Abstract
We evaluated the use of oral fluid as an alternative to serum samples for Classical swine fever virus (CSFV) detection. Individual oral fluid and serum samples were collected at different times post-infection from pigs that were experimentally inoculated with CSFV Alfort 187 strain. We found no evidence of CSFV neutralizing antibodies in swine oral fluid samples under our experimental conditions. In contrast, real-time reverse transcription–polymerase chain reaction could detect CSFV nucleic acid from the oral fluid as early as 8 d postinfection, which also coincided with the time of initial detection in blood samples. The probability of CSFV detection in oral fluid was identical or even higher than in the corresponding blood sample. Our results support the feasibility of using this sampling method for CSFV genome detection, which may represent an additional cost-effective tool for CSF control.
Keywords
Classical swine fever (CSF) is an acute viral infection of pigs and wild boar that causes major economic losses especially in countries with dense populations of domestic pigs. 6 The disease is caused by species Classical swine fever virus (CSFV; genus Pestivirus, family Flaviviridae). 20 Although eradicated in many areas of the world, epizootics in disease-free regions occur regularly, and CSF continues to cause serious problems in various parts of the world. The disease is currently endemic in Asia, areas of Central and South America, and in some Eastern European countries; sporadic occurrence in Western Europe has been observed. Early detection is the key to ensuring that control measures will minimize the spread and impact of such a devastating disease. 14 Vaccination against CSFV was banned in the European Union (EU) at the end of the 1980s, and the current control strategy is based on strict stamping out. Council Directive 2001/89/EC has permitted the use of emergency vaccinations 8 only under exceptional circumstances and following an approved emergency plan.
CSFV antibodies may represent good markers of infection. Antibody assays are economical, compatible with automation, and suitable for high-throughput screening. Therefore, antibody testing may be the best option for CSFV surveillance. The limitation of this approach is the expense of collecting and testing blood samples.
Oral fluid (OF) samples contain serum transudate, saliva, and buccopharyngeal secretions from the animal. 25 It is possible to use OF as diagnostic samples because serum transudate contains a variety of pathogens and antibodies from the animal. The veterinary literature reports the presence of antibodies, pathogens, and acute phase proteins in OF. 19 Infectious agents, cortisol, acute phase proteins, and progesterone have been detected in OF samples of swine in both experimental and field conditions. 11 Furthermore, a variety of pathogens and pathogen-specific antibodies have been demonstrated in swine OF, 19 including Bovine viral diarrhea virus, Foot-and-mouth disease virus (FMDV), Porcine circovirus 2, Porcine reproductive and respiratory syndrome virus, pseudorabies virus (Suid herpesvirus 1), Vesicular stomatitis virus, and Influenza A virus.1,2,16–18,21,23
The ‘‘rope-in-a-bait’’ sampling method has been proven to detect CSFV infection in wild boars experimentally infected, although the animals had a very short viremic phase and virus excretion via saliva was very poor. 13 A subsequent study 9 provided evidence of simultaneous detection of African swine fever virus, CSFV, and FMDV in swine OF by multiplex real-time reverse transcription–polymerase chain reaction (rtRT-PCR).
There is very little experimental information available concerning the use of OF samples for the diagnosis of CSF, especially with regard to antibody detection. The disease is predominantly septicemic, and the main portal of entry for pathogenic virus is the nasopharynx; therefore, early after infection, the virus is found in the tonsils. 12 Induction of an immune response in the upper respiratory tract can be expected to result in early resolution of the viral infection. Nevertheless, the correlation between detection results in serum and OF of individual animals is currently insufficiently addressed. However, such a correlation is important for a better understanding and interpretation of results obtained during pen-based sampling.
Therefore, it was our objective to compare the detection of CSFV antibodies and genomic material by virus neutralization test (VNT) and rtRT-PCR in serum and OF samples collected from individually housed animals after experimental infection with CSFV.
Eight 2-mo-old crossbred pigs were obtained from a commercial local supplier. The pigs were clinically healthy and free of antibodies against pestiviruses. They were housed in high-containment animal facilities and were fed twice a day with a diet for weaning pigs and water ad libitum. The maintenance and experimental protocols were established according to the European legislation on the protection of animals used for scientific purpose. 7 The experiments were carried out under the approval of the Italian Ministry of Health (678/2015-PR). Before starting the experiment, the pigs were acclimated for 7 d. During this period, the animals were trained to chew a rope before feeding.
All pigs were infected intranasally with CSFV Alfort 187 at a 50% tissue culture infectious dose (TCID50) of 103/pig in 2 mL of Eagle minimum essential medium. Clinical signs and rectal temperatures were evaluated daily for 51 d. OF, serum samples, and heparinized blood were collected from each animal at 0, 3, 8, 10, 15, 17, 22, 24, 30, 37, 45, and 51 d postinfection (dpi) for serologic and virologic investigations. The serum samples were stored at −20°C. OF and peripheral blood mononuclear cells (PBMCs) collected from heparinized blood were stored at −80°C until tested. All the animals were subjected to an autopsy.
