 64 to both BVDV-1 and -2, the herd was considered negative for BVDV. A herd was also considered positive if any animal in any management group was found to be positive for BVDV by RT-PCR assay on skin samples.
Information from each herd collected included number of management groups, herd history, and previous BVDV test results. Data was described using descriptive statistics. The sensitivity and specificity of the sentinel calf testing strategy was determined using results of the RT-PCR assay on ear notches as the gold standard. Correlation between the RT-PCR assay and sentinel animal serology as herd-based tests was calculated using the κ test statistic.
Ear notches from 3 calves from 1 herd (n = 24) that was new to the study in 2009 were positive for BVDV. Those calves were confirmed to be persistently infected with BVDV on follow-up testing. BVDV genotype 1a was isolated from all 3 calves. The same management group was also positive for BVDV based on VN titers of ≥ 128 from 3 outof 5 sentinel calves for BVDV-1 and -2. The other 46 management groups tested did not contain PI cattle based on either RT-PCR or VN results (Table 1).
Distribution of Bovine viral diarrhea virus 1 and 2 (BVDV-1 and -2, respectively) virus neutralization (VN) titers in sentinel calves from each herd.*
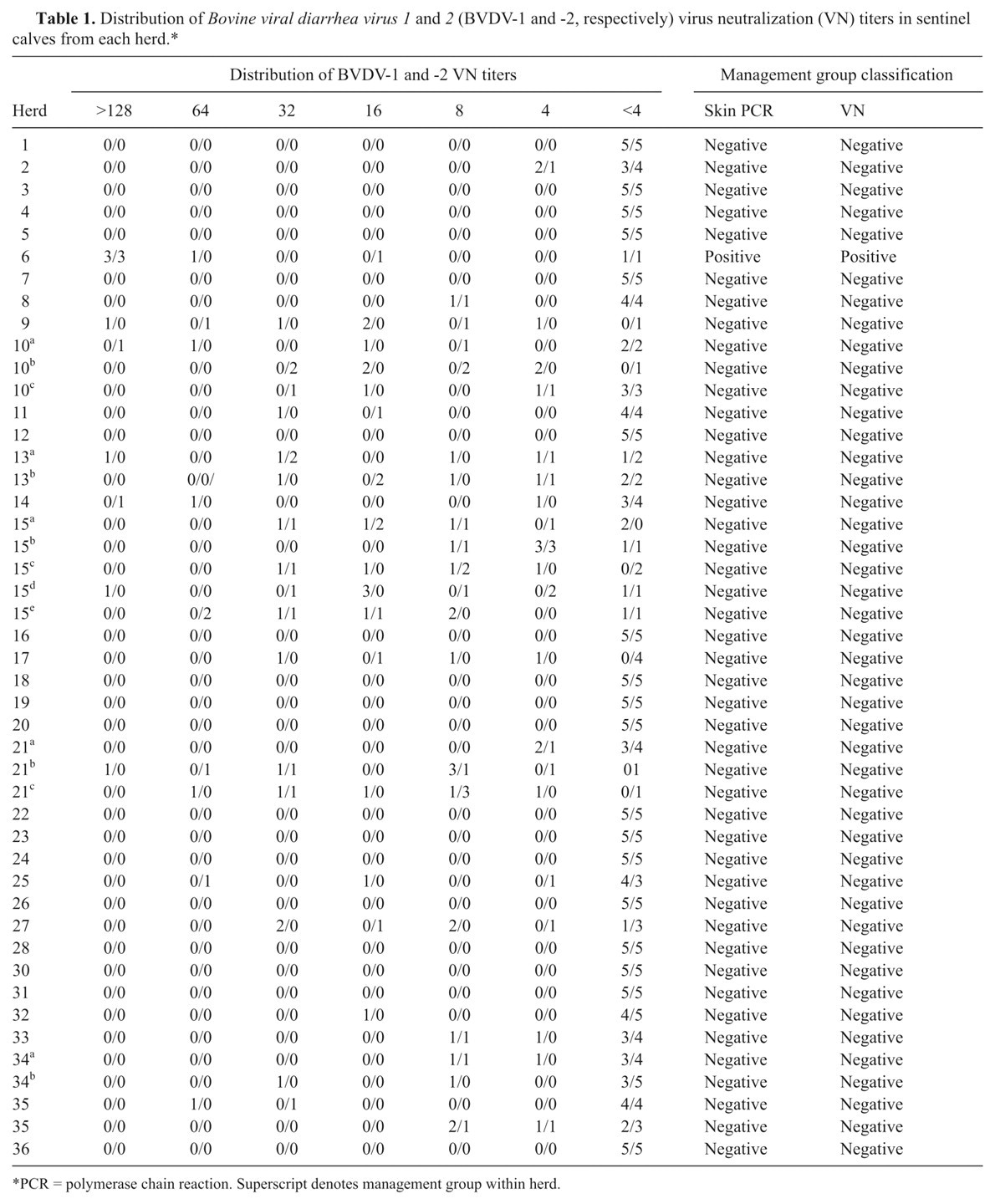
PCR = polymerase chain reaction. Superscript denotes management group within herd.
