Abstract
The reported prevalence of naturally occurring anti–dog erythrocyte antigen (DEA) 7 antibodies in DEA 7–negative dogs is as high as 50%. Characterization of these antibodies may better define their importance in canine transfusion medicine. We determined in vitro activity, specificity, and titer of anti–DEA 7 antibodies in DEA 7–negative dogs. Plasma samples from 317 DEA 7–negative dogs were cross-matched with DEA 7–positive red blood cells (RBCs) using gel column technology. Agglutination occurred with DEA 7–positive RBCs but not with DEA 7–negative RBCs in 73 samples (23%), which were hence classified as containing anti–DEA 7 antibodies. These samples were evaluated for hemolytic and agglutinating activity, strength of agglutination, and antibody specificity and titers. All samples showed agglutination but none showed hemolysis. Gel agglutination was graded as 1+ for 20 samples (27%), 2+ for 49 samples (67%), 3+ for 4 samples (6%); no samples were graded 4+. The agglutination titer was <1:2 for 51 samples (73%), 1:2 for 13 samples (19%), 1:4 for 4 samples (5%), and 1:8 for 2 samples (3%). Of 16 samples treated with 2-mercaptoethanol, 11 samples (69%) contained only IgM, 4 samples (25%) exhibited only IgG activity, and 1 sample (6%) had both IgG and IgM activity. Low titers of warm, weakly agglutinating, mostly naturally occurring IgM anti–DEA 7 antibodies were found in 23% of DEA 7–negative dogs. The presence of naturally occurring anti–DEA 7 antibodies suggests that cross-matching of canine blood recipients is advisable, even at first transfusion, to minimize delayed transfusion reactions.
Introduction
The presence of a number of canine blood groups and natural blood group antibodies has a significant impact on canine blood transfusion medicine. As the average life span of pet dogs increases, repeated blood transfusions to the same animal become more likely, and the importance of determining blood groups and blood compatibility grows. The presence and potential activity of antibodies against blood type antigens has little consequence to a surgical patient with normal hematology, but may be significant in transfusion-dependent patients, such as those with severe aplastic anemia, myelodysplastic syndromes, and other congenital or acquired chronic anemias (such as immune-mediated hemolytic anemia) who require frequent and long-term transfusion support. 16
Dog erythrocyte antigen (DEA) 7 is not an integral antigen of the canine red cell membrane, but is produced elsewhere in the body in soluble form, secreted into the plasma, and is adsorbed onto the cell membrane. DEA 7 is structurally related to a common bacterial antigen, 4 and consists of 3 distinct bands with molecular weights of 53, 58, and 66 kD. 5 The reported prevalence of DEA 7 varies in the range of 6–82% in various canine populations.2,3,8,11,15,17,18,21,23,24
The American Association of Blood Banks Standards defines a clinically significant antibody as one that causes decreased red blood cell (RBC) survival. 6 The characterization of anti–DEA 7 antibodies may help to better define their role and importance in canine transfusion medicine. Although clinical significance is often predicted by evaluating serology, these tests do not always distinguish between clinically significant and clinically benign antibodies. 20 Naturally occurring canine anti–DEA 7 antibodies (alloantibodies) have been identified in up to 50% of all DEA 7–negative dogs that have never received transfusions,2,8,12,25 and have been implicated in causing delayed transfusion reactions through clearance of incompatible transfused RBCs.9,10,16,22 In addition, anti–DEA 7 antibodies can be produced by isoimmunization. 3 DEA 7–negative dogs without anti–DEA 7 antibodies receiving unmatched DEA 7–positive blood at a first blood transfusion can be sensitized and have a delayed transfusion reaction at the second unmatched DEA 7 blood transfusion.
Little is known about the specificity and activity of anti–DEA 7 antibodies. Determination of the characteristics of naturally occurring anti–DEA 7 antibodies may help in assessment of the risk of transfusion reactions following unmatched transfusions in any dog population, in deciding which blood typing and compatibility tests to perform before a blood transfusion, and in selection of the most suitable blood donors. We determined the activity, specificity, and titer of naturally occurring anti–DEA 7 antibodies in DEA 7–negative dogs.