OF were collected from each animal using a cotton rope 12 mm in diameter and 25 cm in length. a Before feeding, each animal chewed a cotton rope for 10–15 min until the rope was sufficiently wet. The wet end of the rope was cut, inserted into a 50-mL syringe, and compressed to recover OF. A volume of 3–5 mL was obtained from each pig on each collection day. Later, the OF was centrifuged at 400 × g for 10 min at 4°C, and the supernatant was stored at −80°C until the tests were performed.
To date, there are no CSFV antibody ELISAs available for OF matrix, therefore serum samples and OF were tested using the VNT, performed according to the World Organization for Animal Health (OIE) protocol. 24 Viral RNA was extracted from both OF and PBMCs with a commercial kit. b According to the manufacturer’s recommendation, any DNA contamination by host cells was previously removed by filtration and centrifugation (1,500 × g for 10 min) of OF samples. Subsequently, viral RNA was detected using primers and FAM-labeled probe according to the rtRT-PCR protocol described previously. 10 The assay amplified a 90-nucleotide fragment of the envelope 2 (E2) glycoprotein.
Seven animals had an increase in temperature >40°C at 8 dpi that lasted 6 d. The following clinical signs were observed during the infection period: fever, anorexia, piling into a corner, apathy, constipation, and diarrhea. One subject was ataxic. In general, anorexia and apathy were concomitant with high fever. Notably, no erythema, hemorrhages, or petechiae of the skin were observed. Five pigs died at 11–35 dpi; the remaining 3 animals were euthanized at 51 dpi in good condition. Postmortem examination was performed on all pigs. Animals had generalized lymphadenopathy; the mandibular, lateral, and medial retropharyngeal lymph nodes were the most affected. Splenic infarcts and petechiae in different organs were only observed on a few occasions. The most commonly observed lung lesion was acute bronchopneumonia.
Neutralizing antibody activity was detected in serum at 10–15 dpi when 6 pigs seroconverted, displaying titers between 1/5 and 1/640. Two pigs that died at 15 and 22 dpi did not seroconvert. When the OF samples were tested, no antibody reactivity was seen by VNT (Table 1).
Detection of Classical swine fever virus (CSFV)–specific antibodies by virus neutralization test in serum and oral fluid samples of 8 pigs experimentally infected with the CSFV Alfort 187 strain.*
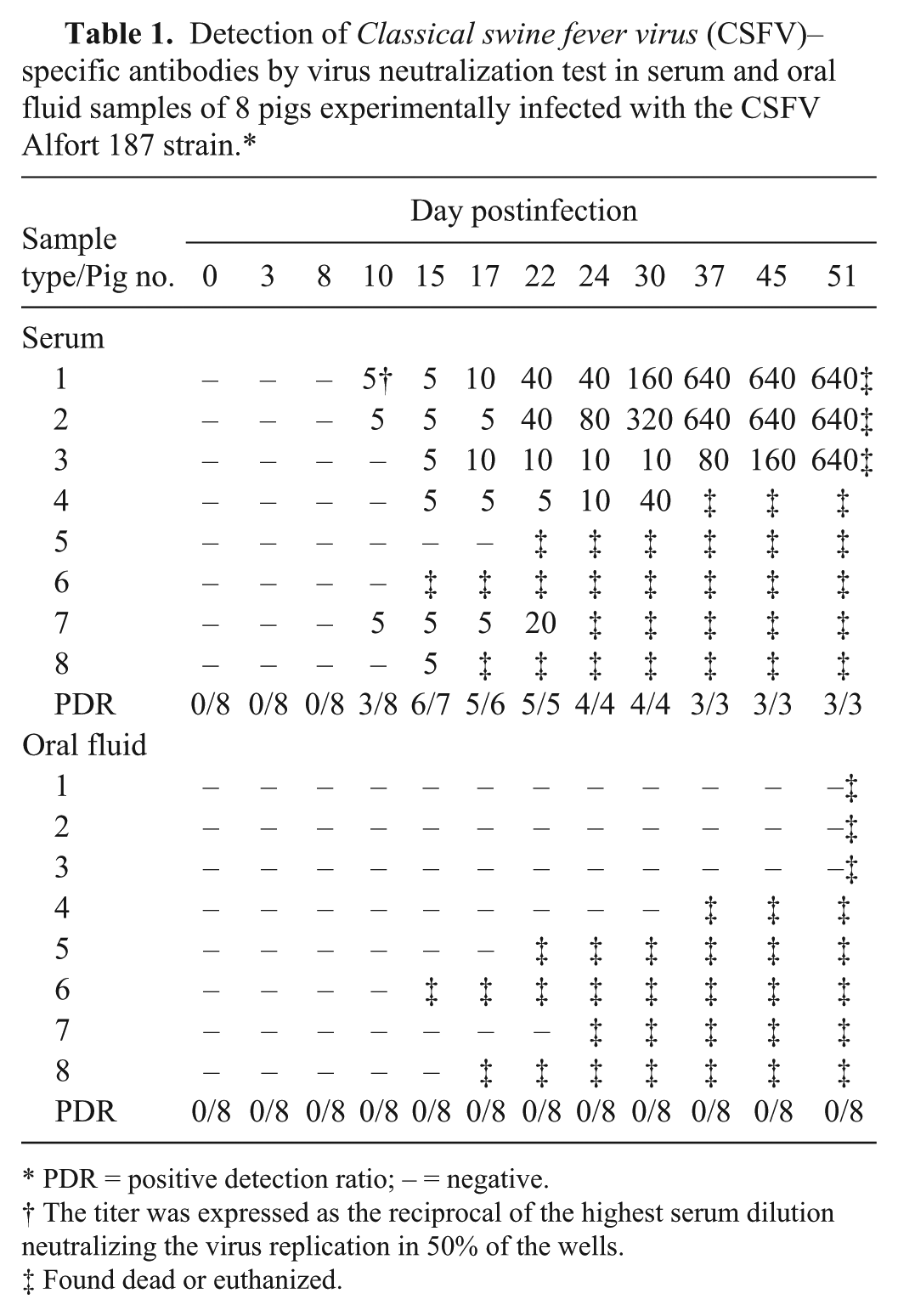
PDR = positive detection ratio; – = negative.