Virus neutralization results from non-vaccinated sentinel calves from each management group were compared with the results for herd testing using RT-PCR. The herd sensitivity for VN testing was 100% (95% confidence interval [CI]: 0.05–1.0) and herd specificity was 100% (95% CI: 0.90–1.0). The κ value for agreement between the 2 tests was 1.0 (95% CI: 1.0–1.0). To determine the influence of changing the VN cutoff value or the number of calves above or below the VN cutoff value on the accuracy of this strategy, results of serologic testing from each management group were compared with various cutoff values for number of positive calves and minimum antibody concentration. As the number of positive calves and minimum antibody concentration were lowered, the diagnostic specificity of the test decreased (Table 2). The sensitivity remained unchanged throughout.
Sensitivity and specificity of serological evaluation of virus neutralization antibodies against Bovine viral diarrhea virus (BVDV) for detection of herd infection with BVDV using different diagnostic criteria and when compared with pooled reverse transcription polymerase chain reaction of skin biopsy samples.
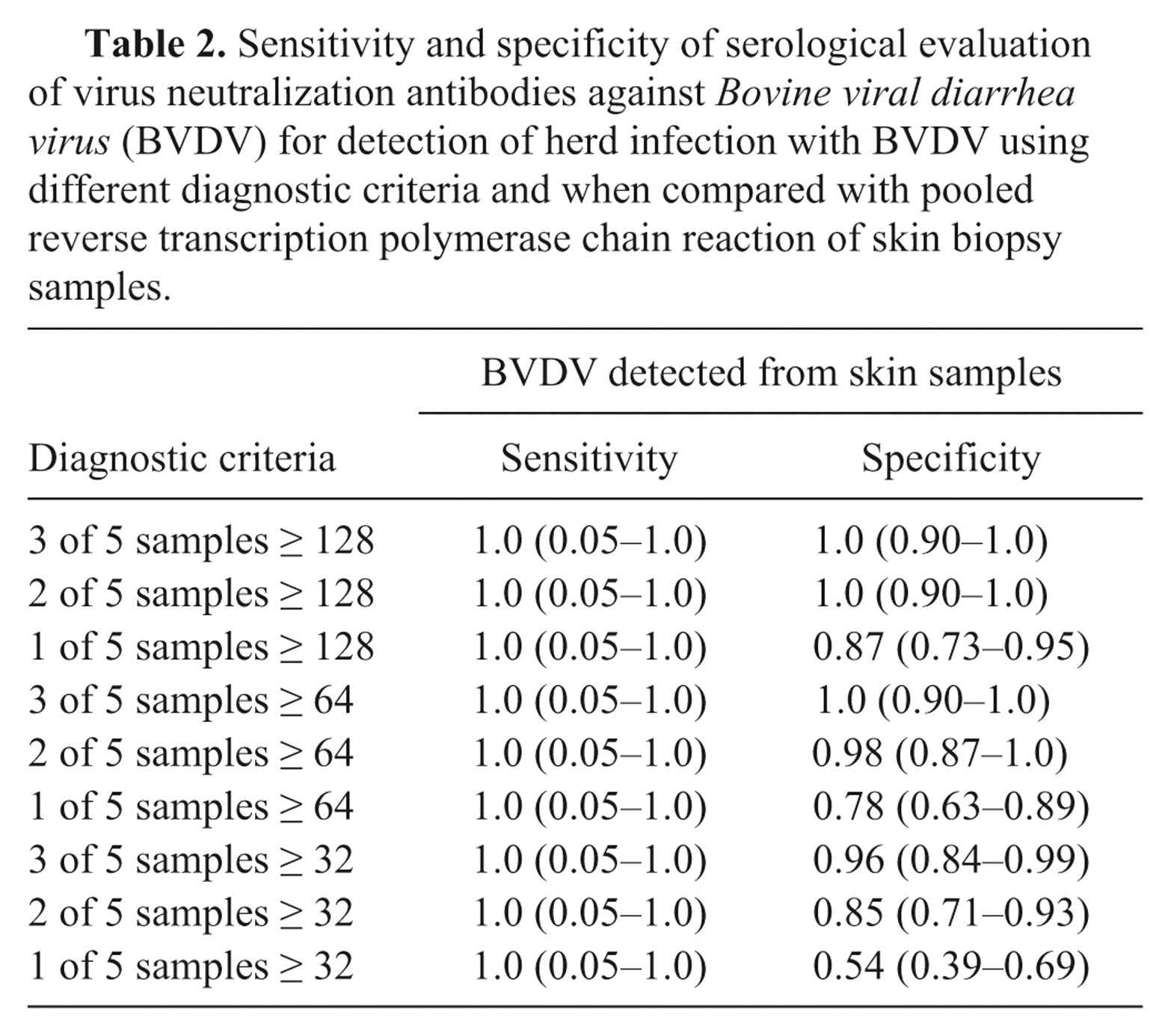
The results from the present study support and extend earlier studies that found sentinel serologic testing useful for screening herds for infection with BVDV. 9,16,18 The premise is that a high prevalence of seropositive calves in a herd is indirect evidence that a PI animal is present. 10 The calves serve as sentinels for virus being shed by PI herd mate(s). Calves selected as sentinels should be non-vaccinated and ≥ 6 months old because colostral antibodies may be present in the serum of calves < 6 months old. In the current study, serological evaluation of 5 non-vaccinated calves per management group was an accurate herd screening method for predicting the presence of PI cattle in a herd, or just as importantly, the lack of PI animals in the herd. There was 100% correlation between detection of PI cattle in a herd using PCR on ear notches and the predicted presence of PI cattle in a herd using sentinel serology. However, since only 1 management group was positive in the study, the sensitivity cannot be accurately assessed. Previous studies have reported lower sensitivity than in the current report. 