Materials and methods
Plasma samples were collected from 317 DEA 7–negative (and DEA 4–positive, DEA 1–negative or positive) canine blood donors from the Veterinary Transfusion Unit (REV) of University of Milan, Italy; these dogs had never received a blood transfusion. The dogs included purebreds and cross-breeds, some of which had been included in previous studies23,25; others were specifically tested for DEA 7 for this research. Owner consent was obtained both for blood collection, as part of the evaluation of the dogs before inclusion in the voluntary canine blood donation program, and for the use of the surplus blood samples in this study. Based on University of Milan animal use regulations, formal ethical approval was not needed as dogs were sampled with the informed consent of the owners during routine visits.
Alloantibody screening—testing for antibodies in an animal’s serum or plasma using different RBC suspensions of known blood type—is already performed in veterinary medicine.1,25 Initially, plasma from all DEA 7–negative dogs was screened against DEA 7–positive, DEA 1–negative, and DEA 4–positive RBCs. All tests were performed using gel column technology a as described previously.1,25 This technique uses low-ionic-strength salt solution (LISS) for preparation of red cell suspensions for cross-matching. LISS increases the rate of antigen–antibody complex formation and thus enhances antigen–antibody reactions. Additionally, because antibody uptake is increased, incubation times of antigen–antibody reactions can be reduced. 6 Briefly, 0.8% RBC-LISS suspension was obtained by adding 10 μL of packed RBCs of DEA 7–positive, DEA 1–negative, and DEA 4–positive RBCs to 1 mL of modified LISS. b Twenty-five microliters of plasma from each DEA 7–negative dog and 50 μL of 0.8% RBC-LISS suspension were mixed in the reaction chamber of the gel column a and incubated at 37°C for 15 min. Gel columns were centrifuged in a special column gel card centrifuge c for 10 min and examined for signs of hemolysis (based on the macroscopic color of the plasma samples) and for agglutination. The strength of agglutination was scored from 1+ to 4+ according to the manufacturer’s instructions. 13 Agglutination ≥1+ was considered positive for the presence of antibodies. Autocontrols (i.e., patient plasma incubated with each patient’s own RBCs) were also performed with each cross-match test to exclude the presence of autoantibodies. Positive agglutination reactions were verified in duplicate by 2 operators. Plasma samples that showed hemolytic or agglutinating reactions were retested against a second panel of DEA 7–negative, DEA 1–negative, and DEA 4–positive RBCs to identify the specific antigen associated with alloantibody production. These plasma samples were further analyzed to characterize the specificity and titer of the anti–DEA 7 antibodies.
The agglutinin titer of antibodies is defined as the highest dilution of plasma in which agglutination against DEA 7–positive RBCs can still be detected. This was determined by creating 2-fold serial dilutions (starting from 1:2) of the plasma sample in phosphate buffered saline solution up to the highest dilution at which agglutination could be detected.1,6,8 The gel column cross-match test was then repeated using these serodilutions. The various suspensions were incubated at 37°C for 15 min and evaluated for the presence and strength of agglutination as described above.
The specificity of antibodies (i.e., IgG vs. IgM) was measured by treating the plasma samples with an equal volume of 0.1 M 2-mercaptoethanol and incubating at 37°C for 60 min. 2-Mercaptoethanol abolishes agglutination and complement-binding activities of IgM antibodies (by cleaving their disulfide bonds), allowing IgG antibodies to be detected. 6 After incubation, the agglutinin specificity was determined based on the presence or absence of agglutination as described above.
Results
A total of 73 samples (73 of 317, 23%) produced agglutination when cross-matched with DEA 7–positive RBCs, but not with DEA 7–negative RBCs, and these were designated as source samples containing anti–DEA 7 antibodies. These samples were derived from 40 Spanish Greyhounds, 20 Italian Corsos, 3 German Shepherd Dogs, 3 Rhodesian Ridgebacks, 3 Italian Hounds, 2 Doberman Pinschers, 1 Bernese Mountain Dog, and 1 Irish Wolfhound.