The titer was expressed as the reciprocal of the highest serum dilution neutralizing the virus replication in 50% of the wells.
Found dead or euthanized.
The first positive results on rtRT-PCR were detected at 8 dpi from blood and OF samples of 3 and 4 inoculated animals, respectively (Table 2). The other animals developed a positive reaction at 10–22 dpi. Interestingly, 3 OF samples were rtRT-PCR positive 5–8 d before the corresponding blood samples; 1 OF sample remained positive 13 d longer (17–30 dpi), although with very high threshold cycle (Ct) values. In general, Ct values of blood samples were significantly lower than those of OF, reflecting a high level of viremia. In contrast, the positive detection rate in OF was higher (27 of 67 = 0.40 compared with 19 of 67 = 0.28 in blood).
Results of Classical swine fever virus (CSFV) detection by real-time RT-PCR in peripheral blood mononuclear cell (PBMC) and oral fluid samples of 8 pigs experimentally infected with CSFV Alfort 187 strain.*
PDR = positive detection rate; – = negative.
Threshold cycle value.
Found dead or euthanized.
There has been increasing interest in the use of OF for the detection of various animal pathogens. Studies have demonstrated viral neutralizing antibody activity in human and animal OF samples.5,15,22 Despite having at least 3 animals with rather high neutralizing antibody titers (1/640) in our study, VNT failed to detect antibodies in OF samples. This could be attributed to a low antibody level in OF compared to the corresponding serum sample and/or to the influence of background effects, such as the hypotonic nature of saliva or innate antiviral substances that are present in oral fluid. 15 Both conditions may have affected the detection limit of the OIE serologic test used in our study, as it was not optimized for OF samples. The presence of antibodies in buccopharyngeal secretions can be a result of local antibody synthesis and/or passive antibody transudation from blood to local secretions. 4 Two major antibody classes in saliva are secretory IgA and IgG. 3 One can speculate that the antibody class eventually present at the time of sampling may have no neutralizing activity. In addition, it has been demonstrated that the strongest local response in OF was observed after intranasal inoculation with a high viral dose. 4 In our experiment, we inoculated pigs intranasally using a very small virus dose (1,000 TCID50/pig) because the goal of this proof-of-concept study was to extend the course of infection and ensure that the animals would seroconvert. It could be hypothesized that the local immune response was not stimulated adequately under such conditions, thus making it difficult to detect CSFV antibodies by conventional methods.
In any case, antibody detection only provides a retrospective analysis of the disease situation, but is not useful for early warning. Hence, we explored the efficiency of CSFV viral RNA detection in OF samples; rtRT-PCR was capable of detecting CSFV nucleic acid as early as 8 dpi, which also coincided with the first detection in blood samples. Surprisingly, the CSFV detection in OF was identical or even higher than the corresponding blood sample. Another finding was the higher detection rate, despite the fact that the amount of virus detected in PBMCs was significantly greater, as indicated by the lower Ct values of blood samples. No conclusive explanation about the origin of the viral RNA in OF at that time can be given. Nevertheless, because replication occurs in local tissues such as salivary glands and tonsils, our results suggest that they cannot be excluded as a potential source of CSFV.
Footnotes
Authors’ contributions
S Petrini and GM De Mia contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. I Pierini contributed to design of the study and to analysis of data. M Giammarioli contributed to conception and design of the study; contributed to acquisition and interpretation of data; and drafted the manuscript. F Feliziani contributed to conception of the study; contributed to interpretation of data; and drafted the manuscript. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Tego swine oral fluids, ITL Animal Healthcare, Malaysia.
b.
QIAamp viral RNA mini kit, Qiagen, Hilden, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Ministry of Health, RC IZSUM 12/2013 (D. Lgs. 502/92, art. 12; D. Lgs. 229/99, art. 12bis)