16,18 One study done in beef cattle evaluated use of 10 non-vaccinated calves per herd. 18 A herd was classified as likely to contain PI cattle when at least 3 out of 10 calves at weaning had VN titers ≥ 1,000; by this measure, the sensitivity for use of VN titers in sentinel beef calves to detect the presence of a PI animal in a herd was 53% and specificity was 80%. Another group found a sensitivity of 66% and a specificity of 100% in dairy herds when using a diagnostic criterion of 3 out of 5 calves with VN antibody titers ≥ 128. 16 If researchers had changed their diagnostic criterion to 2 put of 5 calves, the sensitivity would have improved to 83% (Pillars RB, 2001: Serological evaluation of 5 unvaccinated heifers to detect herds with cattle persistently infected with bovine viral diarrhea virus. MS Thesis. Michigan State University, East Lansing, MI). In that previous study, the diagnostic cutoff criterion was selected based on cost. It was determined that the cost of extensive testing associated with a false-positive result was of more concern than the cost of a false-negative result. Changing the diagnostic criteria used to define a positive herd would alter the sensitivity and specificity of the test. Sensitivity can be improved by decreasing the positive threshold VN antibody titer. Sensitivity can also be improved by decreasing the number of sentinel calves that must have antibody titers above a designated cutoff. Since the goal of surveillance in an eradication program is to identify any and all potentially positive herds, a premium is placed on sensitivity. Decreasing the number of calves that needed to have a minimum titer of ≥ 128 from 3 to 2 was previously shown to increase sensitivity. 16
Reasons for lowered specificity of sentinel serology discussed in previous studies include vaccination of sentinel calves, persistence of passively acquired antibodies, and acute infections introduced by breakdowns in biosecurity. To address these potential problems, the target population for the current study was unvaccinated calves that were 6–12 months of age. By testing calves older than 6 months of age, the risk of detecting passively acquired antibodies is decreased significantly as these antibodies are usually undetectable by 6 months of age. 7 In addition, all herds involved in the present study were implementing biosecurity measures to reduce the risk of herd infection. This included reducing contact with neighboring cattle farms and screening new cattle for BVDV. These practices help to reduce the risk of herd misclassification.