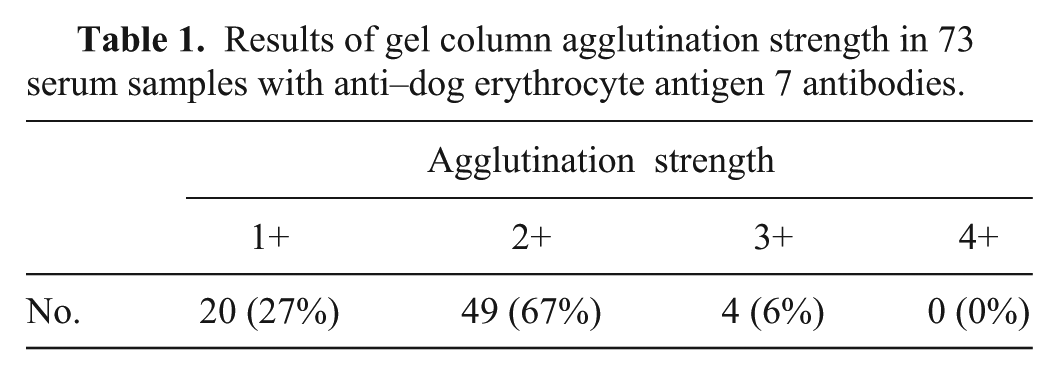
All 73 samples with anti–DEA 7 antibodies showed agglutination, but none showed hemolysis (Tables 1, 2). Sixteen samples were treated with 2-mercaptoethanol, and 11 samples were found to contain only IgM, 4 samples had only IgG activity, and 1 sample had both IgG and IgM activity. In most samples, determination of specificity was not possible because the agglutination titer was <1:2. For 3 samples, there was insufficient volume for titer and determination of specificity.
Results of gel column agglutination strength in 73 serum samples with anti–dog erythrocyte antigen 7 antibodies.
Titer of naturally occurring anti–dog erythrocyte antigen (DEA) 7 antibodies in 70 serum samples from DEA 7–negative dogs.

Discussion
Blood group antibodies (both naturally occurring and as a result of a prior red cell transfusion) could have pathologic effects that result in the destruction of allogeneic RBCs manifesting as a hemolytic transfusion reaction (HTR). The severity of the reaction can vary from mild, with reduced efficacy of the transfusion, to extremely severe, with rapid death of the recipient. 20 Antibody detection and identification are not only fundamental to transfusion practice but also provide information that aids in the selection of suitable blood for transfusion.
Serologic tests (e.g., antibody strength, mode of reactivity, thermal range, specificity, immunoglobulin class, affinity, and ability to bind complement) can be used to identify RBC antibodies and to determine characteristics that may indicate their clinical significance. The most important single result of serologic testing is the thermal amplitude of the antibody. If the antibody does not react at 37°C, it should cause no significant in vivo RBC destruction and should not produce immediate clinical effects. When alloantibodies are active at 37°C, they are potentially clinically significant, and reactions may vary from slightly decreased cell survival to clinically obvious reactions (e.g., jaundice). 7 In our study, the presence and activity of canine anti–DEA 7 antibodies was only evaluated at 37°C, to identify warm antibodies that are likely to be significant in vivo.