Herd size, multiple management groups, and low stocking rates have been cited in previous studies as reasons for lower sensitivity of sentinel serology as a herd-based assay for BVDV. Previous studies conducted in dairy herds showed that herd size did not impact the results of serologic testing and suggested that evaluation of 5 non-vaccinated calves would be effective for herds of varying size as long as the herd was managed as 1 unit with commingling of cattle of various ages. 7,9,10,18 Since most of the herds in the present study (35/36 herds) were beef, serologic testing of any and all management groups was done to increase the sensitivity of the test. In addition, low stocking rates should have been less of a factor, as herds are more contained in the Upper Peninsula of Michigan as compared to other parts of the United States. The average farm size in Michigan is 180 acres, and the average beef cattle herd size is 35 adult cows (http://www.nass.usda.gov/Statistics_by_State/Michigan/index.asp. Accessed on December 23, 2010). Beef cattle in the Upper Peninsula are typically calved in either January-February or April through June. Most calving occurs in confined calving areas to facilitate early management and to reduce risk of carnivorous attacks by coyotes. Cow–calf pairs are typically turned out onto pastures in late spring early summer and rotated through different pastures as needed. Because of the abundance of rain fall and forages in the Upper Midwest, stocking rates are typically higher than other parts of the country and will range from 2–3 acres/cow–calf pair (http://beefteam.msu.edu/LinkClick.aspx?fileticket=mvrM0SbeK4Q%3d&;tabid=550. Accessed on December 23, 2010). Weaning of calves most commonly occurs in September through November depending on feed reserves. During the grazing season, cattle may be grouped together for purposes of intensive grazing schemes, breeding synchronization, fly control, and vaccination of calves as part of reconditioning programs. Because of the higher stocking rates and more confined management that is common in the Upper Midwest, strategies using sentinel animals for detection of herd disease are more likely to be successful than if applied in other regions of the United States where cattle are managed at lower densities.
Serological testing is part of many European BVDV eradication programs. This is based on the high probability of seropositivity in groups of animals where PI animals are present. 7 The eradication programs in these countries do not involve the use of vaccines and calves selected for testing range from 6–12 months of age depending on that nation’s program. 11,13-15 The success of these national programs suggest that serological testing of young stock is a valid and useful screening method that can be applied to in an eradication program in the United States.
Results from the present study provide supportive evidence that serological evaluation of non-vaccinated calves can be utilized as a screening tool for herd infection with BVDV and could be employed as an accurate and economically viable surveillance tool to evaluate herds in a BVDV eradication program. Long-term evaluation of this strategy will provide more information on the usefulness of sentinel serology as a low-cost surveillance tool. Although the strategy was highly sensitive and specific in the herds assayed for the present study, application to herds in different parts of the country and using different management strategies should be evaluated.
Footnotes
Acknowledgements
The authors would like to thank Dr. Roger Maes and the Virology section at the Diagnostic Center for Population and Animal Health for performance of RT-PCR and VN assays.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Funding for this project was provided by the Michigan Agriculture Experiment Station and Pfizer Animal Health.