Other factors that can influence the pathologic effects of an antibody are the amount of antibody present, the quantity of IgG or IgM and/or complement bound to the red cell, and the presence of target antigen in tissues and/or body fluids. 20 In our study, anti–DEA 7 antibodies were mostly IgM (69%). IgM antibodies activate the classical complement pathway leading to formation of the membrane attack complex and puncture of the red cell membrane. Hemolytic transfusion reactions mediated by IgM occur in the intravascular space and are characterized by intravascular liberation of hemoglobin. The usual signs of HTR are chills, shock, hypotension, hemoglobinemia, and hemoglobinuria, which lead to the additional complications of disseminated intravascular coagulation and renal failure. 20 These reactions are rarely seen in dogs that have not been sensitized by previous incompatible blood transfusion, despite the fact that previous reports have shown the prevalence of naturally occurring anti–DEA 7 antibodies to be as high as 50%.2,8,11,25 A decline in packed cell volume several days after transfusion may not be recognized clinically because it may be masked by resolution of the underlying cause of anemia and the recipient’s red cell regenerative response. In addition, other factors that can influence the pathologic effects of an antibody are the quantity and distribution of target antigen on the red cell membrane. The low levels of antigen on DEA 7–positive red cells, or physical factors as yet undefined, result in only small amounts of antibody attaching to the red cells. 3
The anti–DEA 7 antibodies in our study showed no in vitro hemolytic activity. This could be because the plasma was derived from samples collected in EDTA. It is well known that neither of the major complement-activation pathways (i.e., the classical or alternative pathway), necessary for lytic activity of complement, can function in the presence of the metal chelator EDTA. 19
In addition, low titers of anti–DEA 7 antibodies were found in our study, <1:2 in most (73%) samples evaluated. This level may be too low to cause significant intravascular or extravascular hemolysis initially. However, we speculate that patients with low levels of naturally occurring anti–DEA 7 antibodies receiving an incompatible transfusion with DEA 7–positive blood will produce a secondary or anamnestic response. Delayed HTRs usually occur in patients previously immunized to the offending antigen, but in whom antibody levels have dropped to a level too low to cause significant intravascular or extravascular hemolysis (often too low to be detected serologically). Subsequent transfusion of red cells expressing the offending antigen initiates a secondary or anamnestic response. The transfused red cells will be removed from circulation after several days, with complete removal within 2 weeks of transfusion. 20
The main limitation of this study was that we did not demonstrate that the presence of anti–DEA 7 antibodies was correlated with an increased clearance of transfused DEA 7–positive RBCs in vivo. We were unable to do this for ethical reasons, as no DEA 7 incompatible blood units were transfused into the DEA 7–negative patients.
Throughout our study, the neutral gel agglutination assay was used for cross-matching. This blood typing and compatibility technology, widely used in human blood banking, was developed in 1985 in an attempt to achieve more stable agglutination reaction endpoints and to provide more reproducible results in comparison with traditional tube methodology. The procedures used in human tests are standardized and provide clear and stable reactions that improve result interpretation. The results from gel column tests can be saved for up to 24 h, and photographs can be recorded. 13 The use of the gel test for canine cross-matching in a previous study provided clear results, with high sensitivity and specificity when compared with tube agglutination techniques. 2 Based on the results of this and a previous study, 2 the advantages of the gel column technique apply to compatibility tests in canine transfusion medicine.
Based on the results of this study and, in the absence of in vivo clinical studies on the activity of anti–DEA 7 antibodies on incompatible transfused DEA 7–positive RBCs, cross-matching of canine blood recipients is advisable (even at first blood transfusion) to reduce the likelihood of a delayed transfusion reaction caused by anti–DEA 7 antibodies. This is of particular importance in patients with chronic anemia who require frequent and long-term transfusion support. For the same reasons, and based on knowledge of DEA 1 characteristics,8,9,10,14 ideal blood donors should be DEA 1– and DEA 7–negative.
Footnotes
Authors’ contributions
All authors contributed to the conception and design of the study; to acquisition, analysis, or interpretation of data; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. E Spada drafted the manuscript.
a.
ID-Card NaCl enzyme test and cold agglutinins, DiaMed GmbH, Cressier FR, Switzerland.
b.
ID-Diluent 2 (modified LISS solution), DiaMed GmbH, Cressier FR, Switzerland.
c.
ID-Centrifuge 24 S, DiaMed-ID micro typing system, DiaMed GmbH, Cressier FR, Switzerland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Piano di Sostegno alla Ricerca 2015-2016, Linea 2, University of Milan, Milan, Italy.
